Structure, Bonding & Reactivity of Alkenes
Lajoy Tucker
Teacher

Contents
Introduction
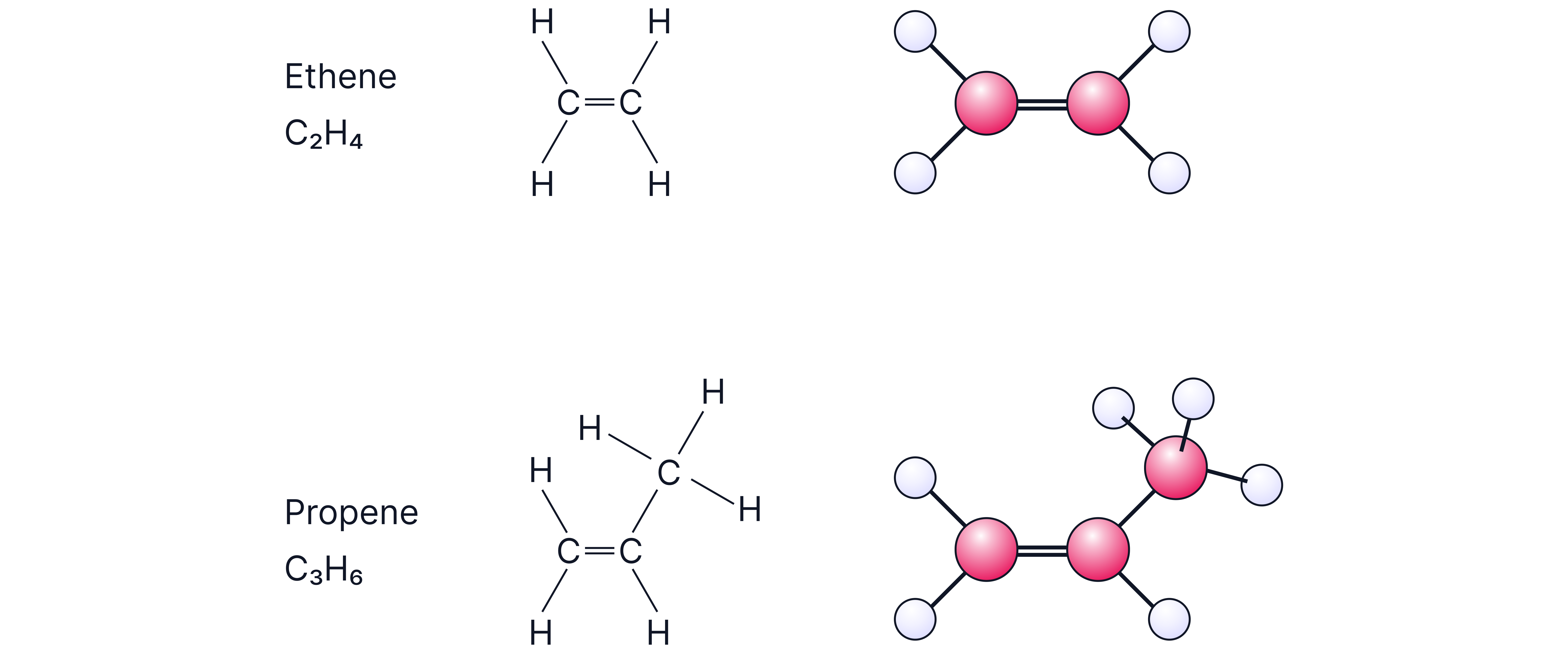
Alkenes are unsaturated hydrocarbons containing at least one C=C double bond.
General formula for acyclic alkenes: CₙH₂ₙ.
The C=C bond is the functional group and controls their chemistry.
Bonding in Alkenes
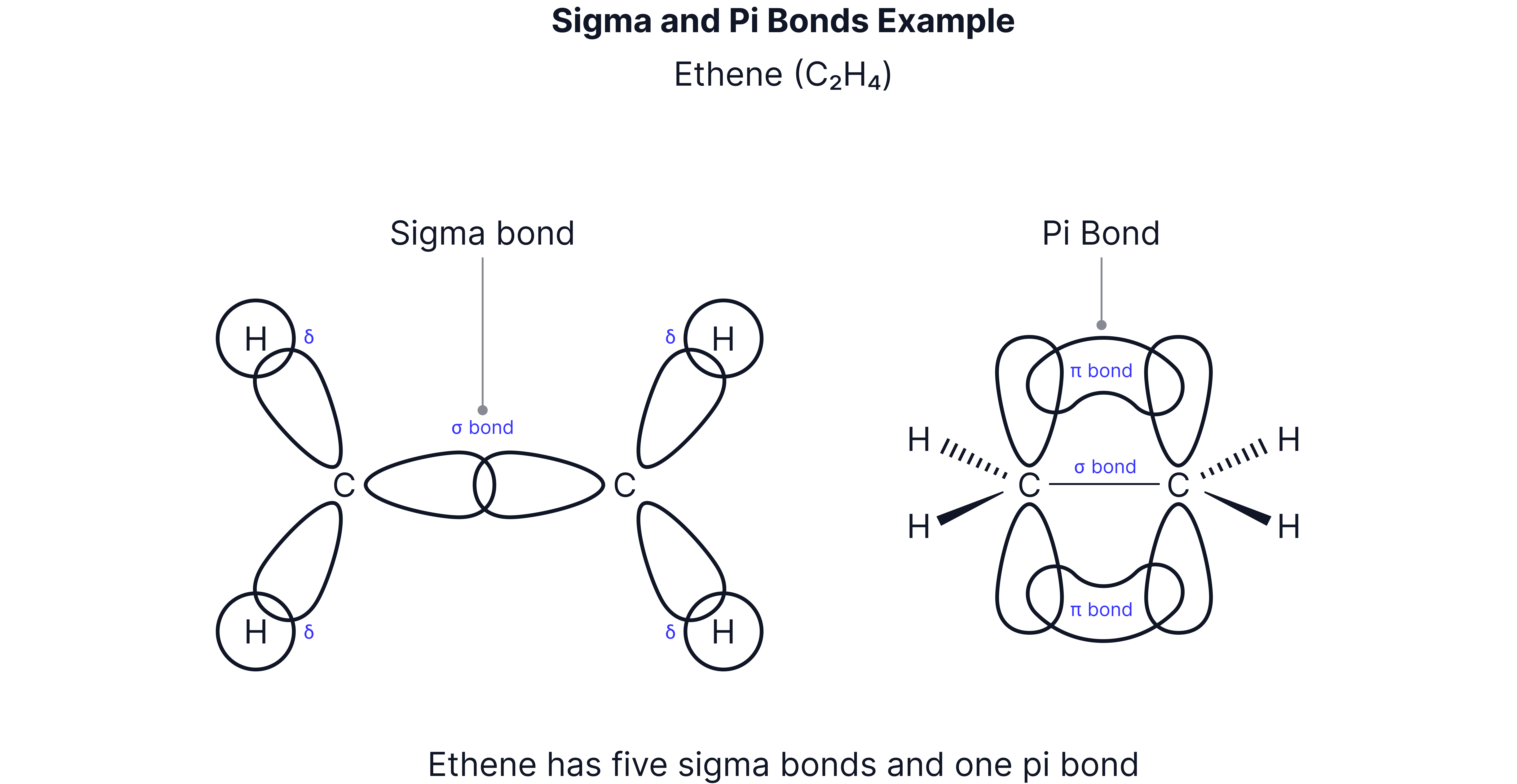
The C=C double bond is made of:
1 sigma (σ) bond – formed by direct overlap of orbitals.
1 pi (π) bond – formed by sideways overlap of p-orbitals.
The π bond lies above and below the plane of the carbon atoms.
This restricts rotation around the C=C bond.
Makes alkenes more reactive than alkanes.
Alkenes are susceptible to attack by electrophiles i.e. species that ‘want’ electrons.
Each carbon is bonded to three regions of electron density.
This results in trigonal planar geometry around eth carbon atom with a bond angle of ~120º.
The π bond restricts rotation, giving rise to stereoisomerism.
Step 1 – Electronic configuration of carbon
Ground state carbon: 1s² 2s² 2p².
To form four bonds, one electron from the 2s orbital is promoted into an empty 2p orbital → configuration becomes 1s² 2s¹ 2p³.
This provides four unpaired electrons, allowing four covalent bonds.
Step 2 – Hybridisation
In alkenes, carbon atoms undergo sp² hybridisation.
The 2s orbital mixes with two of the 2p orbitals → three sp² hybrid orbitals.
One p orbital remains unhybridised.
Step 3 – Sigma (σ) bonds
Each sp² orbital overlaps head-on with another orbital:
Two sp² orbitals overlap with hydrogen 1s orbitals (C–H σ bonds).
One sp² orbital overlaps with the other carbon’s sp² orbital (C–C σ bond).
This gives each carbon three σ bonds.
Step 4 – Pi (π) bond
The unhybridised p-orbitals on each carbon overlap sideways above and below the plane of the σ bonds.
This forms the π bond, the second part of the C=C double bond.
Isomerism in Alkenes
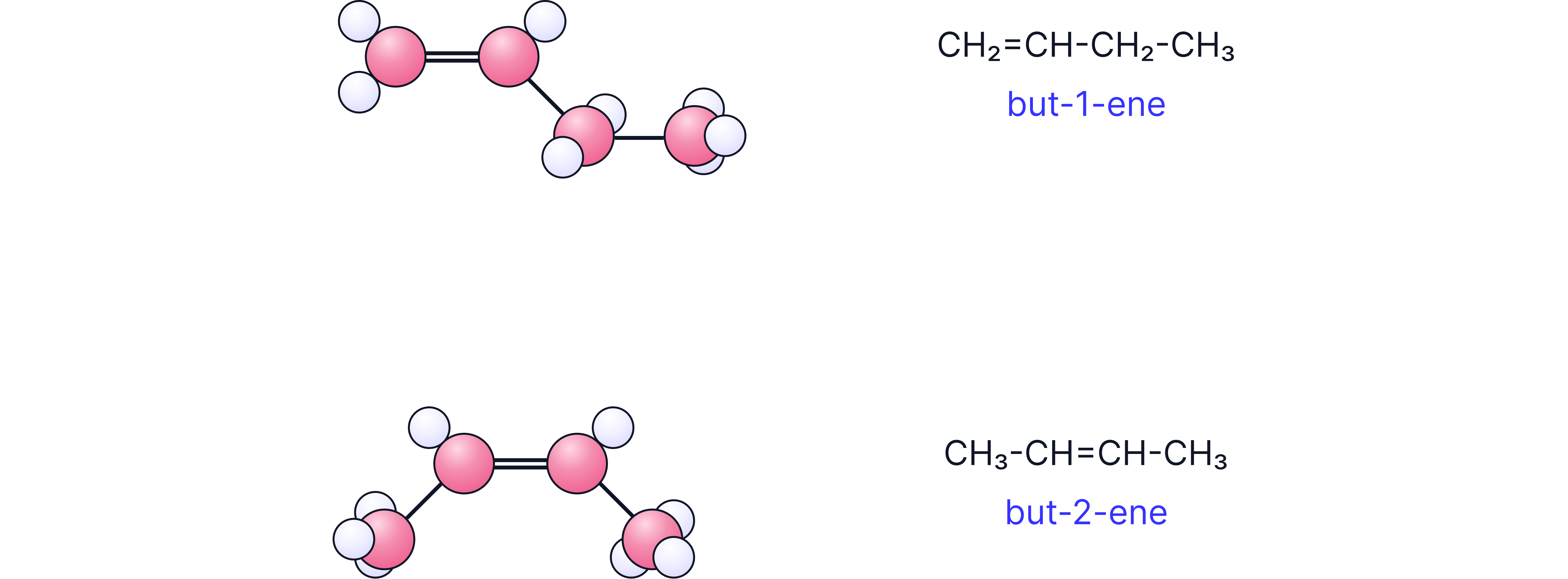
Structural isomerism
Same molecular formula, different atom arrangement.
Example: But-1-ene vs But-2-ene.
E-Z stereoisomerism
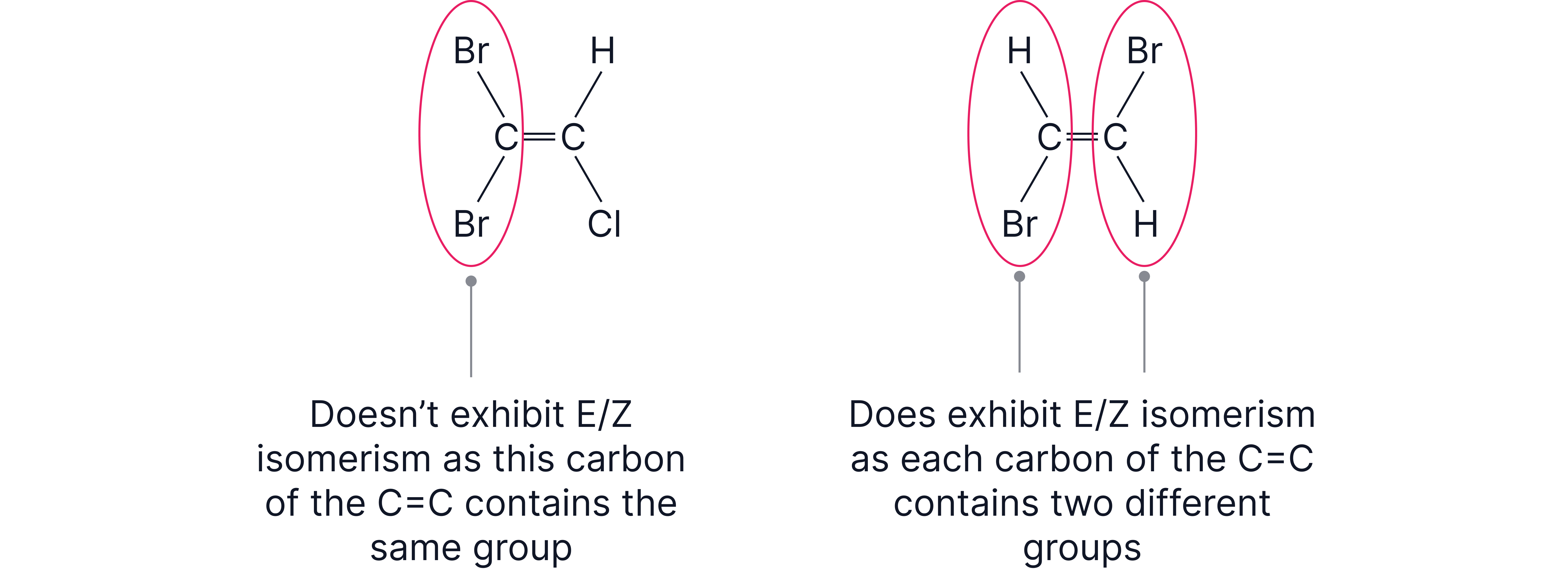
Stereoisomerism is where molecules have the same structural formula but a different arrangement of atoms in space.
Caused by restricted rotation around C=C
Occurs when different groups are attached to both carbon atoms of the double bond.
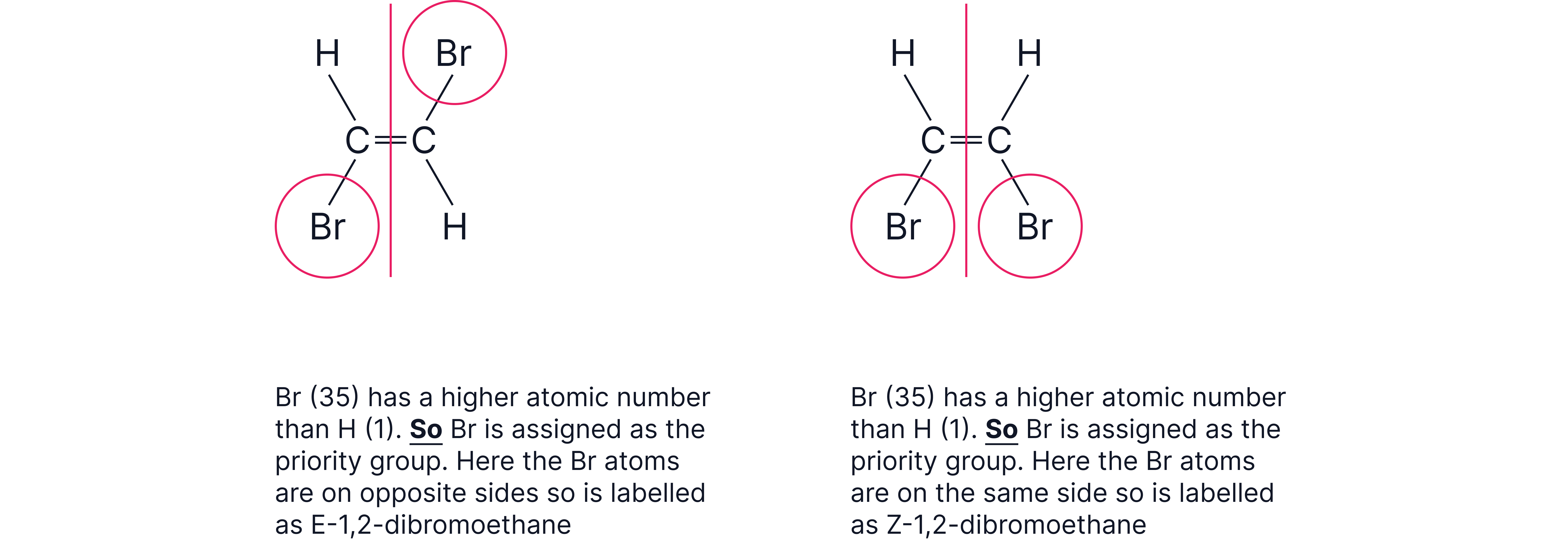
Naming rule:
Z (zusammen, “together”) – higher priority groups on same side.
E (entgegen, “opposite”) – higher priority groups on opposite sides.
Priority decided by atomic number (Cahn–Ingold–Prelog rules).
How to assign priorities (Cahn–Ingold–Prelog)
Look only at the atoms directly attached to each alkene carbon.
Higher atomic number (Z) = higher priority.
Order to remember: Br > Cl > S > P > O > N > C > H.
If there’s a tie, move one bond further out along each substituent.
List the set of atoms attached to that next atom, in descending atomic number, then compare the lists position by position.
The first point of difference decides priority.
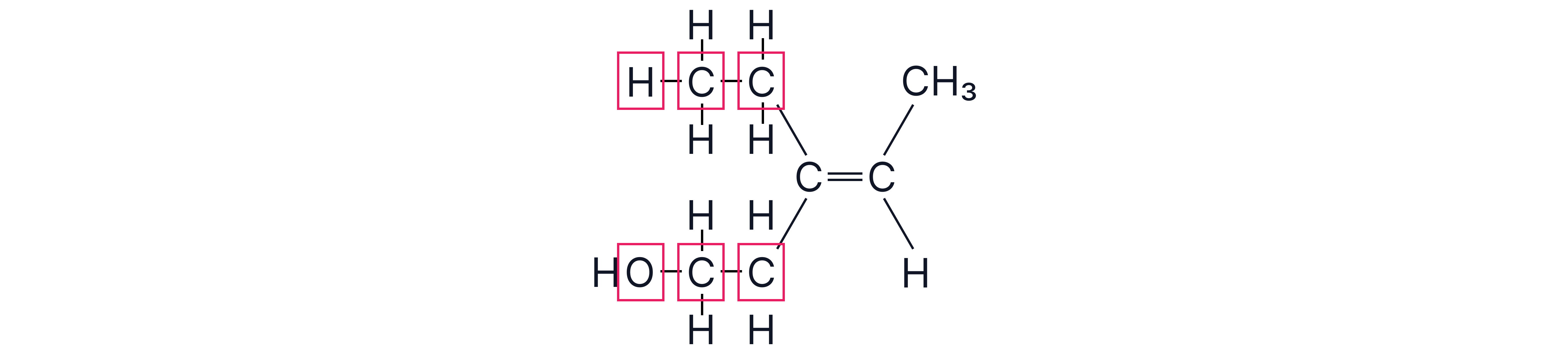
Compare –CH₂CH₃ vs –CH₂CH₂OH.
First atoms both C → tie because –CH₂CH₃ and –CH₂CH2OH:
Next-atom sets:
– For –CH₂CH₃: {C, H, H, H}
– For –CH₂CH₂OH: {C, H, H, O}
• Compare highest members: O (8) vs H (1) → –CH₂ CH₂OH higher priority.
Putting all of it together:
For the following molecule, state whether it is an E or Z-isomer.
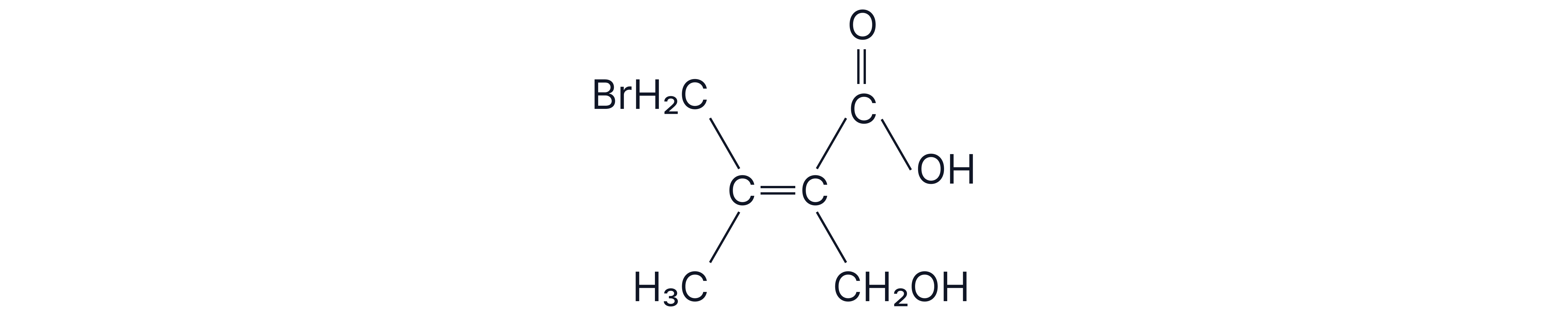
Worked Answer:
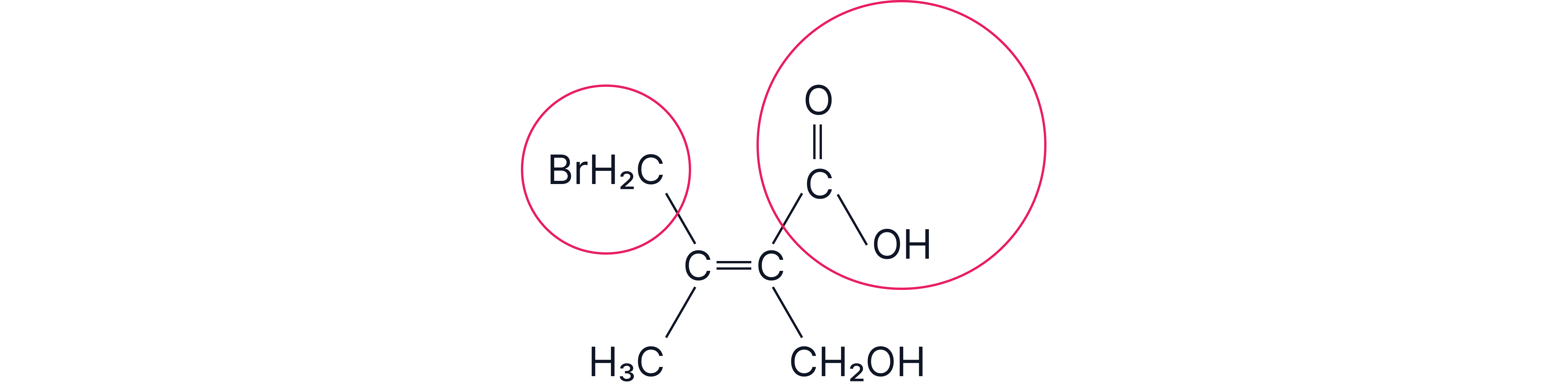
Step 1 – Priorities on the left carbon
Directly attached atoms: both substituents begin with C → tie.
Look one atom further out (exclude the back bond to the C=C):
–CH₂Br carbon is attached to {Br, H, H}
–CH₃ carbon is attached to {H, H, H}
Compare in descending atomic number: Br (35) vs H (1) → –CH₂Br has higher priority.
Step 2 – Priorities on the right carbon
Directly attached atoms: both are C → tie.
Next-atom sets (exclude back bond):
–COOH carbon counts the carbonyl double bond as duplicate → {O, O, O} [Note: For multiple bonds, use “duplicate-atom” counting (C=O counts as C–O and C–O*).]
–CH₂OH carbon → {O, H, H}.
Compare: O vs O (tie), then O vs H → –COOH has higher priority.
Step 3 – Assign E or Z
High-priority pairs: –CH₂Br (left, top) and –COOH (right, top).
High groups on the same side → Z
Answer
Configuration: Z.
Cis/Trans vs E/Z
When cis/trans can be used
Only when each carbon of the C=C bond has one identical group attached.
The “cis” form has the identical groups on the same side; the “trans” form has them on opposite sides.
Example: But-2-ene (CH₃–CH=CH–CH₃):
Each double-bond carbon has an H and a CH₃.
Cis = both CH₃ on same side.
Trans = CH₃ on opposite sides.
In this case, cis = Z, trans = E.
Cis/trans vs E/Z: when they don’t match
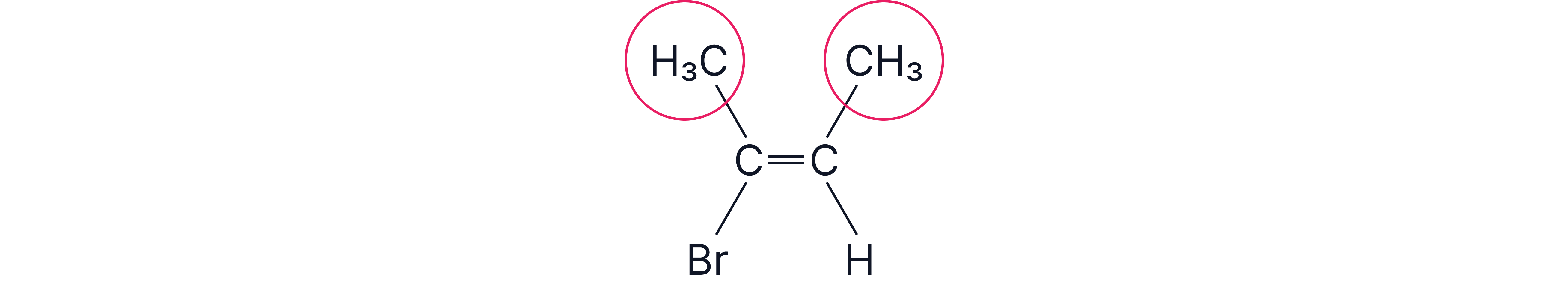
Consider 2-bromo-2-butene.
Step 1 – Cis/trans view
There is a methyl group on each carbon of the double bond.
Looking just at the two methyls, they are on the same side of the C=C.
So by cis/trans language, this molecule is cis.
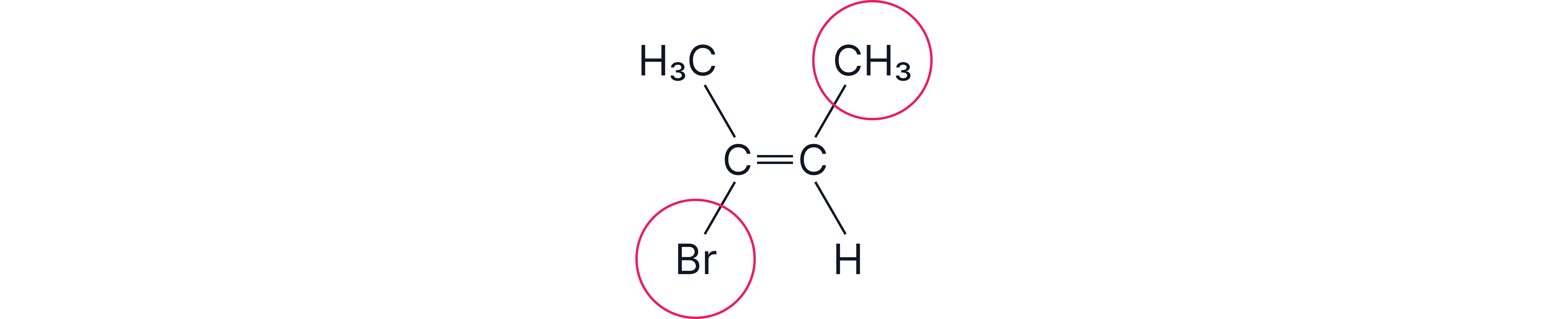
Step 2 – E/Z view
On the right carbon: methyl (–CH₃) vs hydrogen → CH₃ has higher priority.
On the left carbon: bromine (Br) vs methyl (–CH₃) → Br has higher priority.
The priority groups (CH₃ left, Br right) lie on opposite sides of the double bond.
Therefore, by E/Z rules, the molecule is E.
Conclusion
This molecule is cis-2-bromo-2-butene by the cis/trans system, but (E)-2-bromo-2-butene by E/Z.
Cis/trans and E/Z are based on different criteria:
Cis/trans = whether two identical groups are together or opposite.
E/Z = based on CIP priority rules.
Often cis = Z and trans = E (like in but-2-ene), but not always.
Reactivity of Alkenes
The pi bond is relatively weak, making alkenes more reactive than alkanes.
The C=C bond is a region of high electron density, so it attracts electrophiles.
Alkenes typically undergo electrophilic addition reactions.
Electrophilic Addition Mechanism
Key Features:
Electrophile is attracted to the electron-rich double bond.
The pi bond breaks and forms two new single bonds.
Often proceeds via a carbocation intermediate.
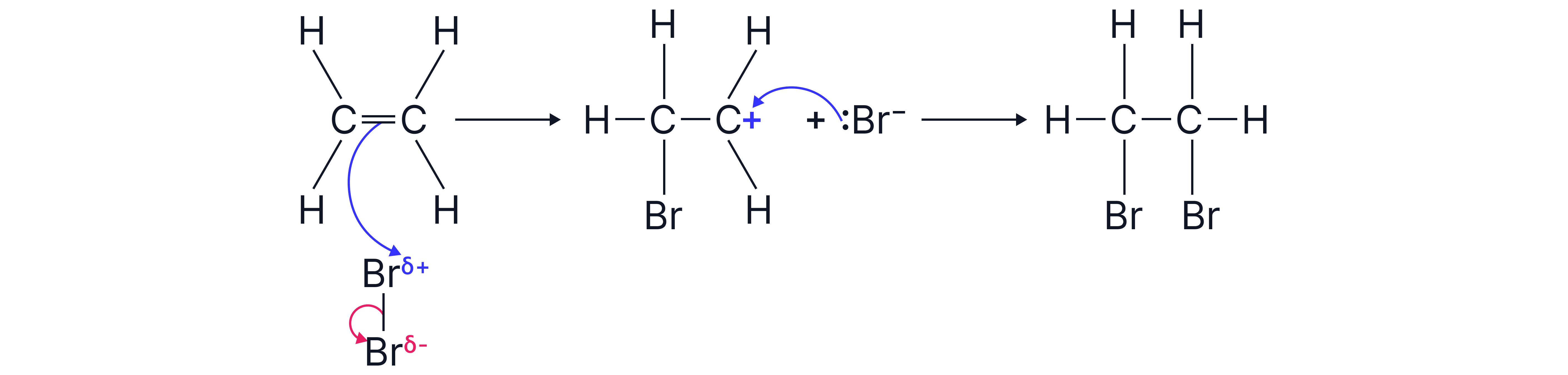
Example: Reaction with
Step 1: The double bond in ethene induces a dipole on the molecule. is the electrophile.
Step 2: The Br-Br bond breaks resulting in one bromine atom attaching to a C of the C=C double bond and one bromide ion () forming
Step 3: The bromide ion () then attacks the the carbocation intermediate resulting in 1,2-dibromoethane
Diagram of mechanism
Comparison with Alkanes
Feature | Alkanes | Alkenes |
Bonding | Only sigma bonds | Sigma + pi bonds |
Saturation | Saturated | Unsaturated |
Reactivity | Relatively unreactive | More reactive due to pi bond |
Type of Reactions | Free radical substitution | Electrophilic addition |
Worked Examples
Example 1
Describe the bonding in the carbon–carbon double bond of ethene.
Answer:
One sigma bond forms from head-on overlap of orbitals. A pi bond forms from sideways overlap of p-orbitals above and below the bonding axis. The pi bond restricts rotation and makes the bond more reactive.
Example 2
What is the shape and bond angle around each carbon atom in ethene?
Answer:
Trigonal planar shape with bond angles of approximately 120°, due to three regions of electron density around each carbon.
Example 3
Explain why alkenes are more reactive than alkanes.
Answer:
Alkenes have a pi bond, which is more exposed and weaker than sigma bonds, making it easier to break during reactions with electrophiles.
Practice Questions
Question 1
State the type of bond formed from the sideways overlap of p-orbitals in an alkene.
Answer:
Pi bond (π bond)
Question 2
Explain why rotation is restricted about the carbon–carbon double bond in ethene.
Answer:
The pi bond, formed by sideways overlap of p-orbitals, locks the bond in place above and below the plane of the molecule, preventing rotation.
Question 3
Describe and explain the bond angle around the carbon atoms in ethene.
Answer:
Bond angle is approximately 120° because the three bonding regions of electron density repel equally, resulting in a trigonal planar shape.