Amino Acids
Lajoy Tucker
Teacher

Contents
Introduction to Amino Acids
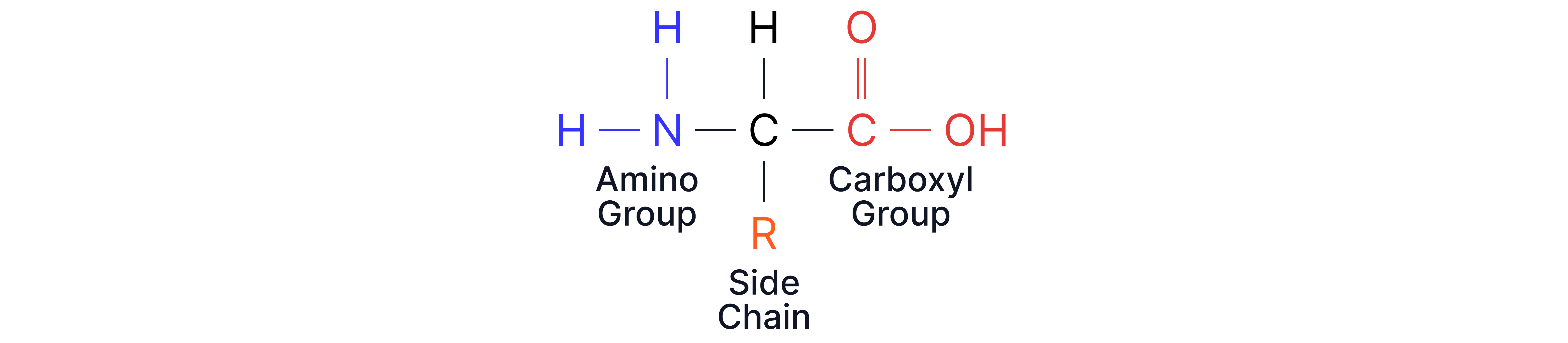
Amino acids are organic compounds containing:
An amine group
A carboxylic acid group
A variable R group (side chain)
General structure:
Amino Acids Explainer Video
Zwitterions
Due to the presence of both acidic and basic groups, amino acids exist as zwitterions – molecules with both a positive and negative charge
These charges cancel out to give an overall neutral molecule.
Zwitterion structure:
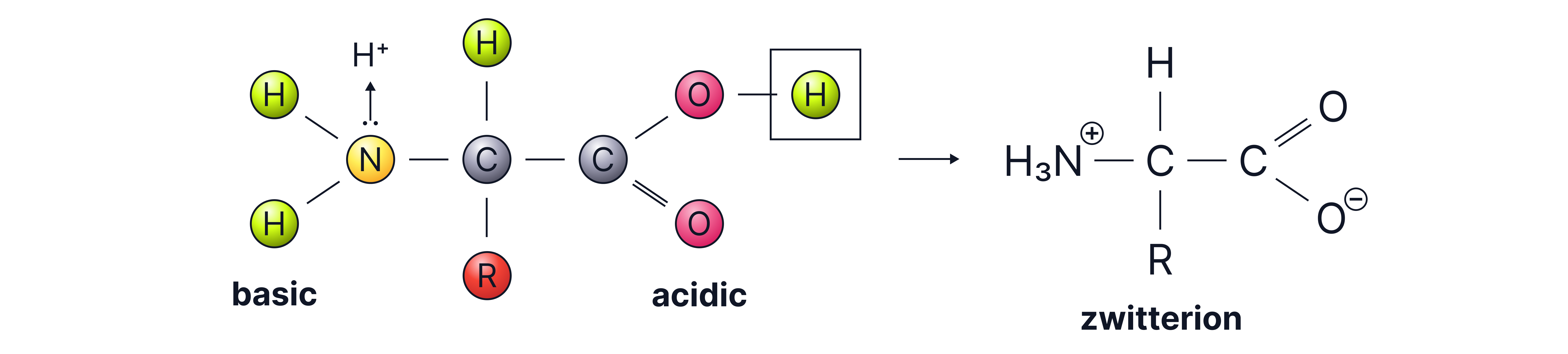
accepts a proton →
donates a proton →
Note – zwitterions must have no overall charge.
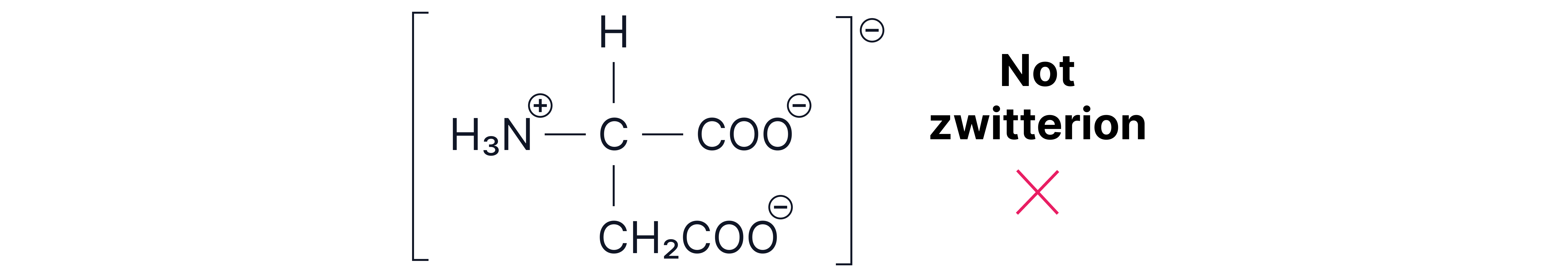
Behaviour in Acidic and Basic Conditions
The pH will determine the structure of an amino acid.
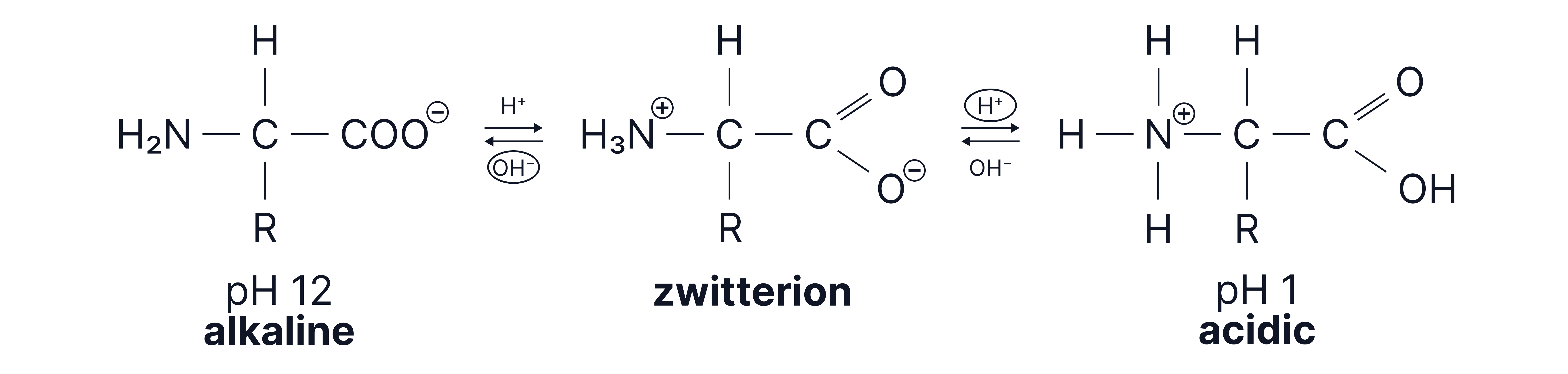
Condition | Behaviour | Ion Form |
Acidic (pH ) | Gains | |
Basic (pH ) | Loses |
Optical Isomerism in Amino Acids
Many amino acids are chiral, meaning the carbon bonded to , , , and has four different groups.
Results in non-superimposable mirror images (enantiomers)

Most amino acids exist naturally as the L (left-handed) enantiomer.
Peptide Formation
A peptide bond forms via a condensation reaction between:
The group of one amino acid
The group of another
Forms a dipeptide + water:
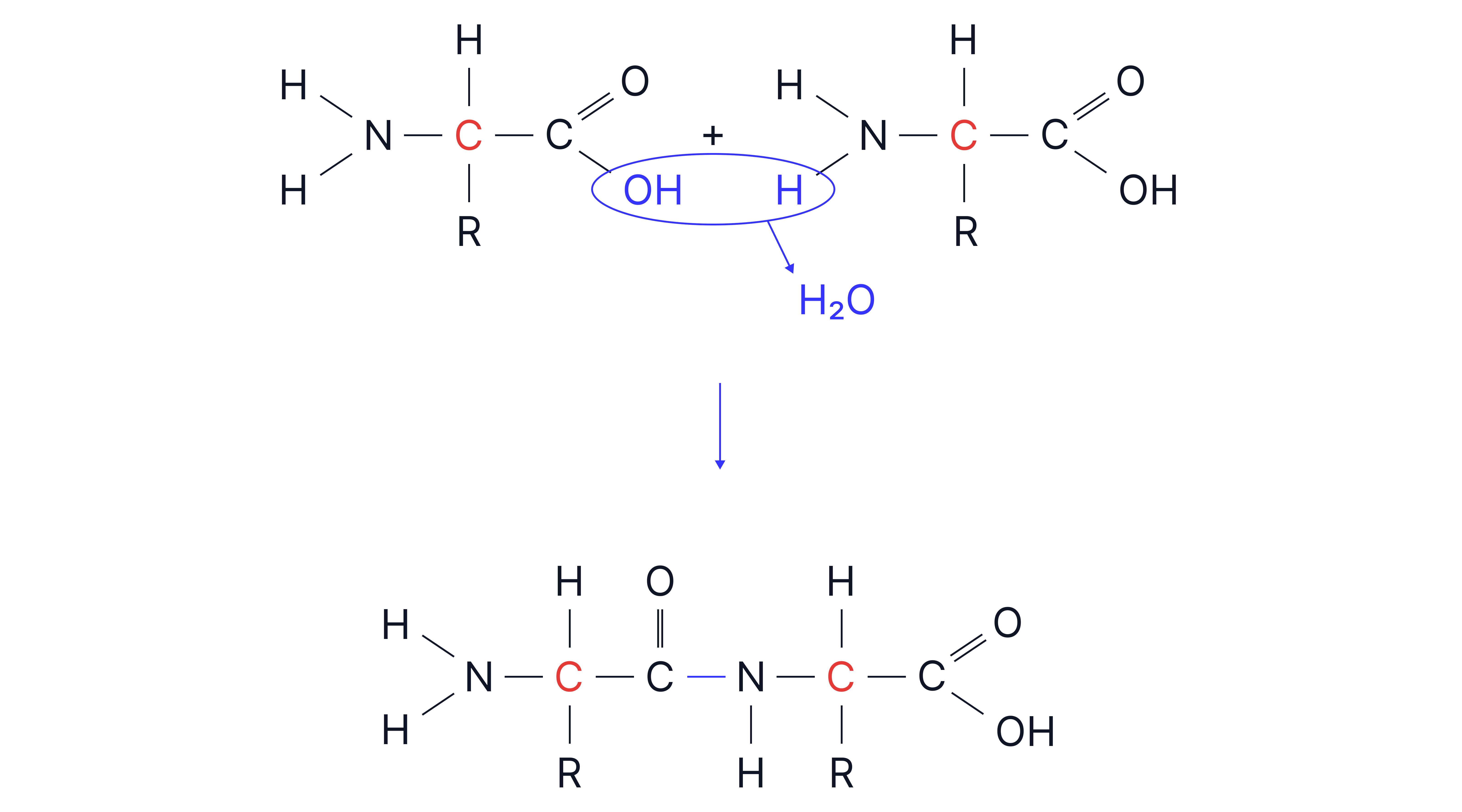
Peptides are covered in the Proteins and Enzymes note
Practice Questions
Question 1
Give the structural formula of the zwitterion formed by alanine.
Answer
Question 2
Explain why glycine does not show optical isomerism, but most other amino acids do.
Answer
Glycine has two hydrogen atoms attached to the central carbon (1 mark)
So it is not chiral (1 mark)
Question 3
Give the structure of the main species present in an aqueous solution of lysine at pH 13?
Answer
Alkaline solution therefore -COOH group deprotonated
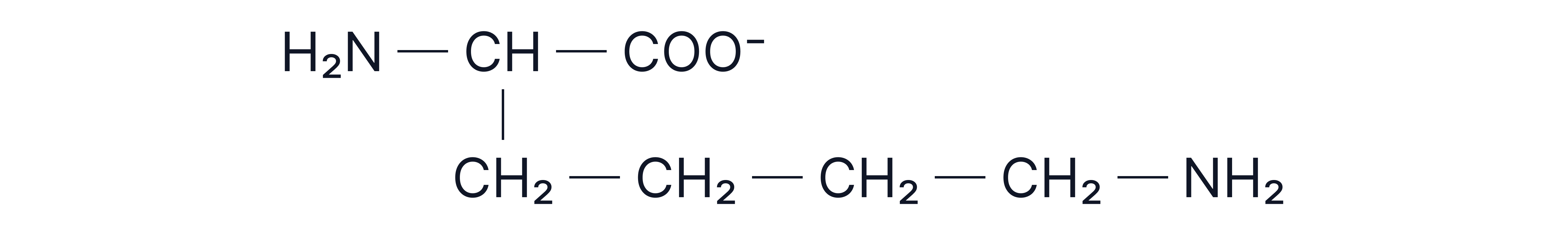
Question 4
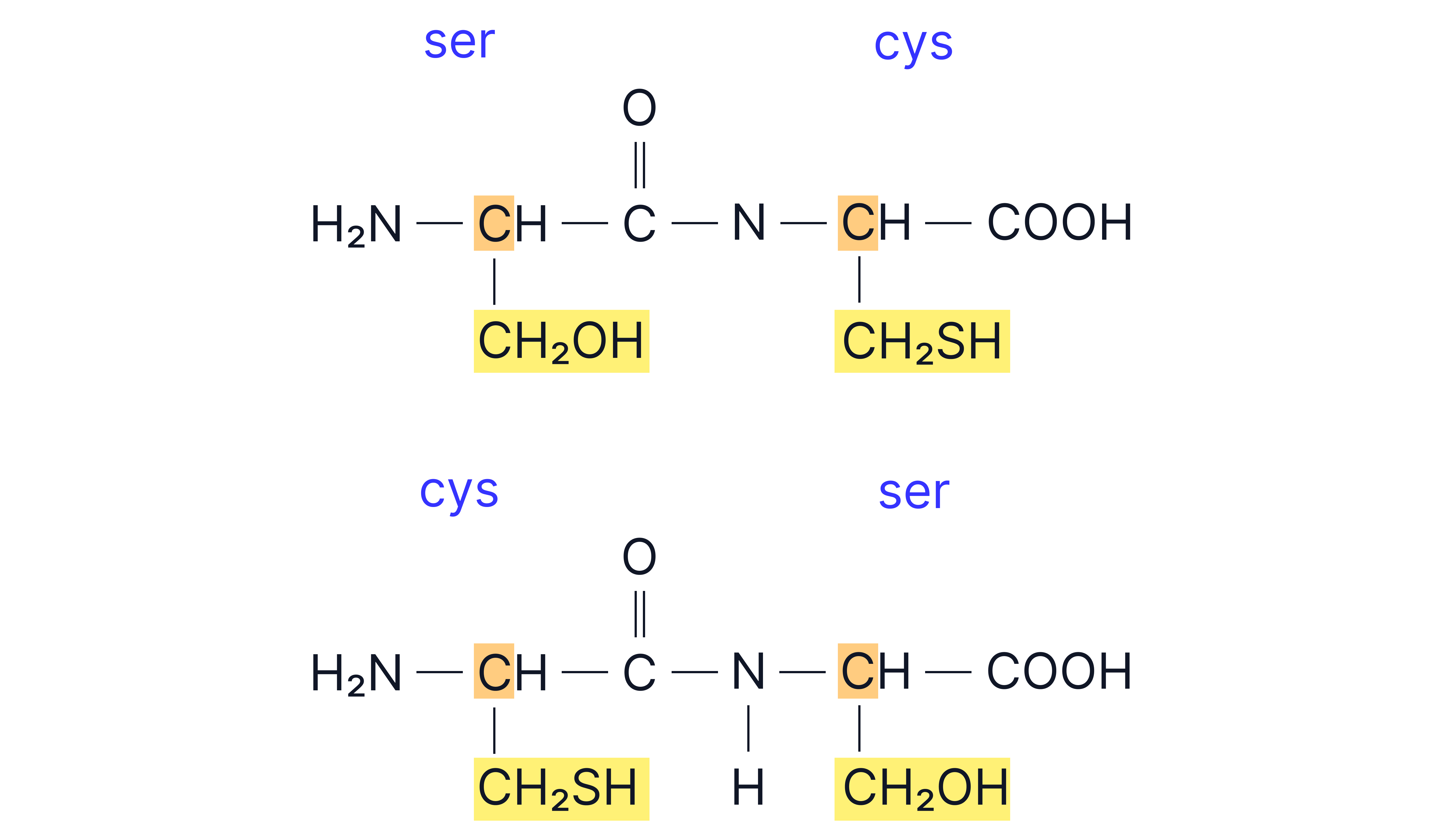
Draw the two possible dipeptides formed from serine and cysteine
Answer