Proteins and Enzymes
Lajoy Tucker
Teacher

Contents
What Are Proteins?
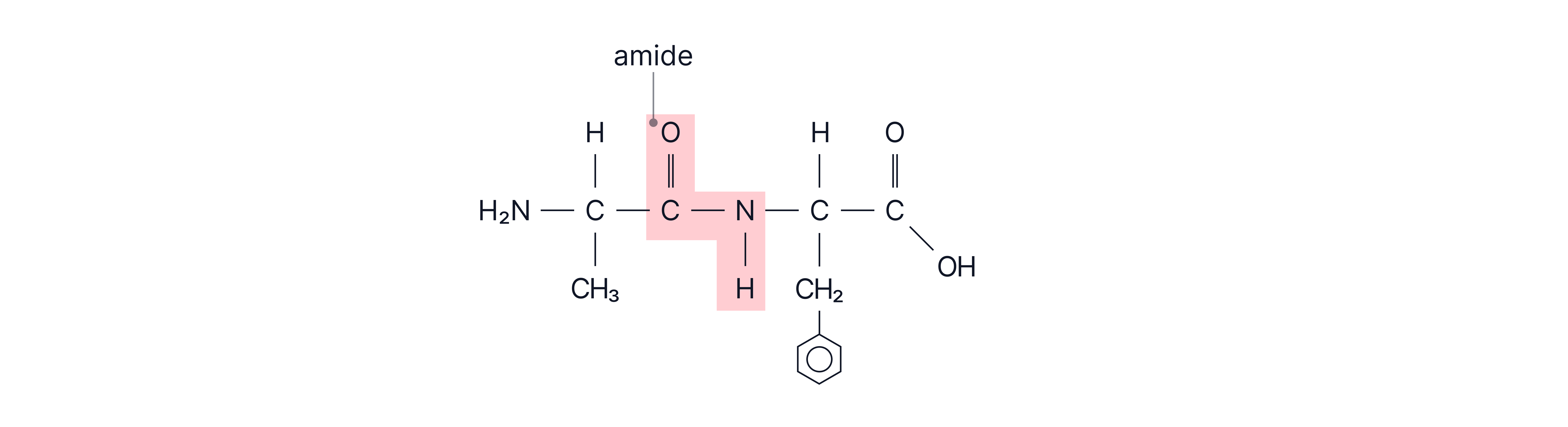
Proteins are large biological molecules made from amino acids joined together via peptide (amide) bonds in a condensation reaction.
Dipeptide formation:
Protein Structure Explainer Video
Protein Structure
There are four levels of protein structure.
Level | Description | Held together by… |
Primary | Sequence of amino acids in the chain | Peptide bonds (covalent) |
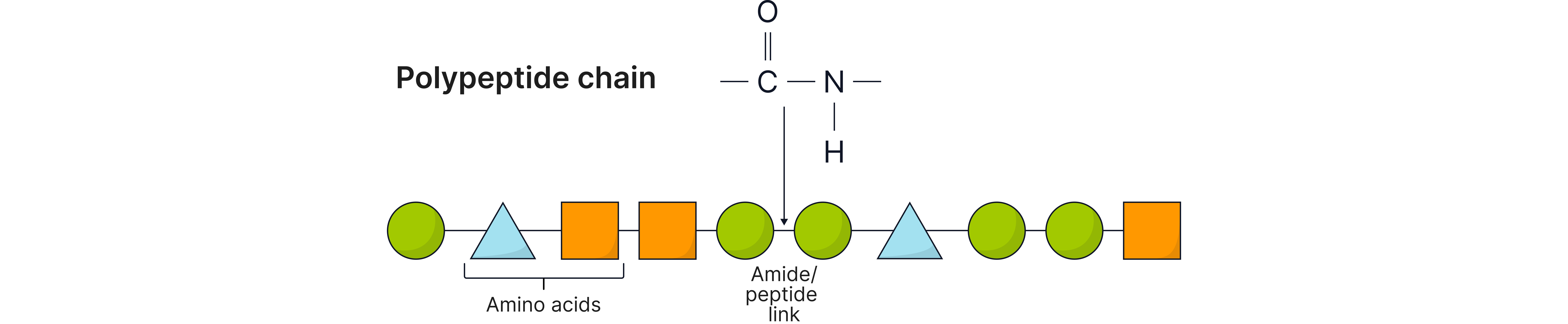
Level | Description | Held together by… |
Secondary | Folding of primary structure into α-helix or β-pleated sheet | Hydrogen bonding |
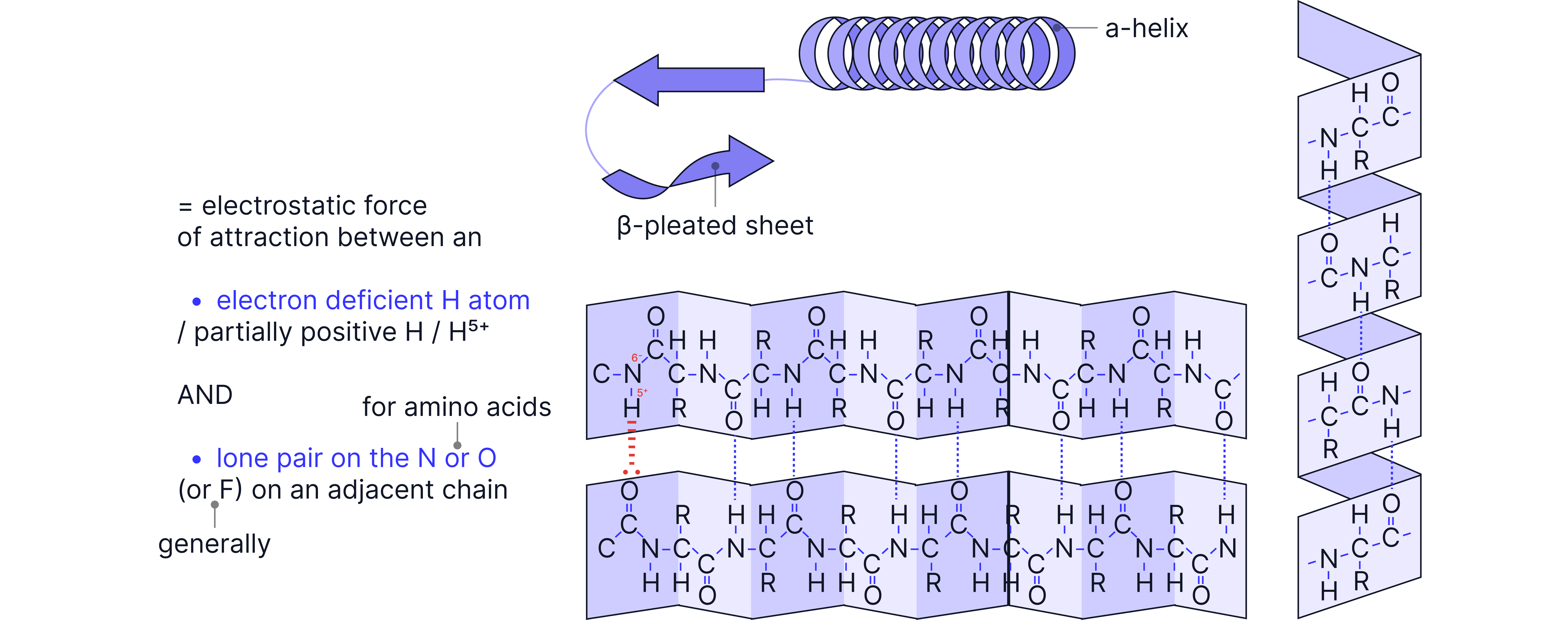
Level | Description | Held together by… |
Tertiary | 3D shape due to R group interactions | Hydrogen bonds, ionic bonds, disulfide bonds |
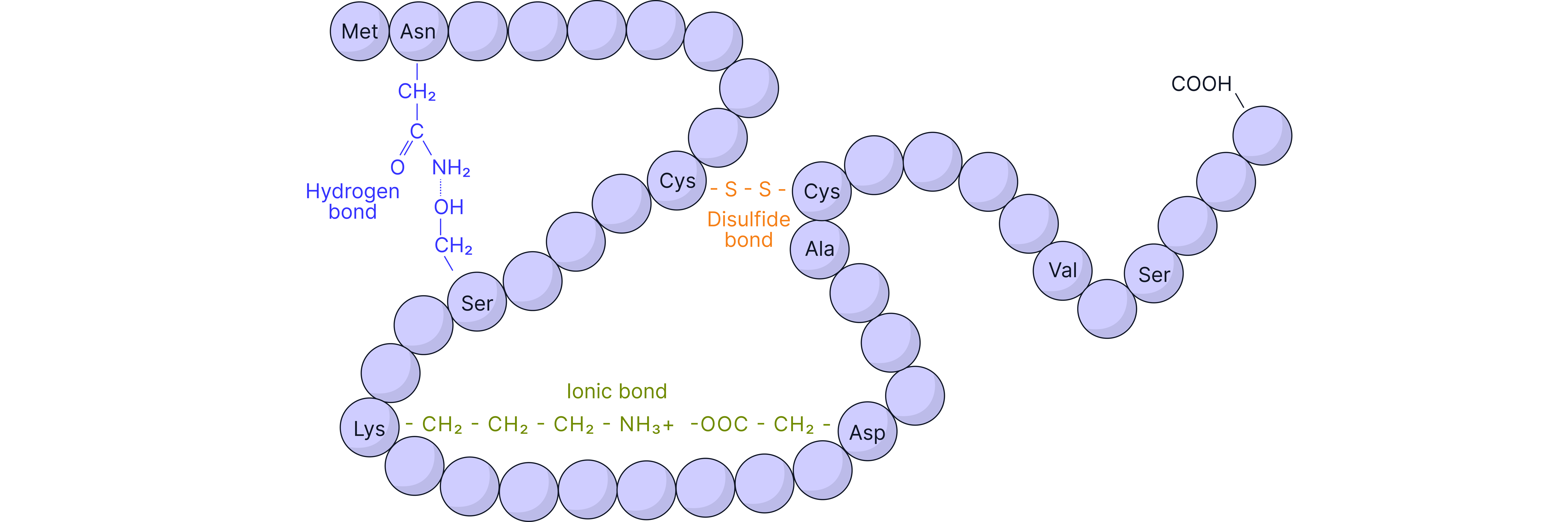
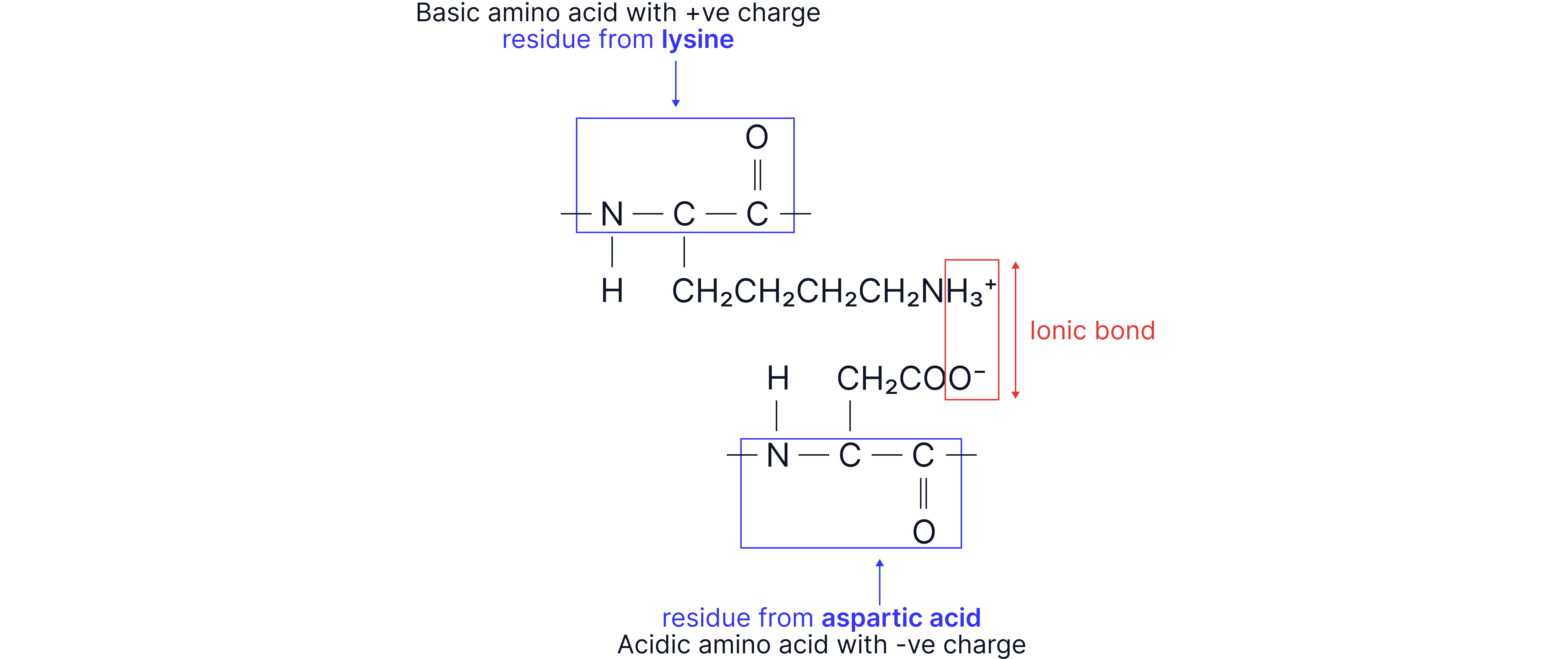
Ionic bonds: electrostatic forces of attraction between oppositely charged ions. Formed between Lysine and Aspartic Acid R-groups due to presence of additional and groups respectively.
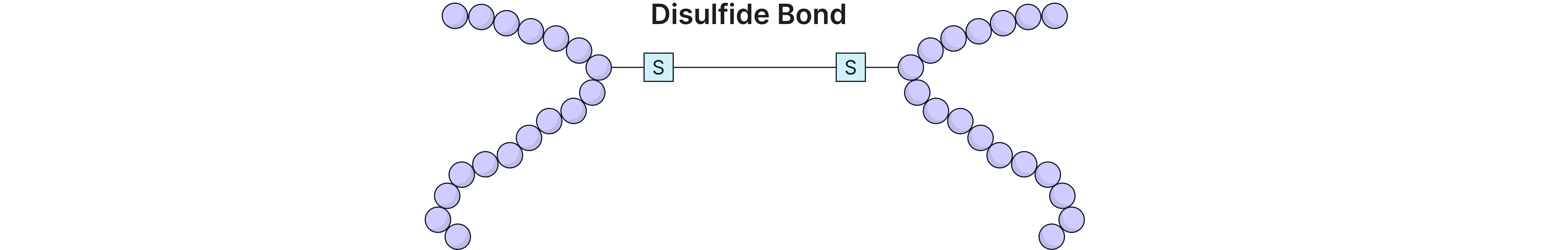
Disulfide bonds: covalent bonds between sulfur atoms on cysteine R-groups
Relative strength of forces: Ionic bonds and disulfide (covalent) bonds are stronger than hydrogen bonds which are intermolecular forces.
Level | Description | Held together by… |
Quaternary (not assessed) | Multiple polypeptides joined | Same as tertiary |
Summary
Hydrogen bonds: Between and )
Disulfide bridges: Covalent S–S bonds between cysteine residues
Ionic bonds: Between oppositely charged R groups on lysine and on aspartic acid
Van der Waals: Weak forces between non-polar side chains
Hydrolysis of Proteins
Proteins can be hydrolysed to amino acids using:
Acid hydrolysis (e.g. HCl under reflux)
Enzymatic hydrolysis (biological systems)
Acid hydrolysis
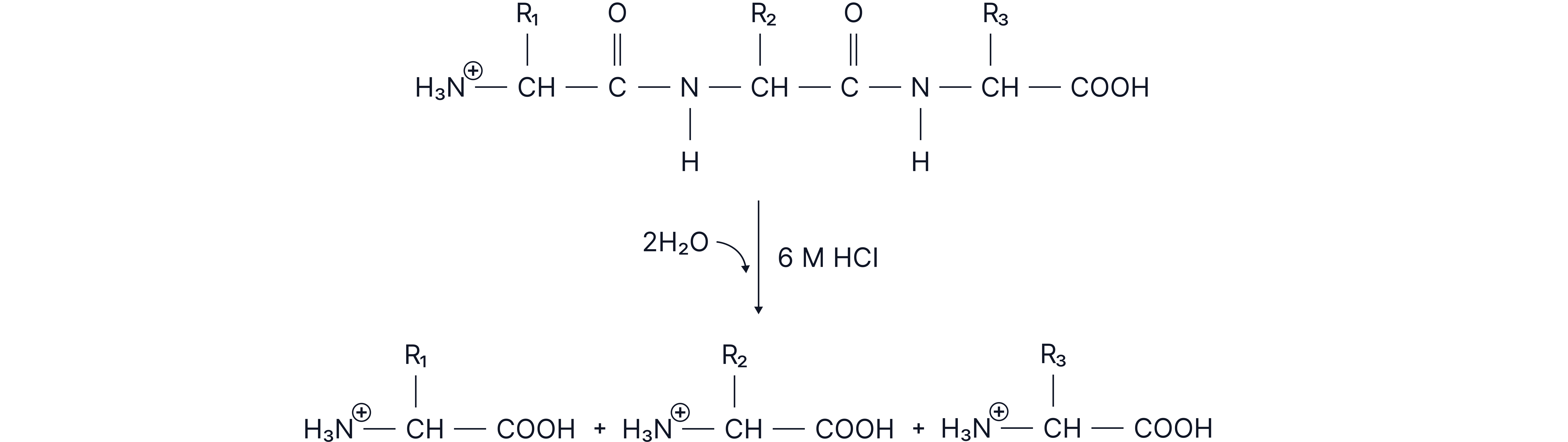
Note: The amine groups have been protonated to as they are basic and therefore accept ions.
Enzymes as Biological Catalysts
Structure
Enzymes are globular proteins with a specific 3D shape.
The active site has a precise shape that matches the substrate.
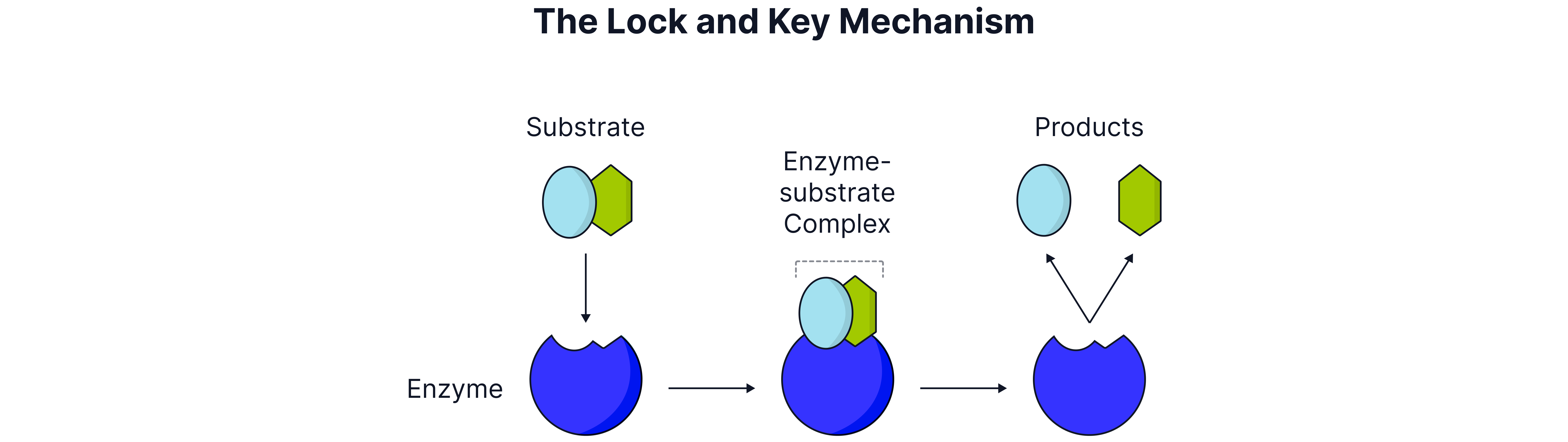
Mechanism
Lock and Key: Substrate fits exactly into active site
Stereochemistry
Enzyme active sites are chiral 3D pockets,
Active site only binds to one enantiomer of a chiral molecule and so are stereospecific
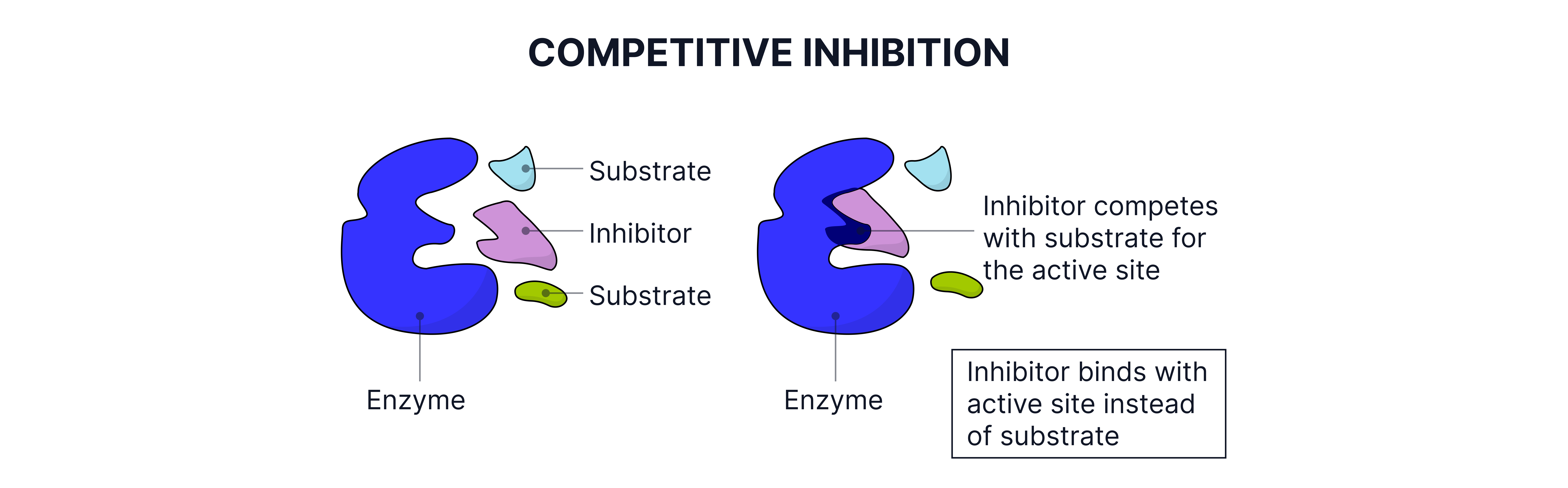
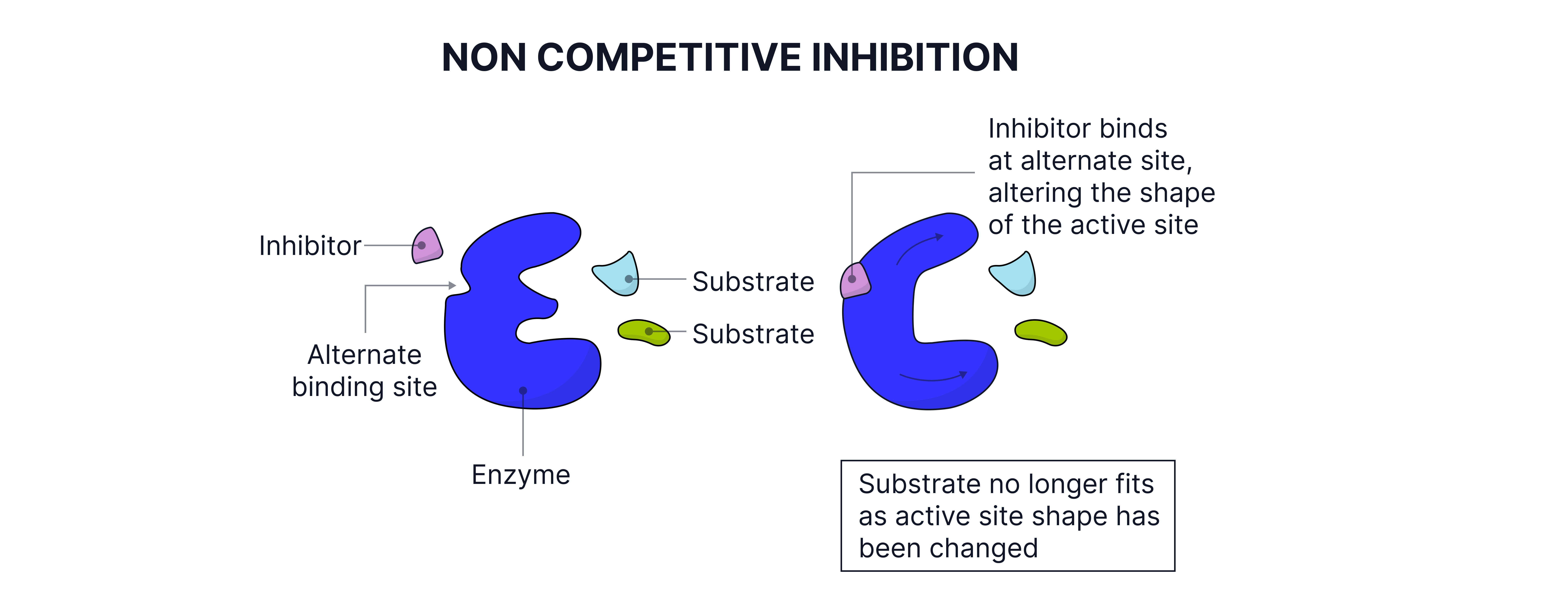
Enzyme Inhibition
Types:
Competitive inhibitors: Similar shape to substrate, bind to active site preventing the original substrate from binding
Non-competitive inhibitors: Bind elsewhere, change active site shape therefore the original substrate can no longer bind.
Drug Design
Many drugs act as enzyme inhibitors
They are designed using computational modelling to fit into the enzyme’s active site
Question 1:
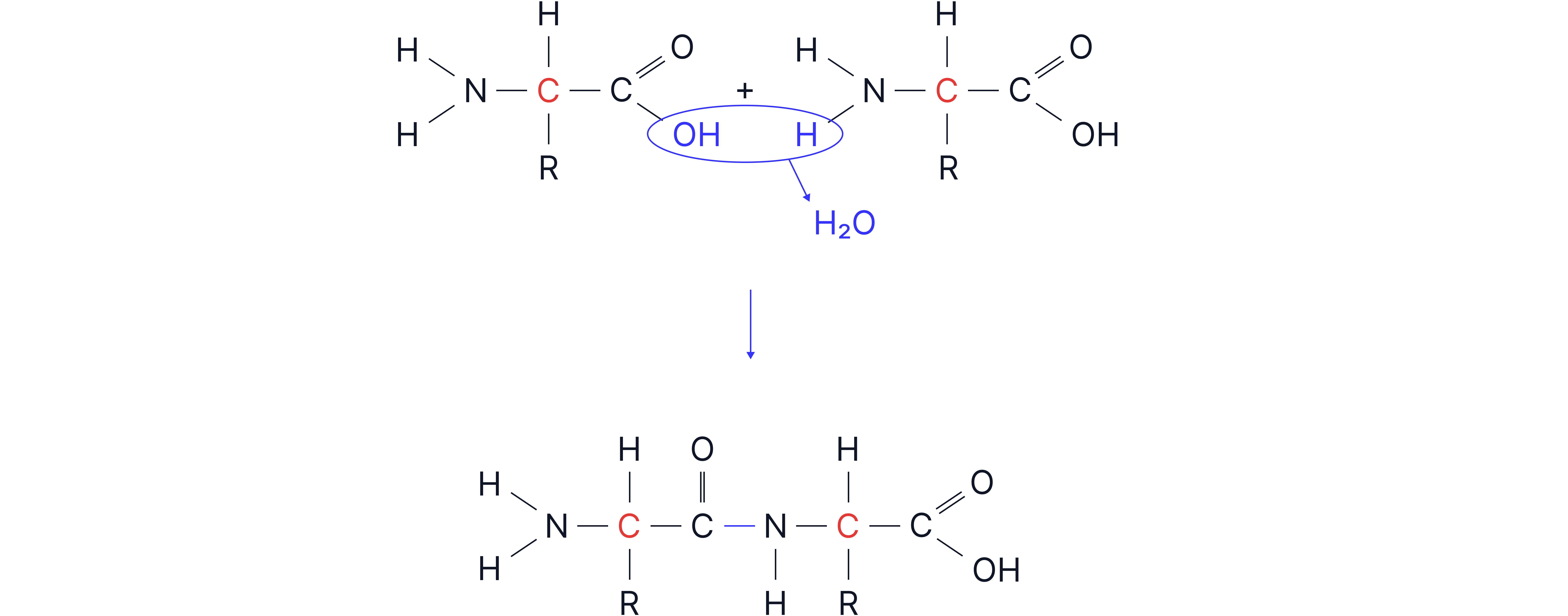
Explain how a dipeptide forms from two amino acids.
Answer:
1. Condensation reaction occurs (1)
2. Between the of one and the of the other (1)
3. Forms a peptide bond and releases water (1)
Question 2:
Draw the alanine-phenylalanine dipeptide
Answer: