Balanced Equations
Lajoy Tucker
Teacher

Introduction & Definitions
Balanced Chemical Equation: A symbolic representation of a chemical reaction in which the number of atoms of each element is the same on both sides of the equation.
Ionic Equation: Shows only the reacting ions and omits spectator ions.
State Symbols:
= solid
= liquid
= gas
= aqueous solution
Basic Principles

Law of Conservation of Mass: Matter cannot be created or destroyed in a chemical reaction → number of atoms of each element must be the same on both sides.
Balancing Equations: ensures that the number of each type of atom is equal on both sides.
Ionic Equations: show species that actually change during the reaction. Spectator ions are not included.
Balancing charges: The overall charge on both sides of the equation must be the same.
Balancing Equations
Worked Example 1
Answer
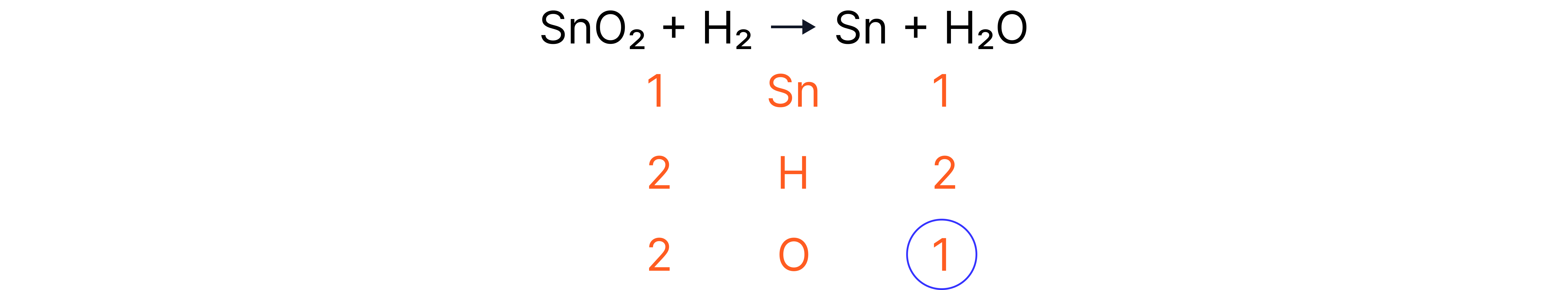
1. Count the number of atoms of each element on each side.
2. Change coefficients to multiply the number of a particular species.
____ + ____ → ____ +
3. Check to see if changing the coefficient affects any of the other elements.
____ + → ____ +
4. Where no balancing numbers are needed, do not include a coefficient.
Worked Example 2
Where there are polyatomic ions that do not change throughout the reaction, it is often easier to treat them as one unit rather than as individual atoms.
Answer
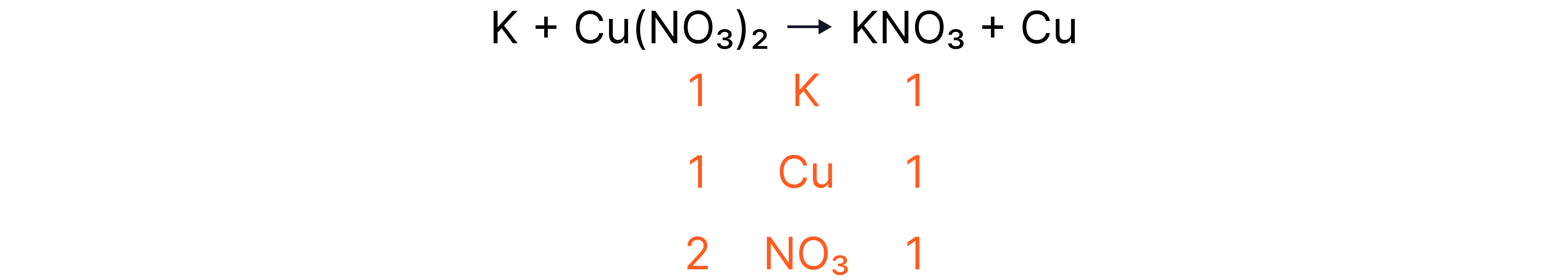
+ ____ + ____
Ionic Equations
When reactions take place in solution, water soluble ionic compounds will dissociate (split up into their constituent ions).
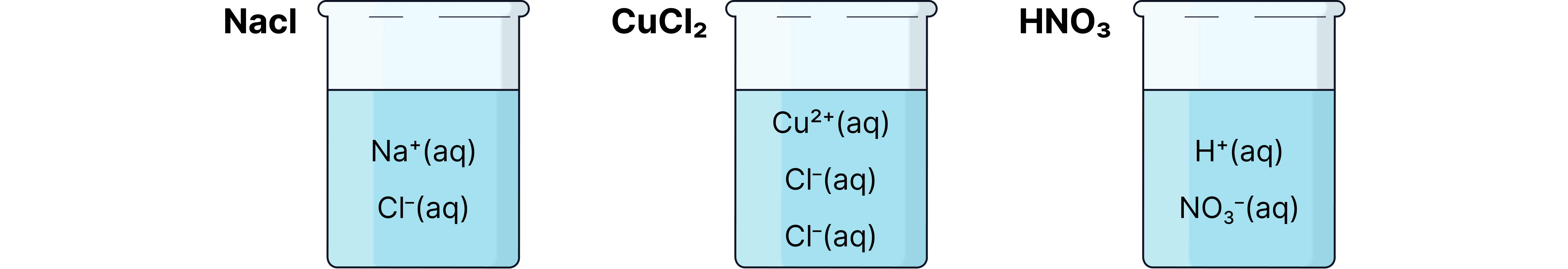
Ionic equations include only the species that are changed in the reaction.
Spectator ions (ions that do not take part in the reaction) are excluded
Example
Full:
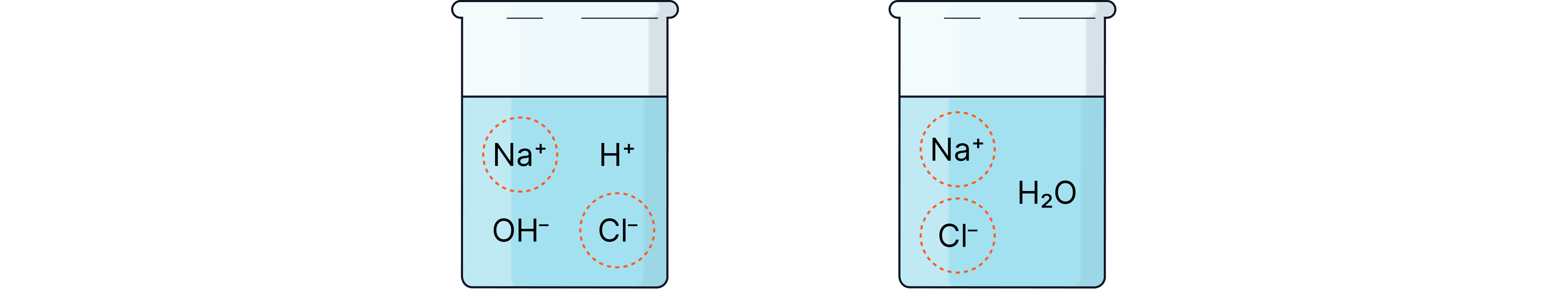
Ionic:
Tip!
State symbols are often required for these and will help to identify which species will dissociate and which will not.
Worked Example
Write the ionic equation for the reaction below
Answer
1. Identify which species will dissociate (aqueous ionic compounds including acids)
2. Remove any spectator ions
Worked Example
Write the ionic equation for the reaction between barium chloride and sulfuric acid to produce the barium sulfate precipitate.
Answer
In precipitation reactions, the insoluble solid (precipitate is formed from its constituent aqueous ions.
This must have been formed from and ions.
Practice Questions
Question 1
Balance the following equations
a) ___ + ___→ ___ + ___
b) ___ + ___ → ___
c) ___ → ___+ ___+ ___
Answer
a)
b)
c) or
Question 2
Write the following as ionic equations. Include state symbols.
a)
b) Production of insoluble silver iodide from reaction between silver (I) nitrate (aq) and potassium iodide (aq)
Answer:
a)
b)