The Mole & the Avogadro Constant
Lajoy Tucker
Teacher

Introduction & Definitions
Mole: A unit of measurement that represents a specific quantity of a substance. That quantity is particles
Avogadro Constant : The number of particles (atoms, molecules, ions, etc.) in one mole of a substance, approximately .
Molar Mass (Mr): The mass of one mole of a substance , numerically equal to its relative molecular/formula mass.
Basic Principles
One mole of any substance contains particles.
Amount in moles links mass, volume, and number of particles.
The ideal gas equation and concentration equations allow chemists to calculate amounts of substance under various conditions.
Worked Examples
Mole calculations using mass:
Moles = Mass
Example 1:
Calculate the number of moles of in ?
Moles
Example 2:
Calculate the number of moles of oxygen atoms in of
Moles
Two atoms per
Moles of atoms
Using Avogadro's Constant
Number of Particles = Moles Avogadro’s constant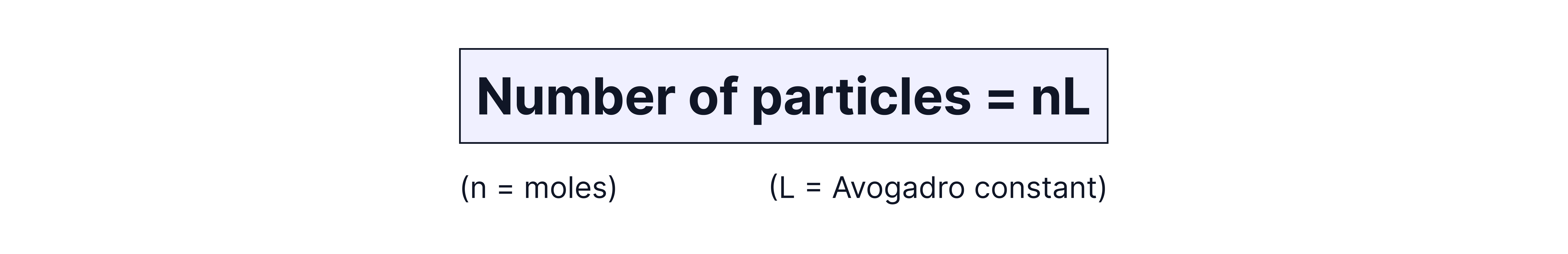
Example 3:
Calculate the number of ions in of
ions
Practice Question
Calculate the number of water molecules in of
Answer
Moles
Molecules
Note: Number of moles can also be calculated for gases using the ideal gas equation and using volume and concentration for solutions.
These are covered in separate revision notes
Mole calculations for solutions:
Moles = Concentration Volume
Mole calculations for gases:
Where:
pressure ,
volume ,
moles,
gas constant ,
temperature
Key Tips & Reminders
Avogadro’s constant will always be given in exams when needed.
Look out for if a question is asking for number of moles or number of particles (atoms, molecules, ions).
If calculating number of particles, multiply number of moles by Avogadro’s constant.
Look out for where questions ask for number of atoms/ions within a larger species (e.g. atoms in , ions on ) and scale accordingly using molar ratios.