Basic Properties of Amines
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Amines are organic derivatives of ammonia , where one or more hydrogen atoms are replaced with alkyl or aryl groups.
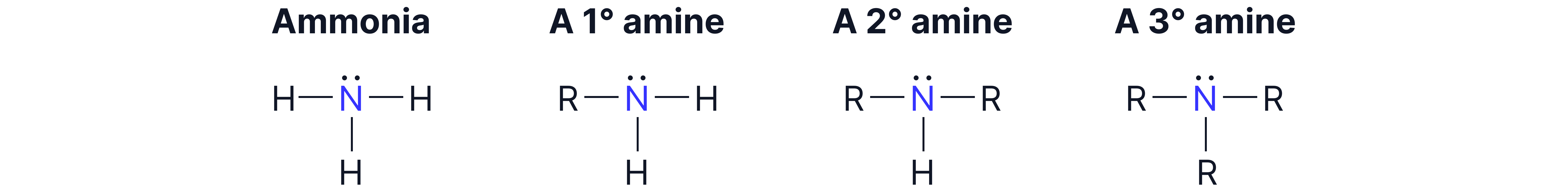
Types of Amines:
Primary amine:
Secondary amine:
Tertiary amine:
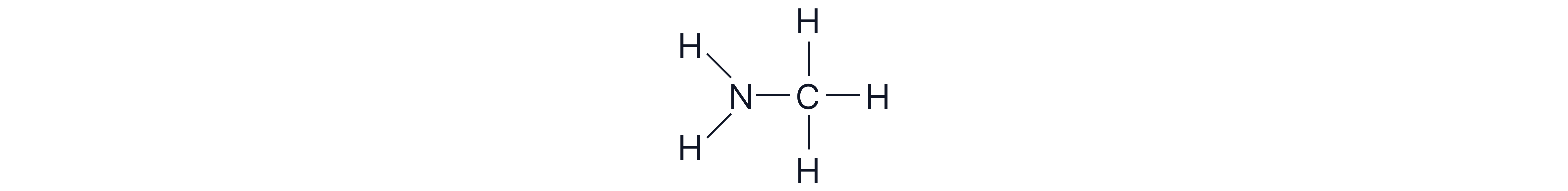
Aliphatic amine: the amino groups are directly attached to non-aromatic groups.
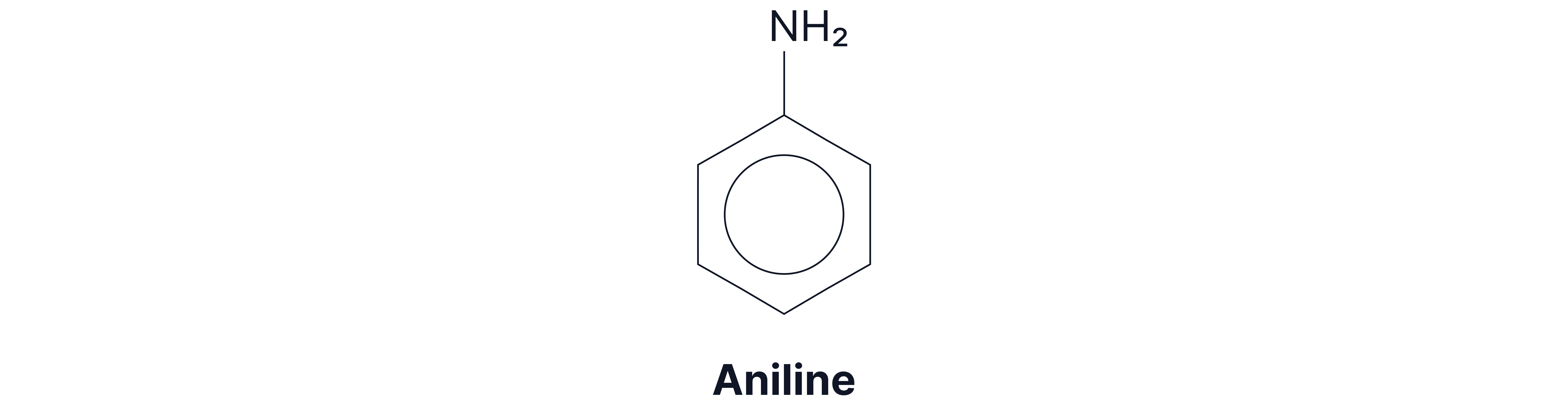
Aromatic amine: the amino group is directly attached to an aromatic ring.
Base: A substance that accepts protons
Basicity: The ability of a molecule to accept a proton (act as a base).
Basicity of Amines Explained
Principles
Amines behave as Brønsted–Lowry bases due to the lone pair on the nitrogen atom, which can accept a proton.
They partially dissociate in water to release ions
Example:
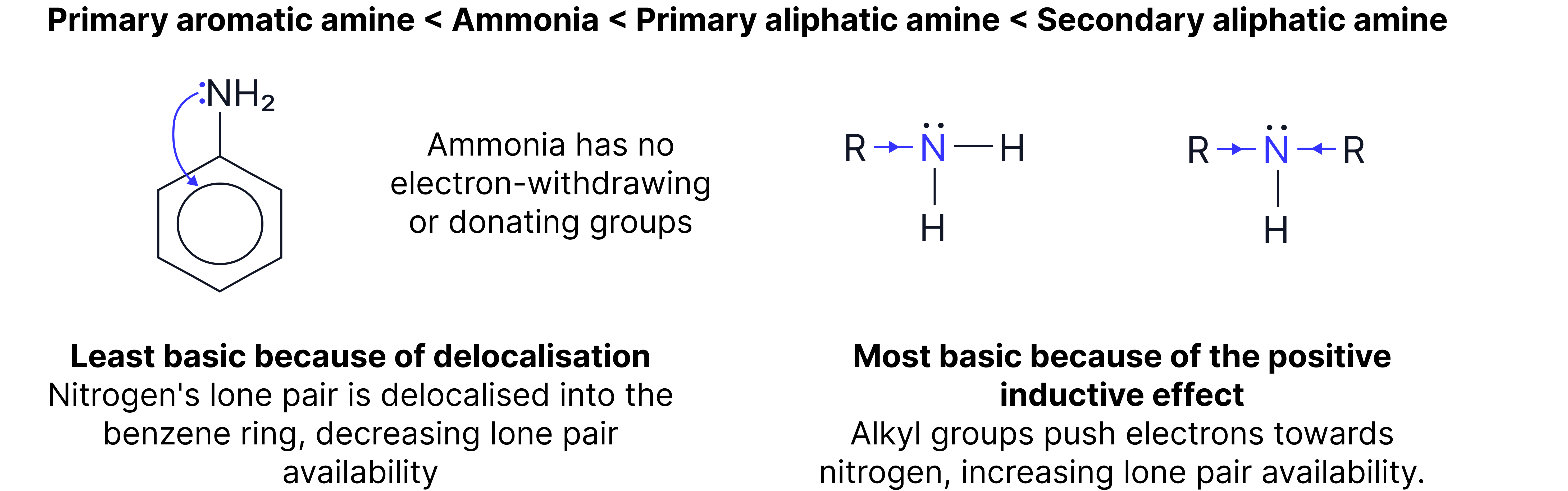
Comparing Basicity of Amines
The strength of a base depends on the availability of the lone pair on nitrogen. Basicity of amines is often compared to ammonia. The order of basicity generally is:
Practice Questions
Question 1
Rank the following in order of basicity and explain and phenylamine.
Answer:
1. - most basic more alkyl groups = greater positive inductive effect lone pair on most available.
2. - Primary alkyl group increases electron density.
3. - No alkyl groups, lone pair less available.
4. Phenylamine - Lone pair delocalised into aromatic ring - lone pair on least available.
Explanation:
Alkyl groups donate electron density, enhancing the lone pair availability for protonation. Aromatic rings withdraw electron density, reducing basicity.
Question 2
Write an equation to show methylamine acting as a Brønsted–Lowry base in aqueous solution.
Answer:
Key Tips and Reminders
Always consider the inductive effect of alkyl groups when comparing aliphatic amines.
Phenylamine is significantly weaker due to delocalisation of nitrogen’s lone pair.
Examiner Insight:
Be specific in comparing basicity: mention the availability of the lone pair.
Use correct terminology: Brønsted–Lowry base, inductive effect, delocalisation, etc.