Preparing Amines
Lajoy Tucker
Teacher

Contents
What are Amines?
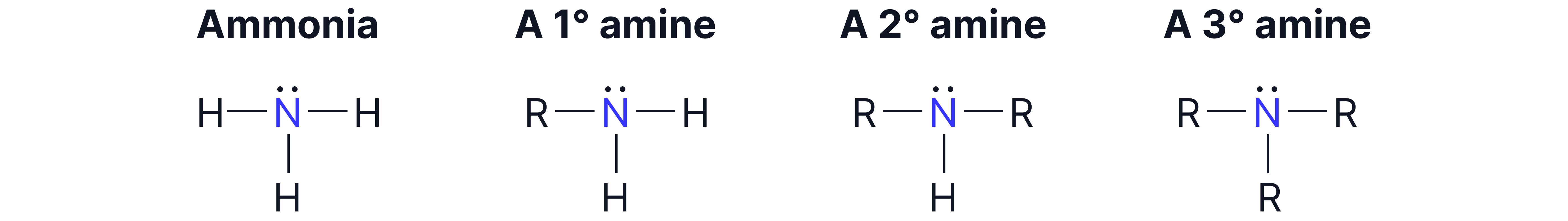
Amines are organic compounds derived from ammonia , where one or more hydrogen atoms are replaced with alkyl or aryl groups.
Types of Amines:
Primary:
Secondary:
Tertiary:
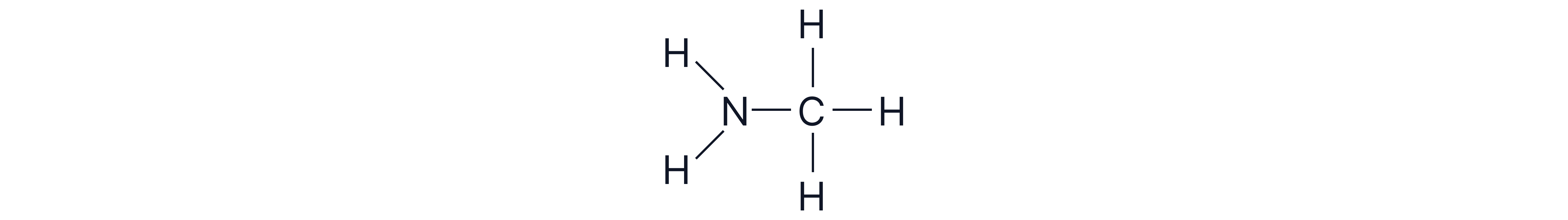
Aliphatic amine: the amino group is directly attached to non-aromatic groups e.g. an alkyl group.
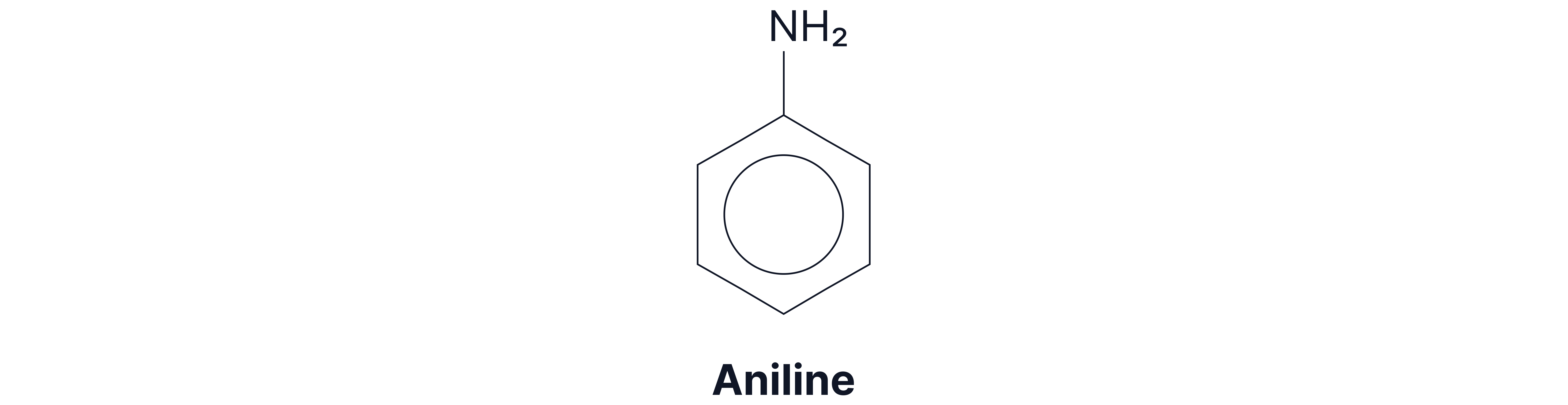
Aromatic amine: the amino group is directly attached to an aromatic ring.
Preparing Amines Explained
Methods of Preparing Amines
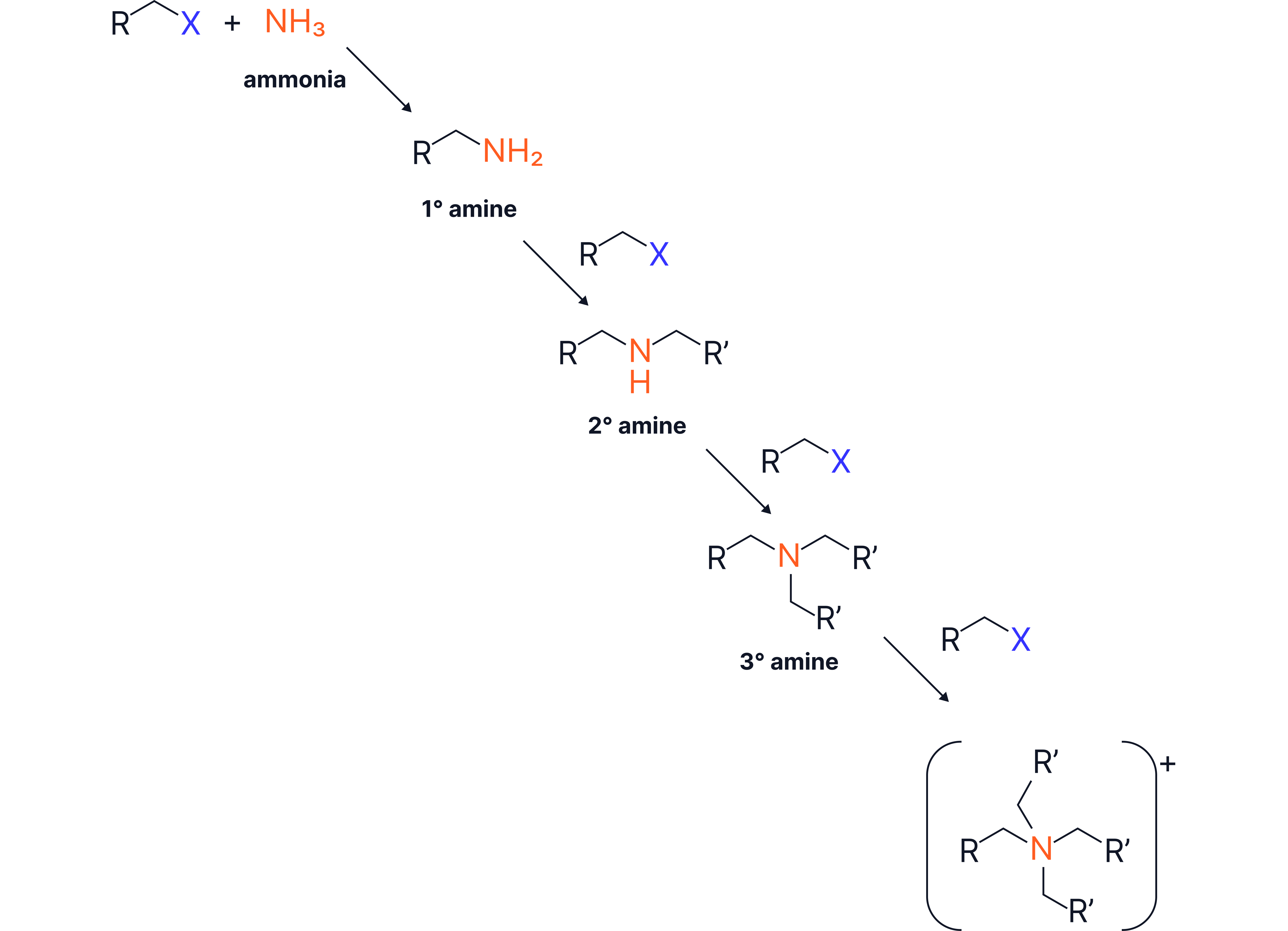
1. From Halogenoalkanes (via Nucleophilic Substitution).
Equation:
Reagents: Excess ammonia
Conditions: Ethanolic solution, sealed tube, heat
Forms primary amines via nucleophilic substitution
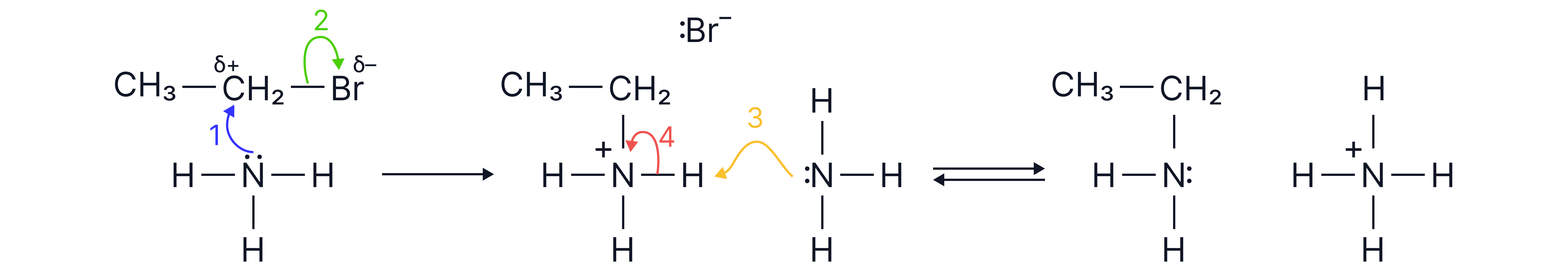
The ammonia must be in excess to reduce the chance of further substitution which leads to secondary and tertiary amines, and quaternary ammonium salts.
If excess halogenoalkane used:
2. From Nitriles (Reduction Method).
Step 1: Convert halogenoalkane nitrile
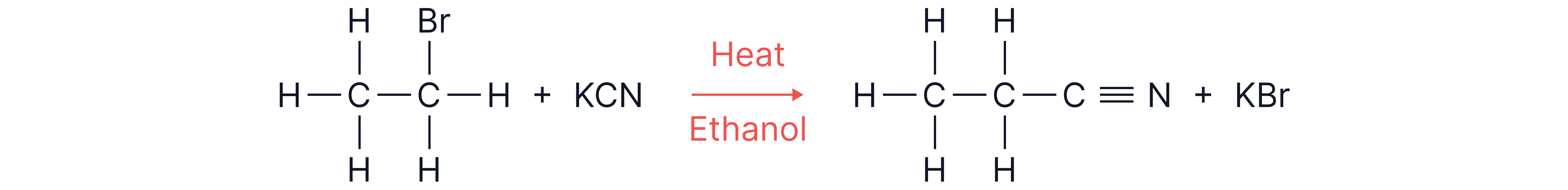
(via nucleophilic substitution)
Step 2: Reduce nitrile primary amine

Reducing agent : Hydrogen () with a catalyst or in dry ether .
This method prevents further substitution, making it better for producing primary amines with greater purity.
This is also a useful method for extending the length of the carbon chain.
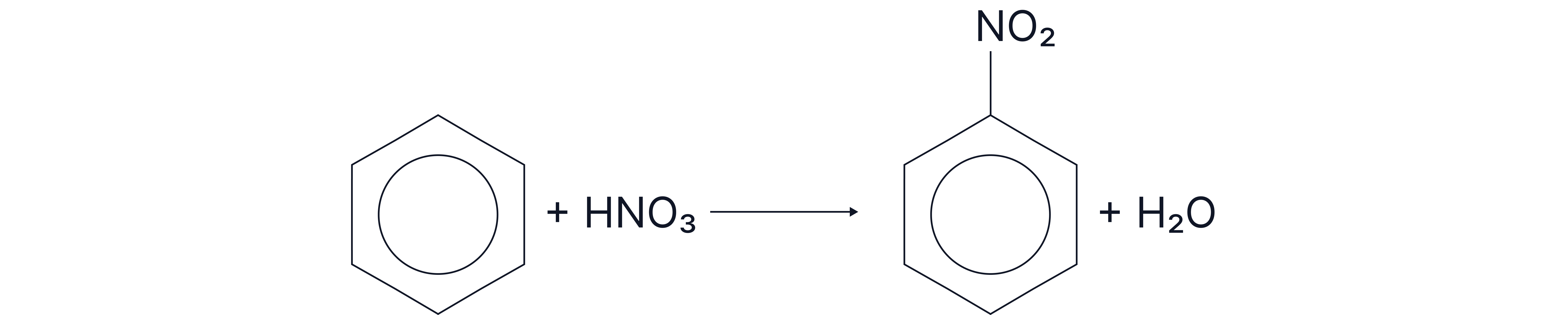
3. Aromatic Amines From Nitrobenzene (Reduction).
Step 1: Nitration of benzene nitrobenzene
Step 2: Reduce nitrobenzene phenylamine
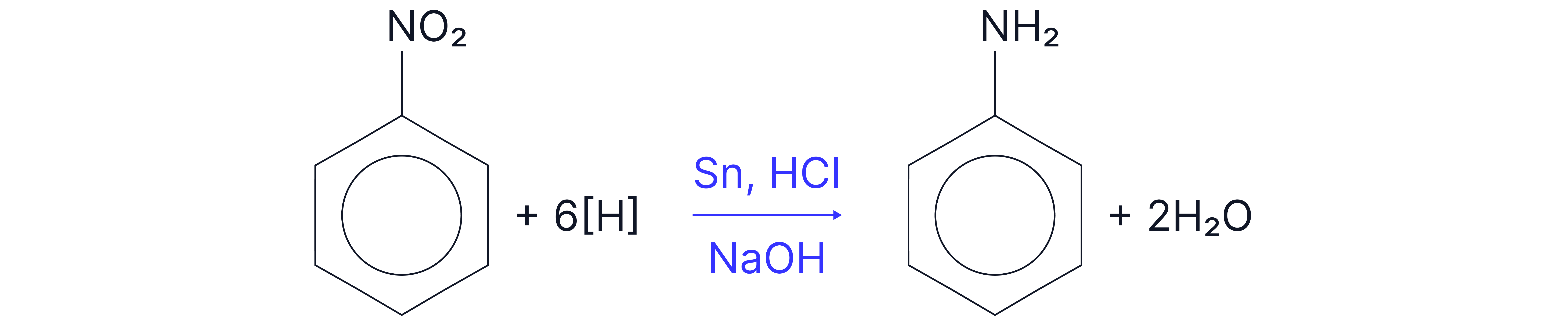
Reagents: Tin and conc. , reflux.
Then add to liberate free amine.
Note – the addition of is not formally assessed by AQA but it is important to acknowledge that under acidic conditions, the amine group would be protonated forming the ammonium salt.

Uses of amines
Quaternary ammonium salts act as cationic surfactants.
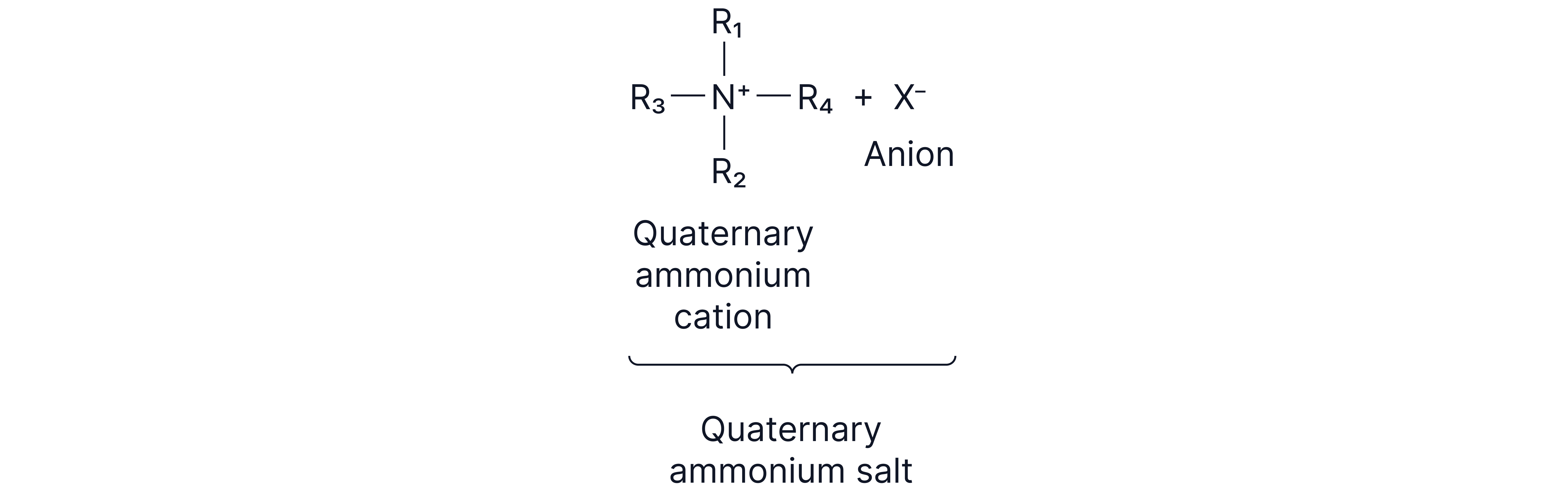
e.g.
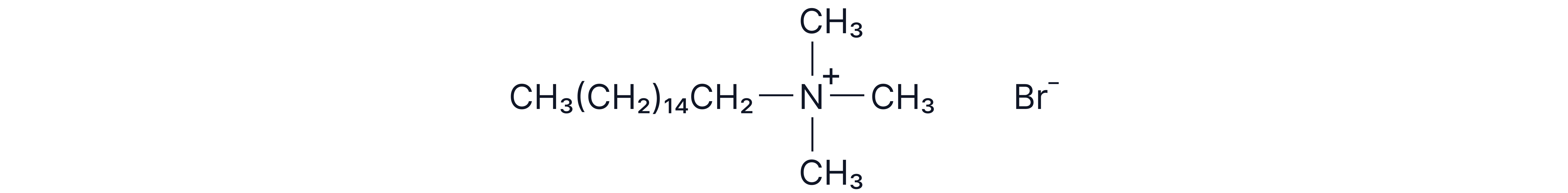
The positive charge on the nitrogen atom makes that part of the molecule hydrophilic (attracted to water)
The non-polar hydrocarbon chains are hydrophobic and do not mix with water.
Cationic surfactants are often used as fabric softeners and hair conditioners as the charged heads are attracted to wet surfaces forming a coating and giving a smoothing effect.
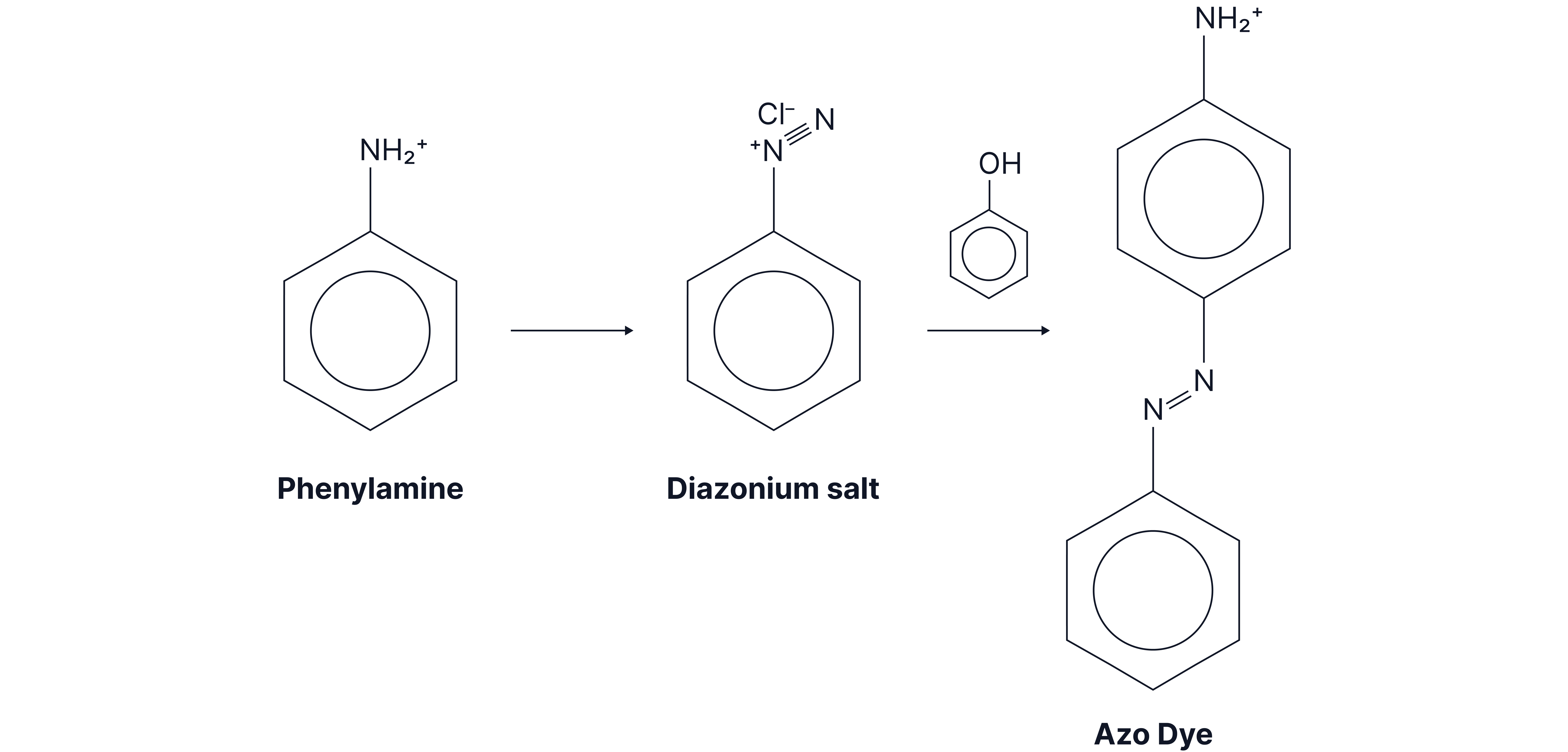
Aromatic amines are used in the manufacture of dyes.
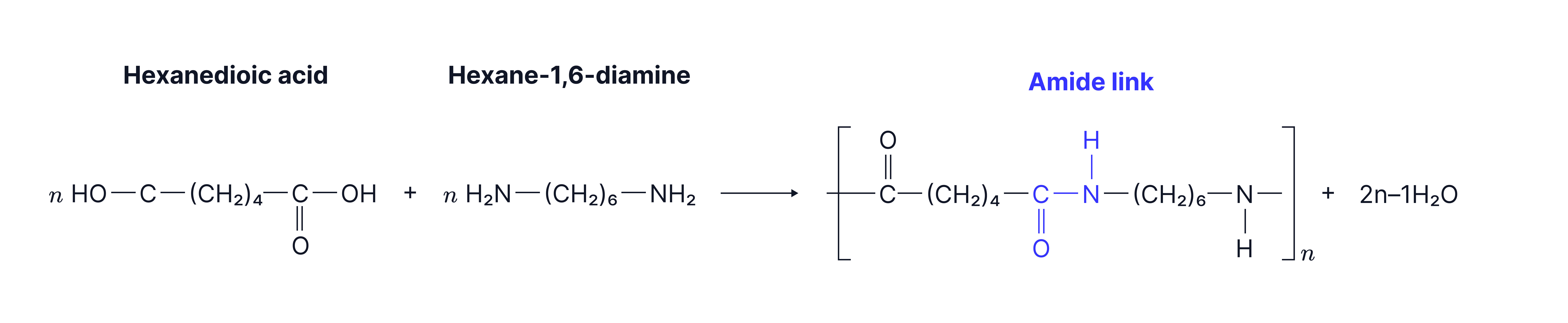
Diamines are used as monomers for condensation polymers called polyamides by reacting them with dicarboxylic acids or acyl chlorides.
Practice Question
Explain why using a nitrile intermediate is better for making a primary amine than direct reaction of a halogenoalkane with ammonia.
Answer
Using ammonia leads to further substitutions forming secondary/tertiary amines (a mixture of products).
The nitrile method produces only one product—primary amine — after reduction therefore giving a higher percentage yield.