Born-Haber Cycles
Dr. Davinder Bhachu
Teacher

Contents
Introduction and Definition
Lattice enthalpy is the energy change when 1 mole of a solid ionic compound is either:
Formed from its gaseous ions (lattice enthalpy of formation), or
Broken apart into its gaseous ions (lattice enthalpy of dissociation)
It’s a measure of the strength of the ionic bonds in a compound. The more negative the lattice enthalpy, the stronger the electrostatic attractions holding the lattice together.
What affects lattice enthalpy?
The strength of ionic bonding can be explained using Coulomb’s Law:
Where:
= electrostatic force of attraction
and = charges on the individual ions
= distance between the centres of the ions (i.e. sum of their ionic radii)
Larger numerator = stronger attraction
Smaller denominator = stronger attraction
From this, we can see that stronger ionic bonds occur when:
The charges on the ions are higher
The ions are smaller, so they can get closer together
This explains why:
(1) has a more negative lattice enthalpy than
Charges:
and → numerator = 2 × 2 = 4
and → numerator = 1 × 1 = 1
Ionic radii:
,
,
has both a larger numerator and a smaller denominator compared to .
Result: has a much stronger ionic bond than .
(2) has a more negative lattice enthalpy than
Charges:
Both compounds involve and a halide ion ( or ), so the numerator (1 × 1 = 1) is the same.
Ionic radii:
Since is smaller, the separation (r) in is less than in .
Result: has a stronger ionic bond than because it has a smaller denominator (smaller r).
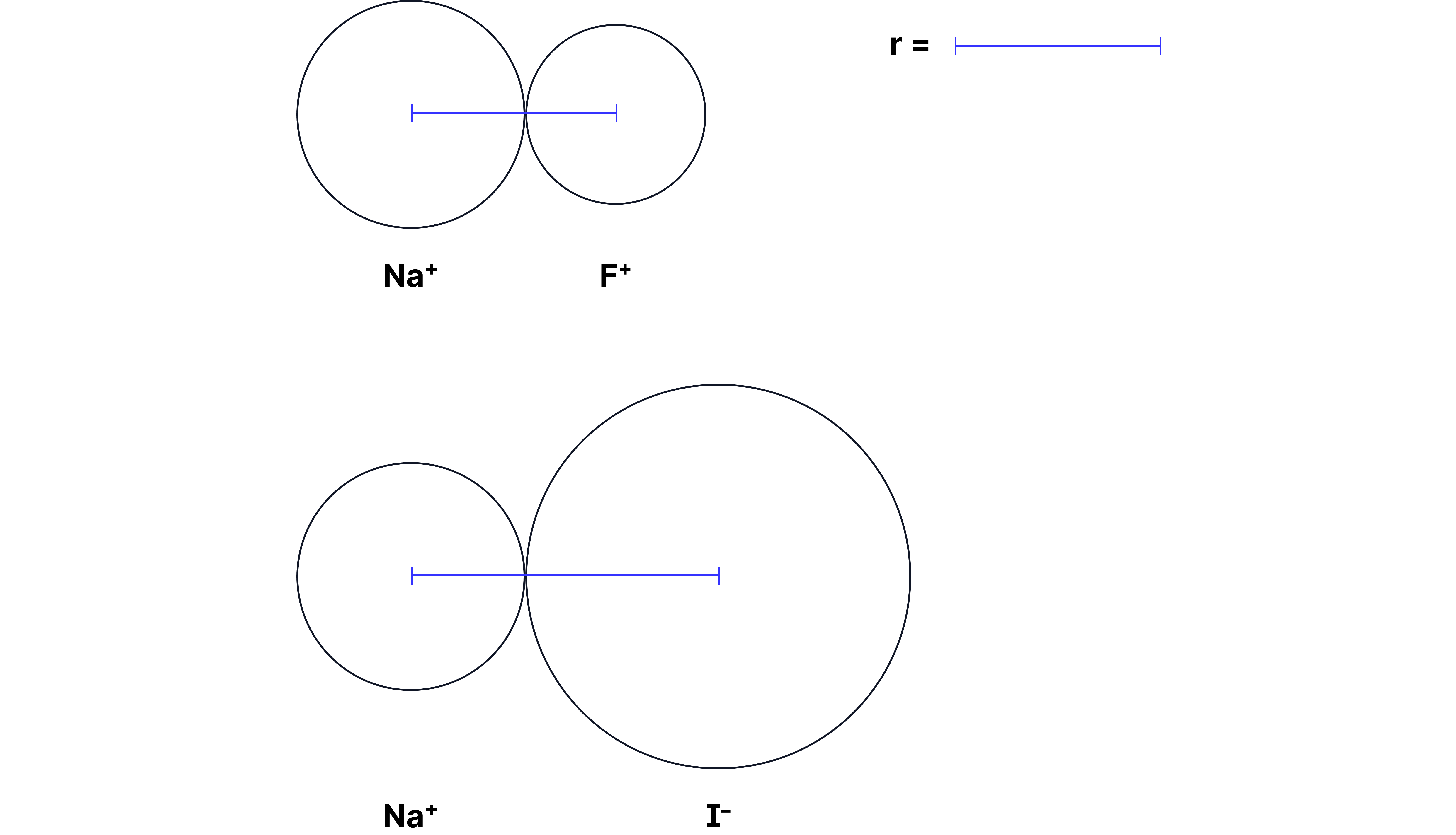
Caption: Diagram showing the variance of r for vs
Common misconception: what if there are two ions?
Some students mistakenly think that in compounds like , you should treat the lithium ion as having a charge of +2 because there are two ions.
This is incorrect.
Coulomb’s Law applies to the interaction between one pair of ions.
In , you consider the attraction between a single ion and the ion. The second forms a separate, identical interaction.
So you always use q₁ = +1 , q₂ = –2 when applying the formula.
Each ion–ion interaction is calculated individually – never combine charges across multiple ions.
Question:
Arrange the following compounds in order of most negative to least negative lattice enthalpy. Explain your reasoning using charge and ionic radius:
magnesium oxide, aluminium oxide, calcium oxide
Answer
Order of decreasing lattice enthalpy:
> >
1. Aluminium oxide
Charge: has a +3 charge, has a –2 charge → very strong electrostatic attraction
Radius: has a very small ionic radius
Conclusion: High charges and small radii give exceptionally strong ionic bonding → most negative lattice enthalpy
2. Magnesium oxide
Charge: and have lower charges than , but still significant
Radius: is smaller than , which increases attraction
Conclusion: Weaker than due to lower charge, but stronger than due to smaller radius
3. Calcium oxide
Charge: Same as → and
Radius: is larger than , so ionic centres are further apart
Conclusion: Weaker electrostatic forces due to larger ion → least negative lattice enthalpy
Measuring Lattice Enthalpy
We can calculate the lattice enthalpy of an ionic compound by using a Born–Haber cycle. This method gives an experimental value because it’s based on data from experiments – not calculations from theory.
A Born–Haber cycle is a type of energy diagram that shows all the enthalpy changes involved in forming an ionic compound from its elements in their standard states.
In these cycles:
The lattice enthalpy of formation is usually shown (this has a negative value, because it’s an exothermic process).
The idea is that you can use the cycle to calculate one unknown enthalpy change (usually the lattice enthalpy) if all the others are known.
You apply Hess’s Law: the total enthalpy change around the cycle = 0.
Key points when drawing a Born–Haber cycle:
Draw every individual step separately – include:
Atomisation of the metal and non-metal
Each ionisation enthalpy (first, second, etc.)
Each electron affinity (don’t combine them!)
Second and third electron affinities are endothermic, not exothermic – so draw these arrows going up.
Label each step clearly. Include either:
The numerical value of the enthalpy change (if given), or
The name of the step (e.g. ΔHat for atomisation, ΔHi for ionisation)
Everything about Born-Haber Cycles
Constructing a Born-Haber Cycle
Consider the reaction of sodium with chlorine:

Enthalpy changes in the Born–Haber cycle for
Atomisation enthalpy of sodium:
First ionisation energy of sodium:
Atomisation enthalpy of chlorine:
First electron affinity of chlorine:
Lattice formation enthalpy:
Standard enthalpy of formation (the arrow from the elements to NaCl):
Step 1 – Apply Hess's Law:
Rearrange to find lattice enthalpy:
Step 2 – Substitute values:
Consider the Born-Haber cycle of
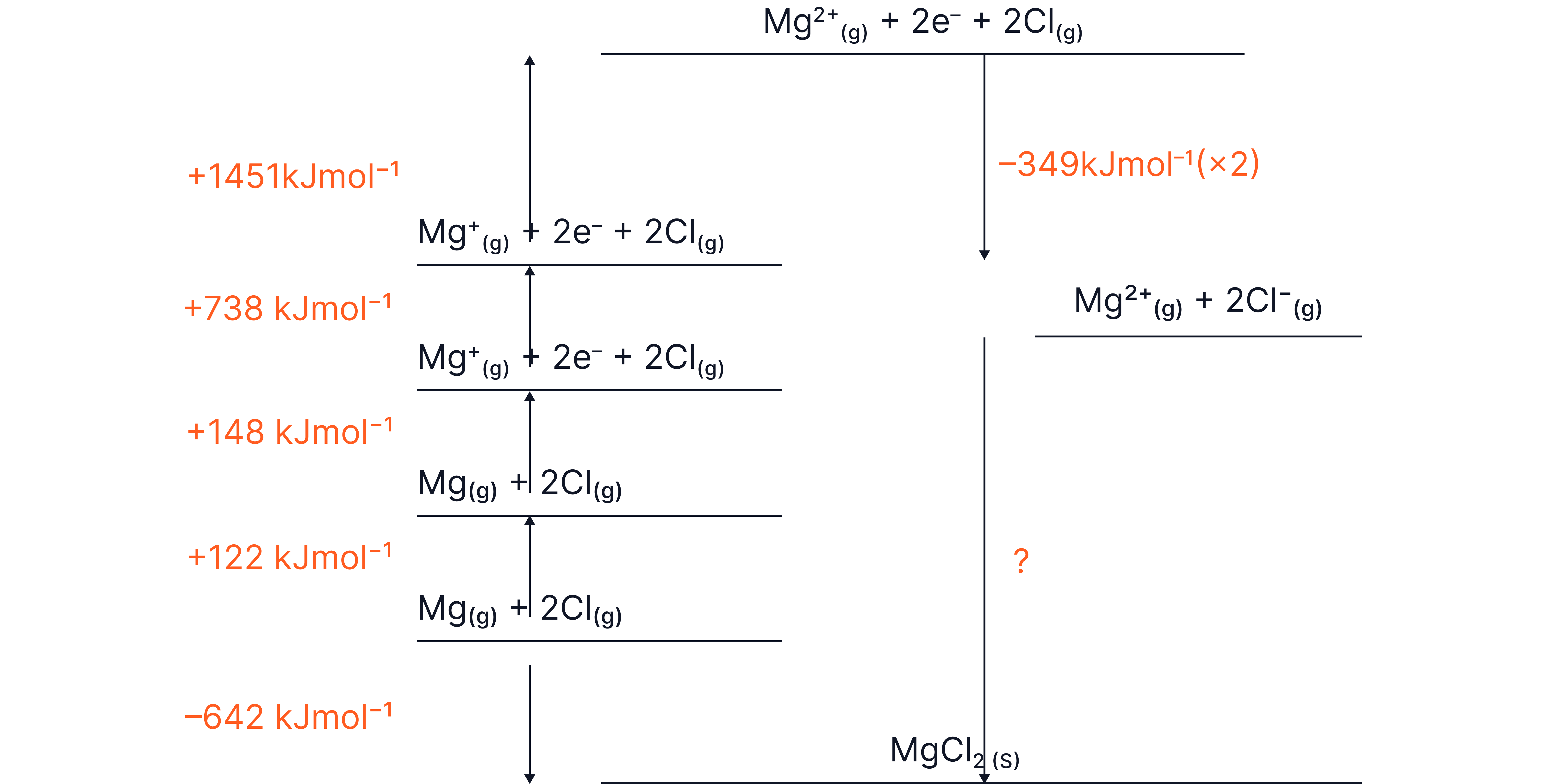
Step | Enthalpy change (kJ mol⁻¹) | Description |
|---|---|---|
–642 | Enthalpy of formation of | |
+148 | Atomisation of : → | |
+738 | 1st ionisation energy: → + | |
+1451 | 2nd ionisation energy: → + | |
+244 | Atomisation of 1 mole : → | |
–349 × 2 = –698 | Electron affinity: + → | |
? | Lattice enthalpy of formation (to be calculated) |
Step 1 – Apply Hess’s Law
Rearranged:
Step 2 – Substitute the values
Practice Questions
enthalpy of atomisation | 107 | 90 | 193 | 314 | 248 | |
1st ionisation enthalpy | 496 | 590 | 577 | |||
2nd ionisation enthalpy | 4562 | 3070 | 1150 | 1820 | ||
3rd ionisation enthalpy | 6910 | 4600 | 4940 | 2740 | ||
1st electron affinity | -142 | -364 | ||||
2nd electron affinity | 798 |
1. Using the data provided, calculate the 1st ionisation enthalpy of potassium, given that the enthalpy of formation of potassium chloride is and its lattice enthalpy is .
Answer
Given:
Enthalpy of formation of =
Lattice enthalpy of =
Enthalpy of atomisation of =
½ Enthalpy of atomisation of =
1st electron affinity of =
1st ionisation enthalpy of = ?
Step 1 – Apply Hess’s Law:
Rearranged:
Step 2 – Substitute values:
2. Calculate the enthalpy of atomisation of chlorine, given that the enthalpy of formation of calcium chloride is and its lattice enthalpy is .
Answer
Given:
Lattice enthalpy
Enthalpy of atomisation of
1st ionisation of
2nd ionisation of
1st electron affinity of
Atomisation of
Step 1 – Apply Hess’s Law:
atomisation of Lattice enthalpy
Rearranged:
Atomisation of
Step 2 – Substitute values:
Atomisation of
Atomisation of
Atomisation of
Atomisation of
Note: This is for 1 mole of → , so the atomisation of 1 mol of atoms .
3. Work out the enthalpy of formation of sodium oxide, given that the lattice enthalpy of formation is .
Answer
Step 1 – Write the formation equation:
Step 2 – Use data provided:
Atomisation of
1st ionisation energy of
Atomisation of
1st electron affinity of
2nd electron affinity of
Lattice enthalpy
Step 3 – Apply Hess’s Law:
Step 4 – Substitute values:
Final answer:
Enthalpy of formation of sodium oxide
4. Determine the lattice enthalpy of formation of aluminium oxide, given that the enthalpy of formation of aluminium oxide is .
Answer
Step 1 – Formation equation:
Step 2 – Data used:
Atomisation of
Ionisation of
1st
2nd
3rd
Total
Atomisation of (to get 3 atoms)
1st electron affinity of
2nd electron affinity of
Enthalpy of formation of
Lattice enthalpy = ?
Step 3 – Apply Hess’s Law:
Rearranged:
Lattice enthalpy
Step 4 – Substitute values:
Lattice enthalpy
Lattice enthalpy
Lattice enthalpy
Final answer:
Lattice enthalpy of formation of