Comparing Lattice Enthalpies
Lajoy Tucker
Teacher

Contents
Comparing Experimental and Theoretical Lattice Enthalpy Values
What is lattice enthalpy?
Lattice enthalpy is the energy change when 1 mole of an ionic solid forms from its gaseous ions. It tells us how strong the ionic bonding is, the more negative the value, the stronger the bonds.
Two Types of Lattice Enthalpy Values
Type | How it’s calculated | What it assumes |
|---|---|---|
Experimental value | Measured using a Born-Haber cycle from experimental data | Based on real compounds |
Theoretical value | Calculated using ion charges and sizes in a perfect ionic model | Assumes the ions are perfect spheres and bonding is 100% ionic |
Which one is correct?
The experimental value is the real one – it reflects what actually happens in nature.
Why might the values be different?
Sometimes, the ions in a compound are not perfect spheres. If the positive ion is small and highly charged, it can pull on (polarise) the electron cloud of the negative ion. This causes distortion, and the bonding is no longer purely ionic – we call this covalent character.
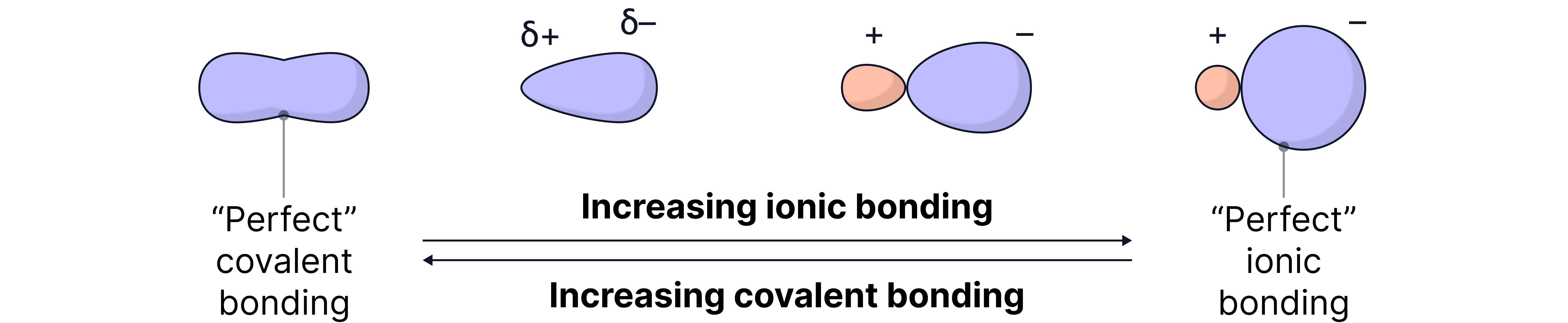
Caption: This diagram shows the spectrum between perfect covalent bonding and perfect ionic bonding. As you move from left to right, ionic character increases – the electron cloud becomes more separated between ions. From right to left, covalent character increases – the positive ion distorts the electron cloud of the negative ion, leading to a more shared (and less perfectly ionic) distribution of electrons.
Key Trends and Explanations
Small, highly charged cations (e.g. ) strongly attract and distort nearby anions.
Large anions (e.g. ) are easily distorted – they are said to be polarisable
The more distortion there is, the more covalent character the bond has.
The bigger the difference between experimental and theoretical values, the greater the covalent character.
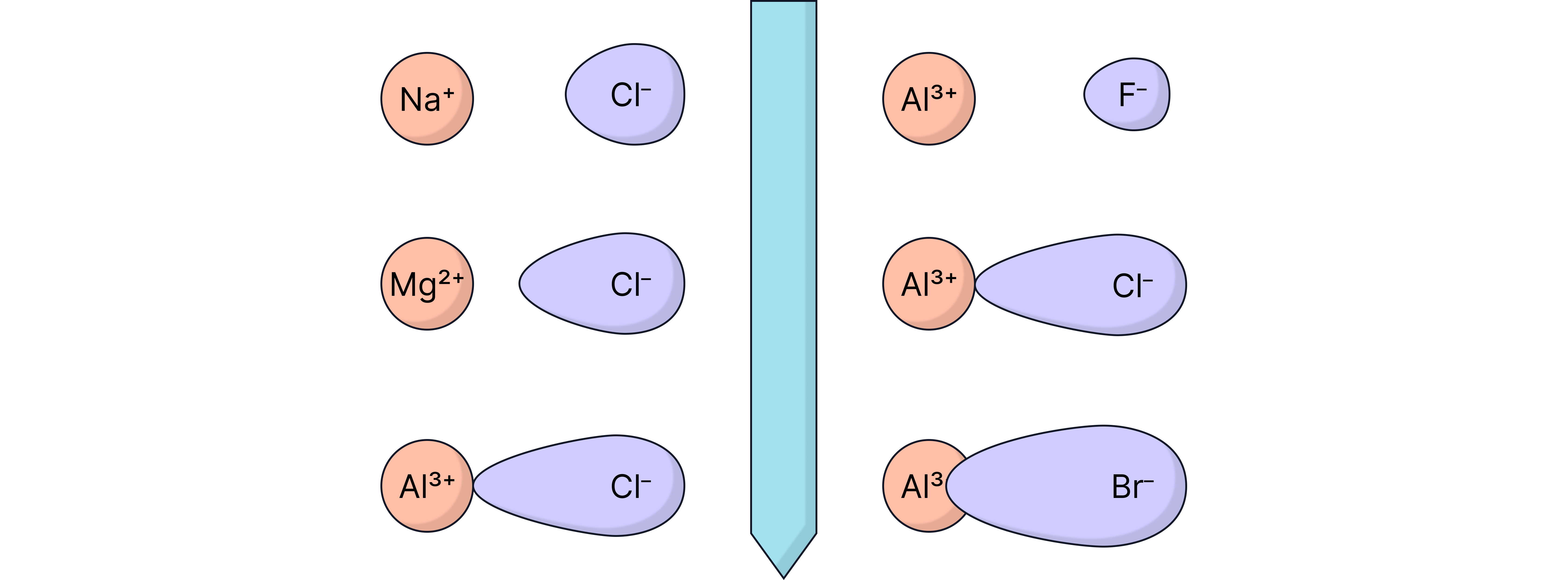
Comparison of Experimental and Theoretical Lattice Enthalpy Values
Compound | Experimental | Theoretical | Difference | % Difference | Notes |
Classic example of almost pure ionic bonding | |||||
Very strong ionic bonding and both highly charged | |||||
Small strongly polarises large more covalent character than | |||||
Large polarisation by and large strong covalent character | |||||
High covalent character – small, soft ion highly distorting | |||||
Very strong bonding overall – small gives partial covalent character |
When the experimental lattice enthalpy is significantly more negative than the theoretical value, it indicates that the bonding is not purely ionic. The presence of covalent character – caused by polarisation of the anion by a small, highly charged cation – strengthens the electrostatic attraction. This results in a more exothermic lattice enthalpy than predicted by the purely ionic model.
Practice Questions
Question 1
Use the data in the table below to answer the questions that follow. All values refer to lattice enthalpies.
Substance | |||
Experimental value | |||
Theoretical value |
a) Do these values represent lattice enthalpies of formation or dissociation? Explain.
b) Which of the compounds has the strongest bonding? How can you tell?
c) Which compound displays the greatest covalent character? Support your answer with data.
d) Copper chloride is poorly soluble in water, whereas potassium chloride dissolves easily. Use the enthalpy data to explain this difference.
Answer
a) These values represent lattice enthalpies of formation because they are negative, indicating that energy is released when 1 mole of an ionic solid forms from its gaseous ions.
b) has the most negative experimental lattice enthalpy , meaning it has the strongest bonding.
c) shows the largest difference between experimental and theoretical values:
This suggests significant covalent character, likely due to the small and polarising Cu⁺ ion distorting the Cl⁻ ion.
d) is poorly soluble in water because the strong bonding due to covalent character makes it harder for water to break apart the lattice.
, which is more purely ionic, dissolves easily because the ionic lattice separates more readily in water.
Question 2
The lattice enthalpy of formation of magnesium oxide is . The value predicted using a perfect ionic model is .
a) Calculate the percentage difference between these two values.
b) Magnesium oxide is almost completely insoluble in water. Explain why in terms of energy changes.
Answer
Magnesium Oxide
a) Experimental value
Theoretical value
Difference
Percentage difference
b) Magnesium oxide is almost insoluble because of its very strong ionic bonding between and . The lattice enthalpy is very large, and the hydration enthalpy is not enough to compensate, making it energetically unfavourable for the lattice to dissolve.