Enthalpy of Solution & Hydration
Dr. Davinder Bhachu
Teacher

Contents
Introduction to Enthalpy of Solution & Hydration
Enthalpy of Solution
The enthalpy of solution is the energy change when 1 mole of an ionic solid dissolves in water to form an aqueous solution of its ions.
Born-Haber Cycles Explainer Video
Hess cycle
It can be calculated using a Hess cycle, because it’s difficult to measure directly.
What steps are involved?
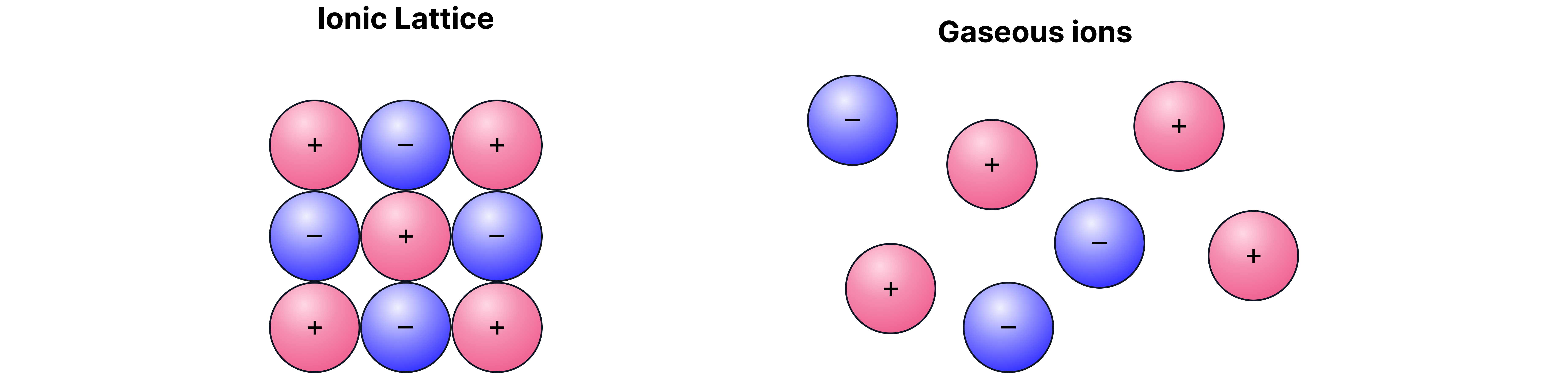
To dissolve an ionic solid like , we imagine it happening in two stages:
1. Break up the solid lattice into separate gaseous ions
→ This is the lattice enthalpy ().
→ Always endothermic ( is positive).
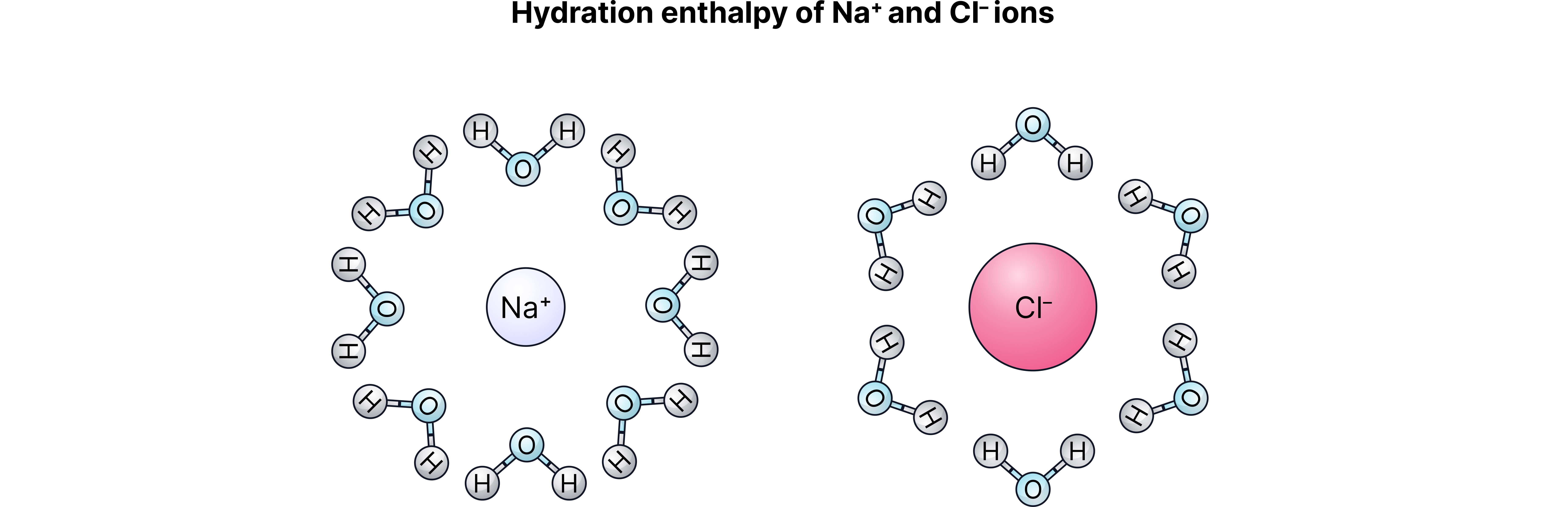
2. Hydrate the gaseous ions by surrounding them with water molecules
→ This is enthalpy of hydration ().
→ Always exothermic ( is negative).
→ This is where the water molecules and ions interact through ion/dipole attractions. Note the on the oxygen oriented towards the cation and the on the anion oriented towards the anion.
Where:
is positive (energy in to break the lattice)
is the sum of negative hydration values
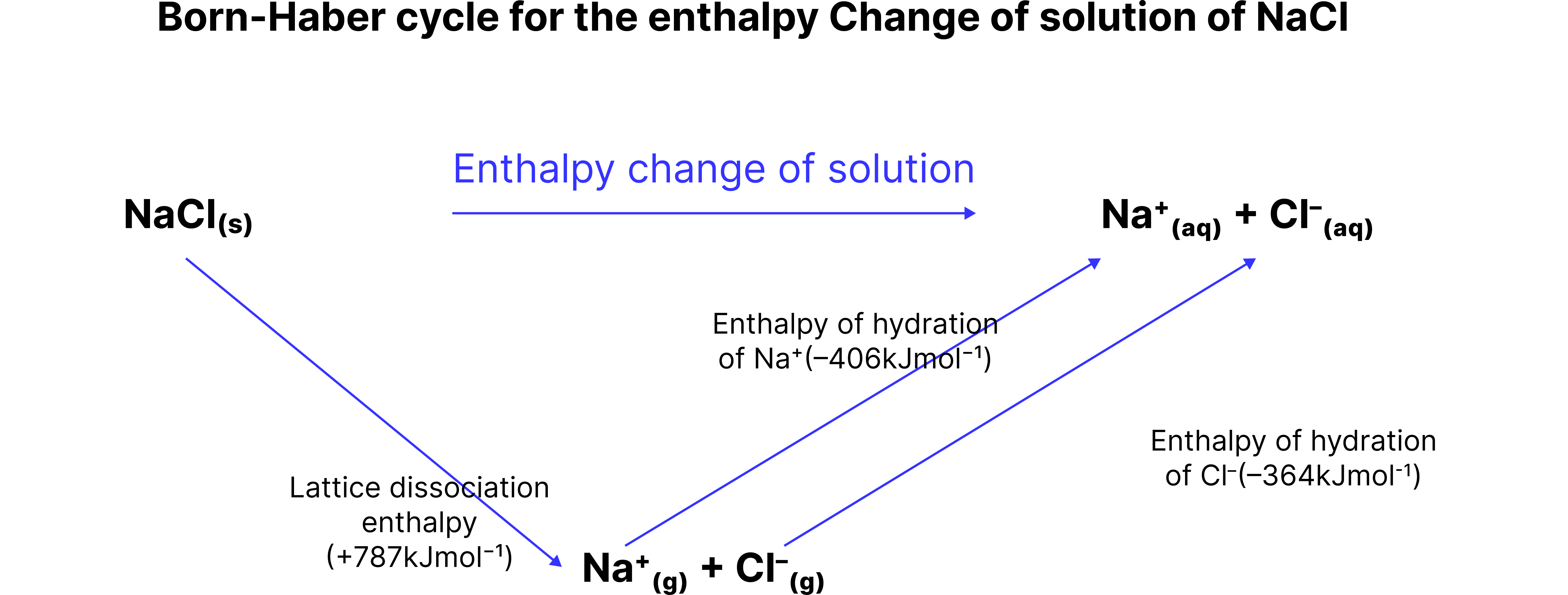
What does the sign of tell you?
If is negative, the solution process is exothermic → often dissolves easily.
If is positive, dissolving requires energy → may not dissolve well unless entropy favours it.
Worked Example
Enthalpy of Solution for
Reaction:
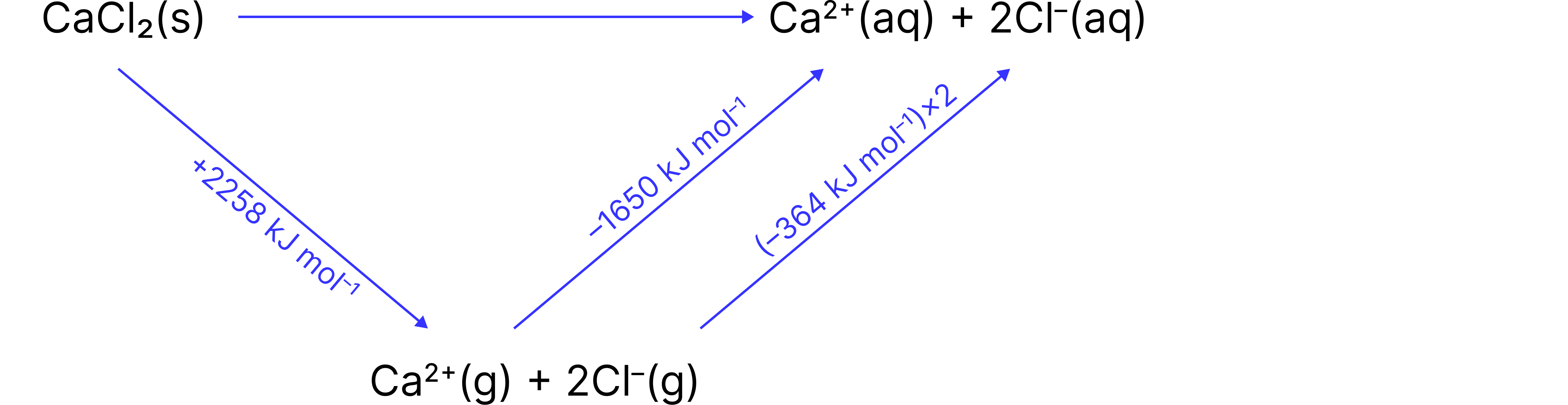
Given data:
Lattice enthalpy of
Enthalpy of hydration of
Enthalpy of hydration of (per ion)
Answer
Step 1: Apply the Hess cycle equation
Step 2: Simplify the hydration enthalpies
Total hydration
Step 3: Calculate
Final Answer:
Conclusion
This process is exothermic. dissolves easily in water and releases heat.