Entropy
Dr. Davinder Bhachu
Teacher

Contents
Introduction and Definitions
Endothermic Process
We have defined enthalpy as the driving force for chemical changes. Processes lead to increased stability if they lead to a lower energy state. Exothermic reactions occur because the reactants are thermodynamically unstable in comparison to their products. Reactions are most often exothermic but may be endothermic.
To predict feasibility, a new concept is needed: the dissipation of energy (Entropy).
Entropy is also a driving force for physical and chemical changes (reactions).
What is entropy?
Entropy (S) is a measure of disorder or randomness in a system.
The more disordered the particles are, the higher the entropy.
Entropy is measured in joules per mole per kelvin – this tells us how much disorder there is per mole of particles for each degree of temperature.
Solids have the lowest entropy – particles are locked in fixed positions and only vibrate slightly.
Liquids have more entropy – particles can move around each other more freely.
Gases have the highest entropy – particles are far apart and move rapidly and randomly.
The Second Law of Thermodynamics states that in any spontaneous process, the total entropy of the universe increases. In other words, things naturally become more disordered over time.
Entropy isn’t just about messiness – it’s a quantifiable measure of how energy and particles are spread out.
State of matter and entropy :
State | Particle behaviour | Entropy level |
|---|---|---|
Solid | Particles vibrate in fixed positions | Low entropy |
Liquid | Particles can move around each other | Medium entropy |
Gas | Particles move freely and randomly | High entropy |
So:
S(gas) > S(liquid) > S(solid)
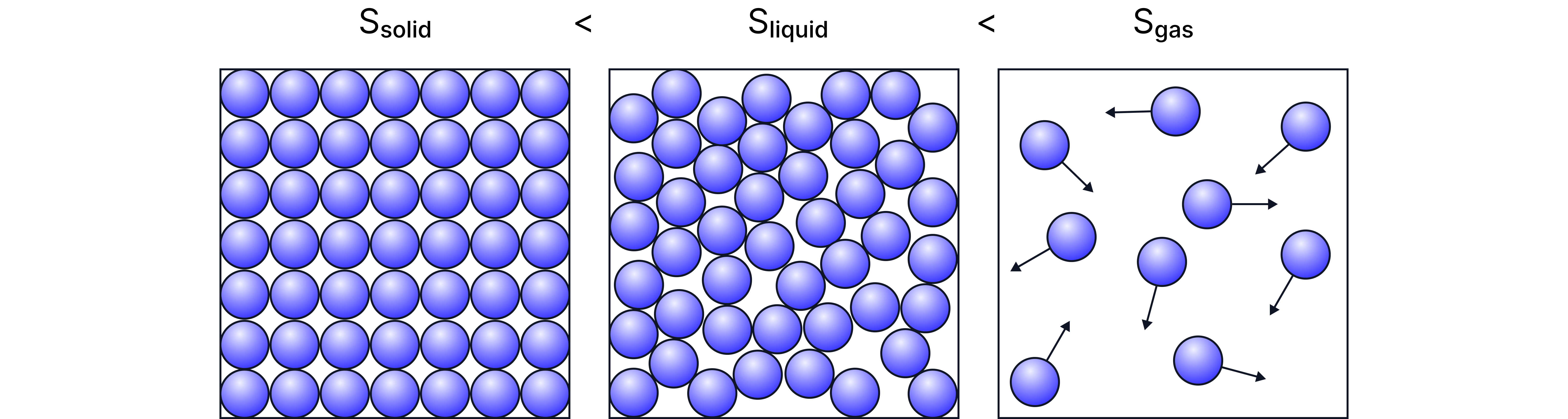
Why are gases so disordered?
Gas particles are spread out and have high kinetic energy, so their positions and movements are highly unpredictable.
This makes them have a huge number of possible arrangements – and entropy is a way of counting those possibilities.
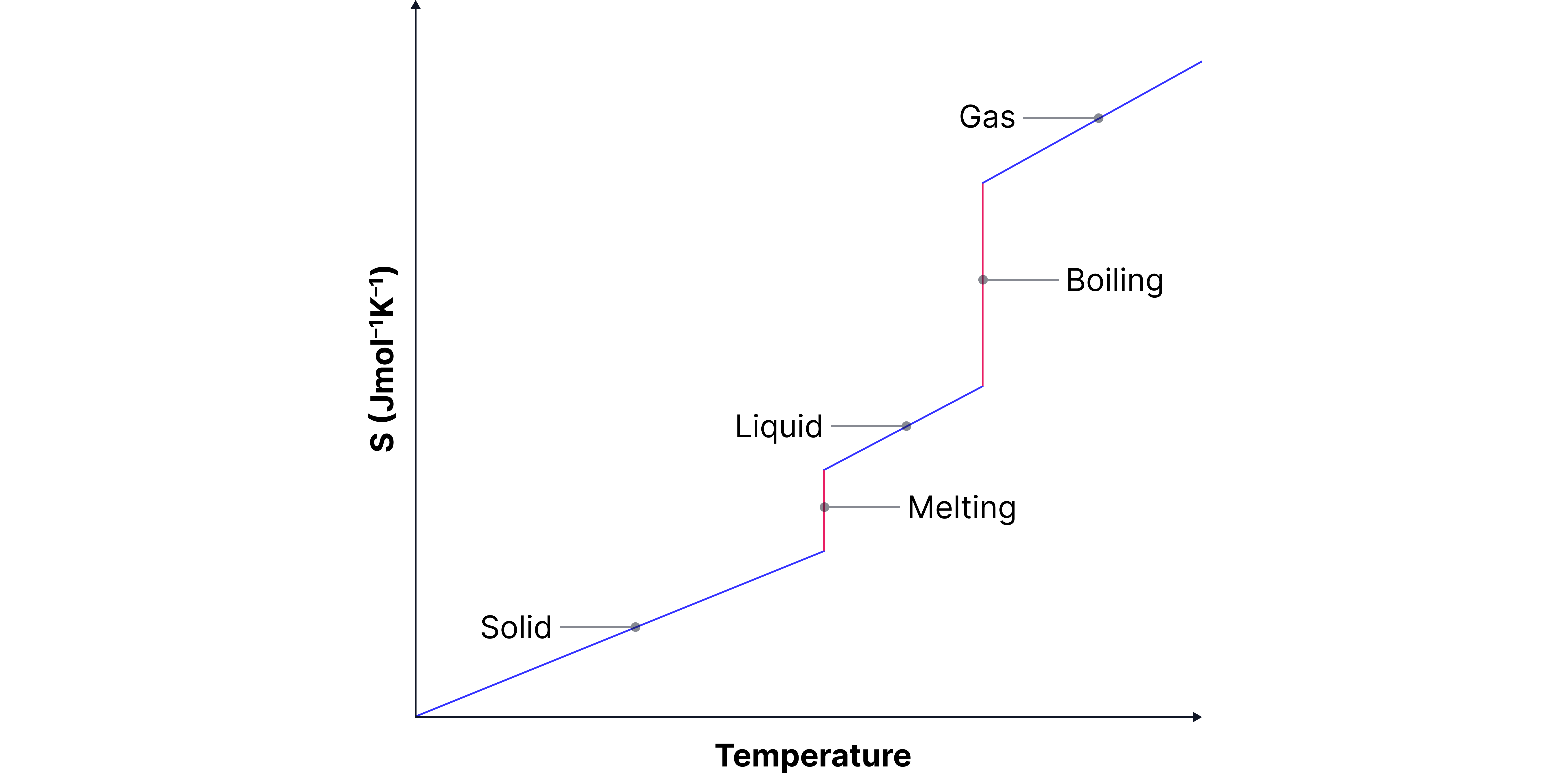
How does entropy change with temperature?
When a substance is cold, particles move slowly → low entropy.
As temperature increases, the particles gain more energy and move more freely – this leads to an increase in entropy (more disorder).
When a substance changes state (e.g. from solid to liquid or liquid to gas), there is a sudden jump in entropy. This is because the particles are no longer restricted in the same way.
The biggest jump in entropy happens during boiling (liquid to gas). Gases are extremely disordered compared to liquids and solids, so the increase in randomness is much greater than it is during melting (solid to liquid).
Important rule:
According to the Third Law of Thermodynamics, a perfectly ordered crystal at absolute zero (0 K) has zero entropy.
Comparing the Entropy of Different Substances
When a substance is very structured, its particles are arranged in an orderly way and can't move around much. This means it has low entropy. For example, in ice, the water molecules are locked into a fixed pattern – they can only vibrate slightly – so the entropy is low.
When ice melts to form liquid water, the molecules can move around each other more freely. This increases the number of possible arrangements of the particles, so the entropy is higher.
When water is heated to form steam (water vapour), the particles spread far apart and move very quickly in all directions. This is the most disordered state, so steam has the highest entropy of the three.
In summary:
Entropy increases from ice → water → steam
S(solid) < S(liquid) < S(gas)
Substance | S/J mol⁻¹ K⁻¹ |
|---|---|
C (diamond) | 2 |
C (graphite) | 6 |
Ice | 41 |
42 | |
72 | |
90 | |
70 | |
189 | |
214 |
Calculating entropy changes in a reaction:
We can work out the entropy change for a chemical reaction using a simple formula:
= total entropy of products – total entropy of reactants
(or: = ∑S(products) – ∑S(reactants))
If entropy increases during the reaction, then is positive. This means the products are more disordered than the reactants.
If entropy decreases, then is negative. This means the system has become more ordered.
According to the Second Law of Thermodynamics, reactions that increase entropy (positive ) are generally more likely to happen – they are energetically favourable.
Example 1: Combustion of methane
Reaction:
Given standard entropy values :
Step 1: Add up entropy of products
Step 2: Add up entropy of reactants
Step 3: Calculate
Conclusion:
Entropy decreases slightly (ΔS is negative) – system becomes a little more ordered.
Example 2: Thermal decomposition of calcium carbonate
Reaction:
Given standard entropy values :
Step 1: Add up entropy of products
Step 2: Entropy of reactant
Step 3: Calculate ΔS
Conclusion:
Entropy increases significantly (ΔS is positive) due to formation of a gas – system becomes more disordered.
Entropy Practice Question
Practice Questions
Reaction:
Standard entropy values :
Calculate the standard entropy change () for this reaction.
Answer
Step 1: Entropy of products
Step 2: Entropy of reactants
Step 3: = S(products) – S(reactants)
Reaction:
Standard entropy values :
Calculate the entropy change for this reaction.
Answer
Step 1: Entropy of products
Step 2: Entropy of reactant
= 94
Step 3:
=
Reaction:
You are given the following standard entropy values (S°, in J mol⁻¹ K⁻¹):
unknown
The standard entropy change for the reaction is
Calculate the standard entropy value of .
Answer
Step 1: Calculate total S°(products):
Step 2: Write ΔS equation and plug in known values:
= S(products) – S(reactants)