Gibbs Free Energy
Dr. Davinder Bhachu
Teacher

Contents
Gibbs Free Energy and Feasibility
This equation combines two key thermodynamic ideas:
= change in enthalpy (heat energy transferred)
= change in entropy (change in disorder)
T = temperature in kelvin (K)
What does ΔG tell us?
If is negative or zero ( ≤ 0), the reaction is said to be feasible – meaning it can happen, from a thermodynamic point of view.
If is positive, the reaction is not feasible under those conditions – it will not happen on its own.
Note: Feasible does not mean the reaction will definitely happen. It just means it's thermodynamically allowed. It might still not occur if:
The activation energy is too high, or
The reaction is too slow
What makes a reaction more likely to be feasible?
A negative (exothermic reaction) helps make negative – this is favourable.
A positive (increase in disorder) also helps make negative – this is favourable too.
Common terminology:
Some textbooks use the word spontaneous instead of feasible.
But be careful – spontaneous doesn’t mean it happens quickly, just that it could happen thermodynamically.
The word feasible is clearer and preferred in this context.
How Temperature Affects Feasibility
Here’s how different combinations of ΔH and ΔS affect whether a reaction is feasible at different temperatures:
Effect on | Feasibility | ||
|---|---|---|---|
is always negative | Feasible at all temperatures | ||
is negative at low T, positive at high T | Feasible only at low temperatures | ||
is positive at low T, negative at high T | Feasible only at high temperatures | ||
is always positive | Never feasible |
Example 1 – Combustion of Hydrogen
Reaction:
Given data:
Answer
Step 1 – Convert to :
Step 2 – Use the equation:
Conclusion:
< 0 → combustion of hydrogen is feasible at room temperature (and highly exothermic).
Example 2 – Thermal decomposition of calcium carbonate
Reaction:
Given data:
Answer
Step 1 – Convert :
Step 2 – calculation:
Conclusion:
At , is still positive, so the reaction is not quite feasible. A slightly higher temperature would be needed to make it thermodynamically favourable.
Example 3 – Reaction of nitrogen and hydrogen to form ammonia
Reaction (Haber process):
Given data:
Answer
Step 1: Convert :
Step 2: calculation:
Conclusion:
At , the reaction is not feasible ( > 0), despite being exothermic.
High temperature increases entropy’s unfavourable effect (–TΔS becomes large and positive).
This is why the Haber process uses a compromise temperature (~450–500 °C) and a catalyst to increase the rate.
Gibbs Free Energy Question Answered
Using Free Energy to Predict Feasibility
You can rearrange the Gibbs free energy equation to find the temperature at which a reaction becomes feasible (i.e. when ΔG reaches zero).
We start with:
To find when a reaction becomes feasible, set = 0:
Rearranging gives:
This tells us the exact temperature at which a reaction becomes just thermodynamically allowed.
Below this temperature, the reaction is not feasible.
Above this temperature, it becomes feasible – depending on the signs of and .
Make sure and are in the same units (usually both in or both in ).
Example 1: Decomposition of Ammonium Nitrate
Reaction:
Data:
Answer
Step 1 – Convert to kJ:
Step 2 – Use :
Conclusion:
The decomposition becomes feasible above 407 K. This makes sense – it requires heating to decompose (used in explosives and propellants).
Example 2: Thermal Decomposition of Calcium Carbonate
Reaction:
Data:
Answer
Convert :
Conclusion:
The decomposition becomes feasible above 1106 K. This aligns with its industrial use in lime kilns at high temperature.
Example 3: Combustion of Carbon Monoxide
Reaction:
Data:
Answer
Convert :
Conclusion:
Although combustion is exothermic, the decrease in entropy means it’s only feasible below 3272 K. Since this is very high, the reaction is feasible at all normal temperatures – as expected for a combustion process.
Changes of State and Gibbs Free Energy
The Gibbs free energy equation also applies to physical changes like melting and boiling – not just chemical reactions.
For a substance to melt or boil, the change must be thermodynamically feasible, which means .
Melting:
Below the melting point, melting is not feasible.
→ is positive, so the solid remains solid.
At the melting point, = 0.
→ The solid and liquid are in equilibrium, and melting can happen.
Above the melting point, becomes negative, and melting is feasible.
Boiling:
Below the boiling point, boiling is not feasible.
→ is positive, so the liquid stays as a liquid.
At the boiling point, = 0.
→ The liquid and gas are in equilibrium, and boiling becomes possible.
Above the boiling point, is negative, and boiling is feasible.
Key Idea:
Just like chemical reactions, phase changes only happen when they are thermodynamically allowed .
You can calculate the melting or boiling temperature using the rearranged Gibbs equation:
(remember to convert units: in , in )
Example: Determining the melting point of ice using ΔG = 0
When ice melts, the process can be analysed using the Gibbs free energy equation.
Reaction:
Given data:
Enthalpy change for melting ice,
Entropy change for melting ice,
Answer
Step 1 – Convert entropy to :
Step 2 – Use the formula:
At the melting point, = 0, so:
Conclusion:
The melting point of ice is .
Below this temperature, melting is not feasible ( > 0).
At and above this temperature, melting becomes thermodynamically feasible.
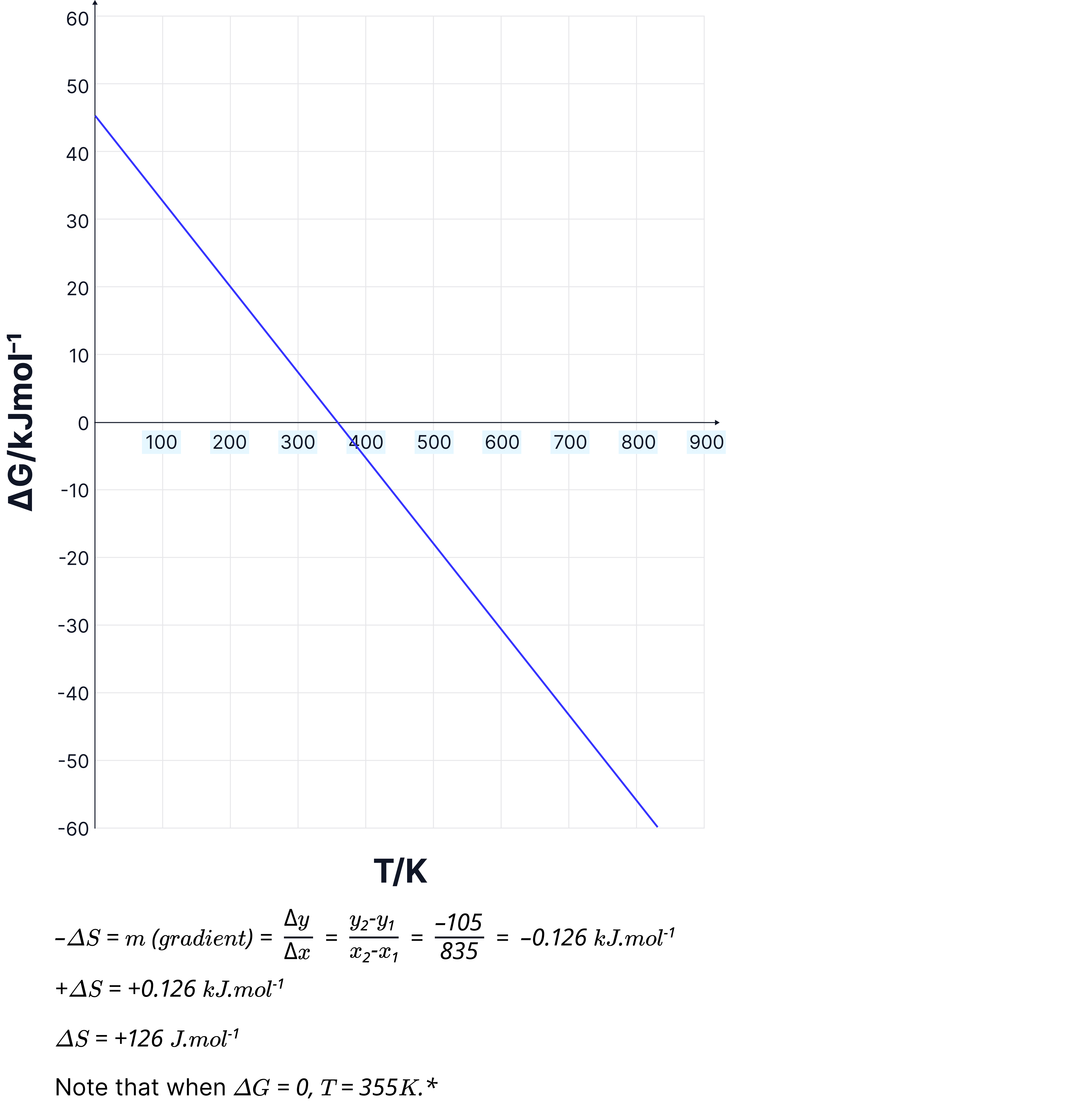
Graphing Gibbs Free Energy
We can turn the Gibbs equation into a straight-line graph to help understand how temperature affects feasibility.
Rewriting the equation:
Start with:
Compare this to the straight-line form:
Now match the terms:
x = T (temperature in K)
m (gradient) =
c (y-intercept) =
What the graph looks like:
The graph is a straight line, plotting (y-axis) against T (x-axis)
The gradient is negative if is positive (and vice versa)
The point where the line crosses the x-axis is when = 0
→ this is the temperature at which the reaction becomes feasible
Key interpretation points:
Below the x-intercept ( > 0) → reaction is not feasible
Above the x-intercept ( < 0) → reaction is feasible
The x-intercept can be found using:
(make sure units match)
Why is this useful?
It helps you visualise how changes with temperature
You can predict feasibility over a range of temperatures
It shows why some reactions are only feasible at high or low temperatures
Example shown below:
Practice Questions
Question 1
a) Calculate the entropy change at for the following reaction:
b) Explain the sign and size of the entropy change in terms of the number and state of particles involved.
Answer
a) Calculate at
Given:
Use the rearranged Gibbs equation:
Substitute values:
=
b) Explain the sign and magnitude of
The entropy change is negative, which makes sense:
The reaction goes from
There's a loss of gas particles, which are highly disordered
Fewer particles overall → less disorder → decrease in entropy
The value is relatively large because gases contribute significantly to entropy, and 1.5 moles of gas are being removed.
Question 2
a) Calculate , , and at 298 K for the reaction below:
Given values , in :
b) Based on your values, decide whether the reaction is feasible at 298 K.
c) Calculate the temperature at which this reaction becomes feasible. Give your answer in kelvin.
Answer
a) Calculate , and
Step 1: (in )
Convert to kJ:
Step 2:
b) Will the reaction be feasible at 298 K?
No – because is positive, the reaction is not feasible at 298 K.
c) Calculate the temperature at which it becomes feasible
Set use:
Conclusion:
The reaction only becomes thermodynamically feasible above ~1071 K.