Buffers
Lajoy Tucker
Teacher

Contents
Introduction to Buffers
Key Definitions:
Buffer solution: A solution that resists changes in when small amounts of acid or base are added and on dilution.
Weak acid: An acid that only partially ionises in aqueous solution ‘
e.g.
Conjugate base: The deprotonated form of an acid e.g. for the acid , the conjugate base is .
Acidic buffer: Formed from a weak acid and its conjugate base e.g. and .
Basic buffer: Formed from a weak base and its conjugate acid e.g. and (calculations NOT required).
Buffers Explained
Buffer action
Buffers resist changes in pH on addition of small amounts of acid or base according to Le Chatelier’s Principle which says:
“When a system is at dynamic equilibrium and a change is made to any of the conditions, then the system responds to counteract the change.”
For an acidic buffer:

Worked Example
Explain how the buffer system in blood maintains an approximately constant pH on addition of small amount of acid.
Answer
increases
Eqm shifts to the left by reaching with . Thus decreasing/restoring the and pH remains approx constant.
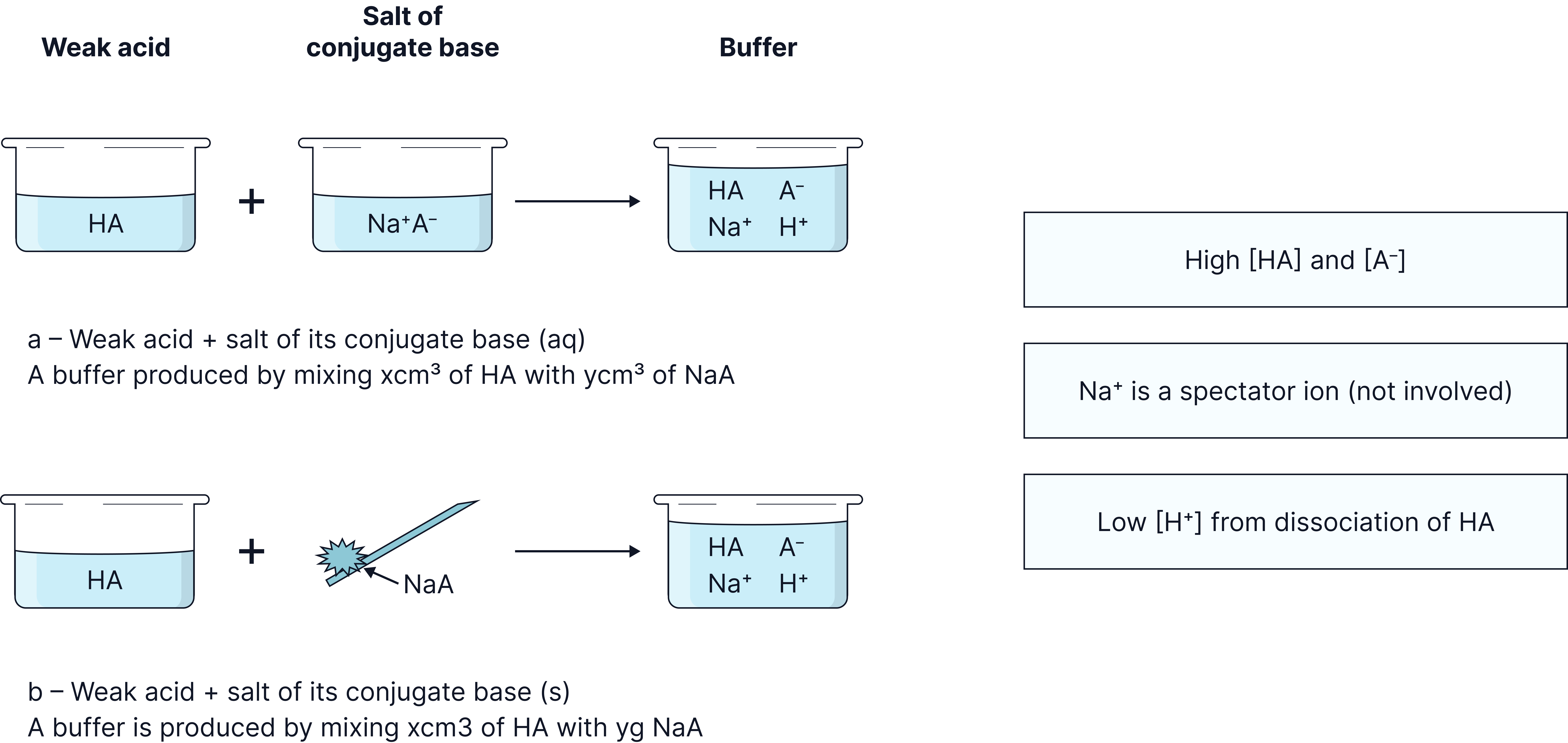
Buffer formation
For the previous systems to work, a buffer solution must contain sufficient weak acid and its conjugate base .
Weak acids only partially ionise meaning that the concentration will be very low.
Therefore, to have enough and , the following methods can be used:
Method 1: Mix the weak acid with the salt of its conjugate base e.g. ethanoic acid and sodium ethanoate .
Calculate moles of and concentrations
Substitute into expression
Worked Example
The concentrations of ethanoic acid and sodium ethanoate, in a buffer solution, are and , respectively. Calculate the pH of this buffer solution.
Answer
Worked Example
A buffer solution is made from of ethanoic acid and of sodium ethanoate. Calculate the pH of this buffer solution.
Answer
Calculate concentrations in combined solution using .
Substitute into rearranged expression.
Calculate from .
Method 2: Partially neutralise a weak acid with a strong base
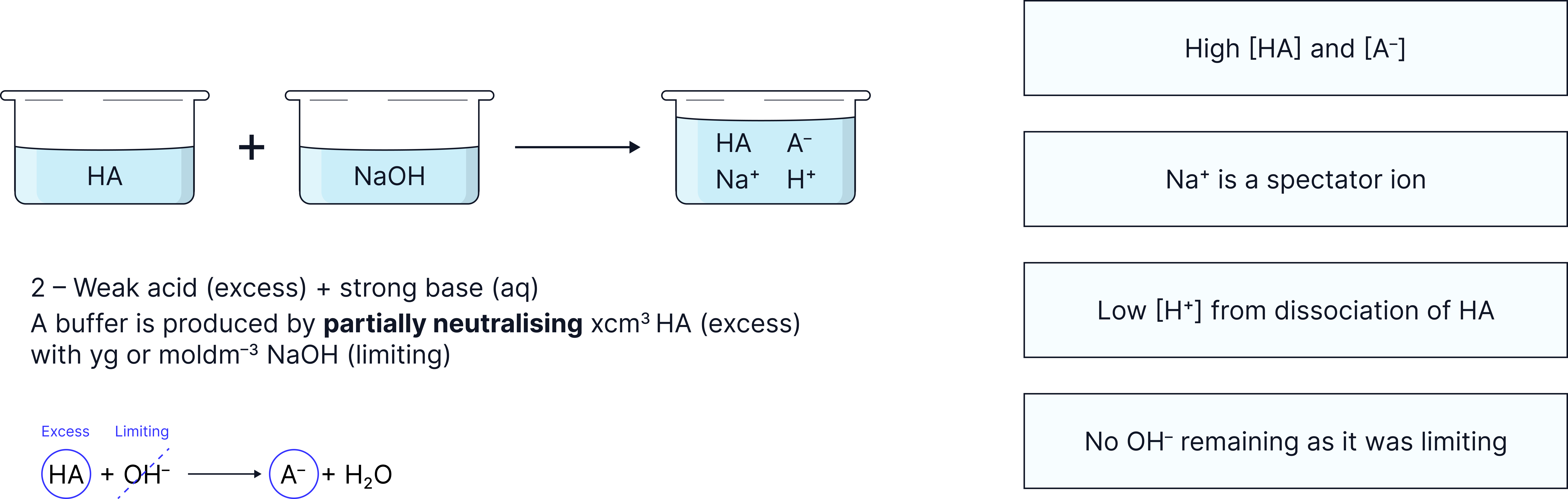
Calculate moles of leftover and produced at the end of reaction .
Substitute into
Worked Example
An acidic buffer is made from reacting of of a weak acid, , with of .
Calculate the pH of the buffer solution.
Answer
Use the ICE method outlined in the ‘Reactions between Acids and Bases’ note to deduce moles of HA and A- once the reaction is complete.
Equation | |||||||
Initial moles (I) | (excess) | (limiting) | Moles of water not required for future pH calculation | ||||
Change in moles (C) | |||||||
End moles (E) |
Total volume is the same so moles can be used
Buffer Action Calculations
change being minimised on addition of small amounts of acid or based can be demonstrated quantitatively.

Worked Example
The concentrations of ethanoic acid and sodium ethanoate, in a buffer solution, are and , respectively.
of is added to of this buffer solution. Calculate the pH of this solution.
Answer
added added
Addition of shifts eqm to the left thus increasing moles of and decreasing moles of .
Buffer system | |||||
Initial moles (I) | To be determined for pH | ||||
Change based on moles of | |||||
End moles (E) |
Tip: Can also be calculated from moles alone as the volumes cancel top and bottom
Worked Example
A buffer is prepared by mixing of propanoic acid, , with of sodium propanoate, , at .
of is then added. Assuming volume effects are negligible, calculate the final of the solution.
Answer
so
Buffer system | |||||
Initial moles (I) | To be determined for pH | ||||
Change based on moles of | |||||
End moles (E) |
Final
Adding metals to buffers
Metals behave similarly to bases in that they react with acids. This shifts the position of the buffer system equilibrium to the right, decreasing and increasing .
e.g. if z moles of Mg is added to a buffer system
The ratio in which the moles increase or decrease depend in the group of the metal.
If z moles of metal added…
Group 1 = 1:1 ratio
Effect on buffer moles
Group 2 = 1:2 ratio
Effect on buffer moles
Group 3 = 1:3 ratio
Effect on buffer moles
Worked Example
A buffer solution contains of sodium ethanoate and of ethanoic acid.
A sample of of magnesium is added to this buffer solution.
Calculate the of the solution formed.
For ethanoic acid, at .
Answer
Buffer system | |||||
Initial moles (I) | To be determined for pH | ||||
Change based on moles of (group 2) | |||||
End moles (E) |
Diluting buffer solutions
Buffer solutions also resist changes in on dilution.
Dilution increases the overall volume of the solution without number of moles present
Learning Check
Which of the following mixtures will form an acidic buffer solution?
A. and
B. and
C. and
D. and
Answer
C – partial neutralisation of a weak acid by a strong base
Key Tips and Reminders
Acidic buffers contain a weak acid and the salt of their conjugate base
changes are minimised based on Le Chatelier’s Principle. Applying this allows most buffer problems to be solved!