pH Curves
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
pH curves show how pH changes during an acid-base titration.
Keywords
Equivalence point: Volume at which moles of acid = moles of base.
End point: The point during the titration when the indicator changes colour.
General pH curve
x-axis (horizontal): Volume of base (or acid) added, usually in .
y-axis (vertical): pH of the solution, measured with a pH probe. When a base is added from a burette into a flask containing an acid, a pH curve can be divided into three main sections.
1. Gradual increase in pH as [] decreases.
2. Vertical sections due to sharp increase in pH when the acid is neutralised.
3. Gradual increase in pH as [] increases.
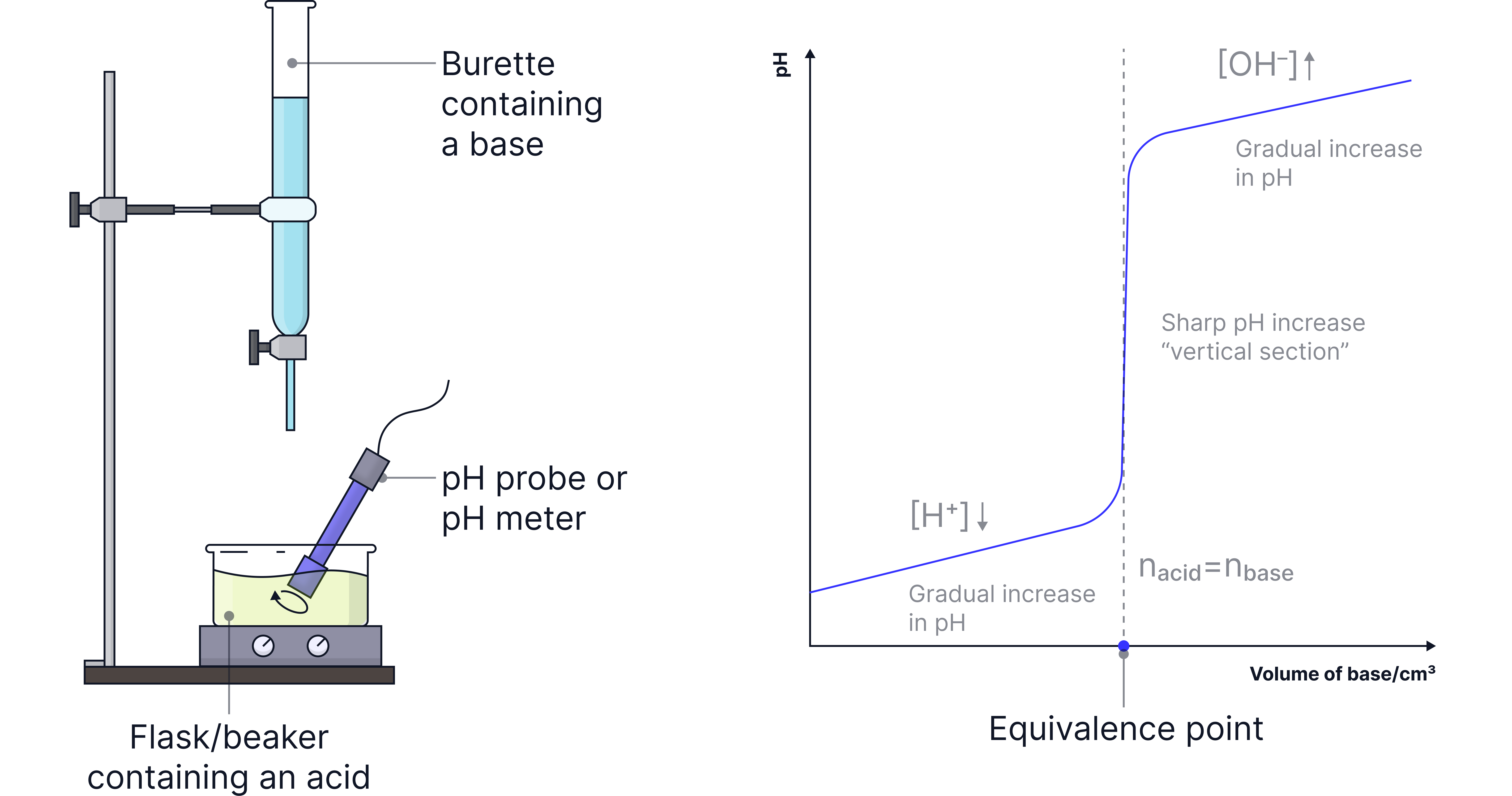
The volume at which the number of moles of acid are equal to the number of moles of base is the equivalence point.
This is the volume at the vertical section of the curve.
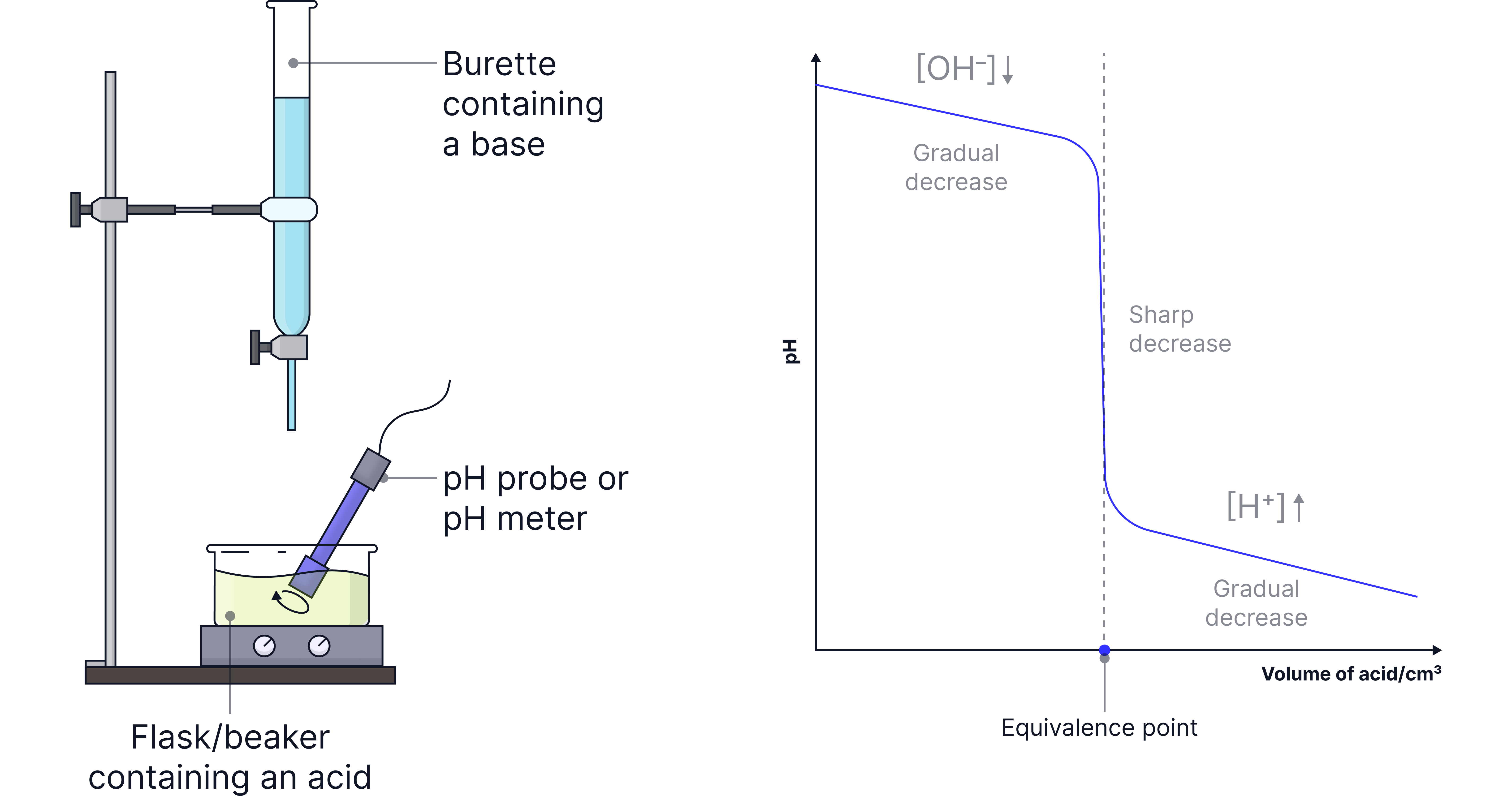
Less commonly assessed, the acid can be added from the burette to a base in a flask.
This results in the general shape below.
Acid-base combinations
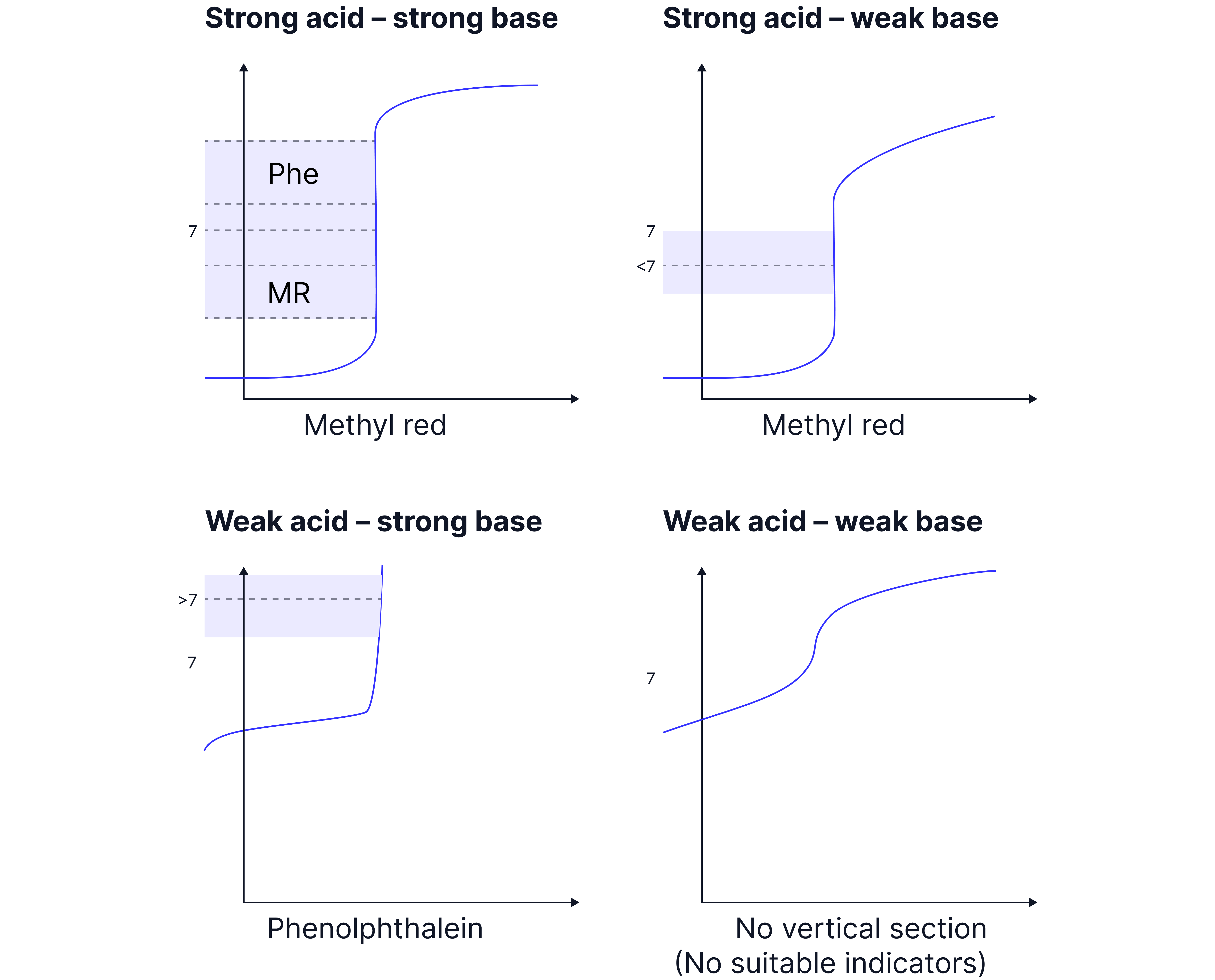
There are 4 types of pH curves to recognise and interpret based on the combination of acid and base used.
Strong acid – strong base e.g. and
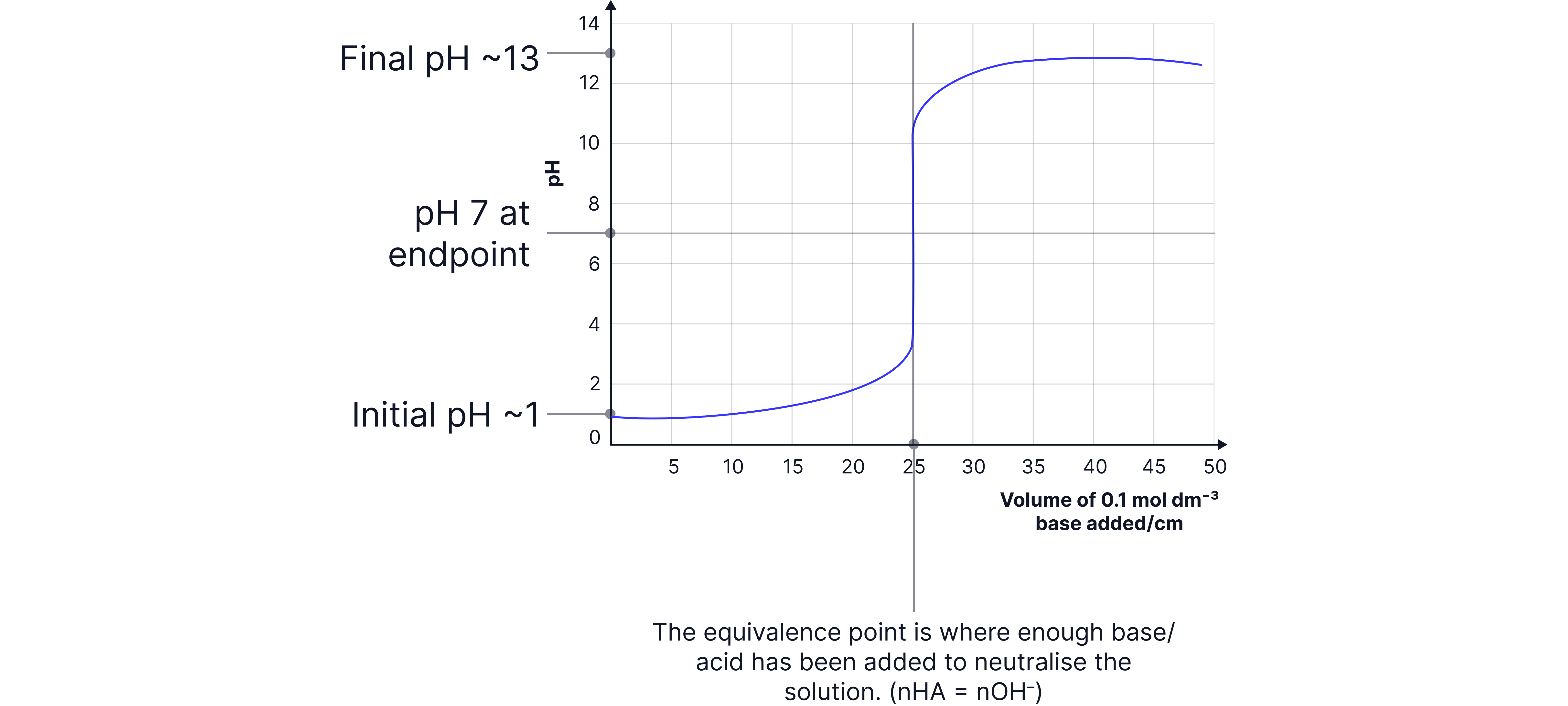
Strong acid – weak base e.g. and
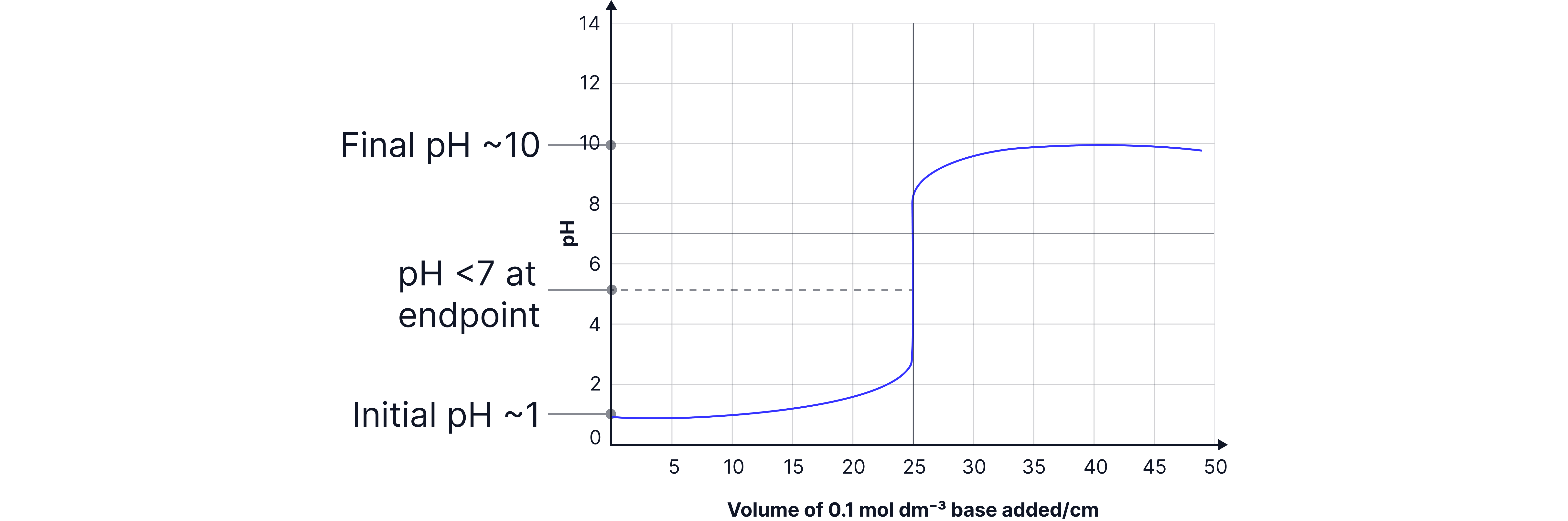
Weak acid – strong base e.g. and
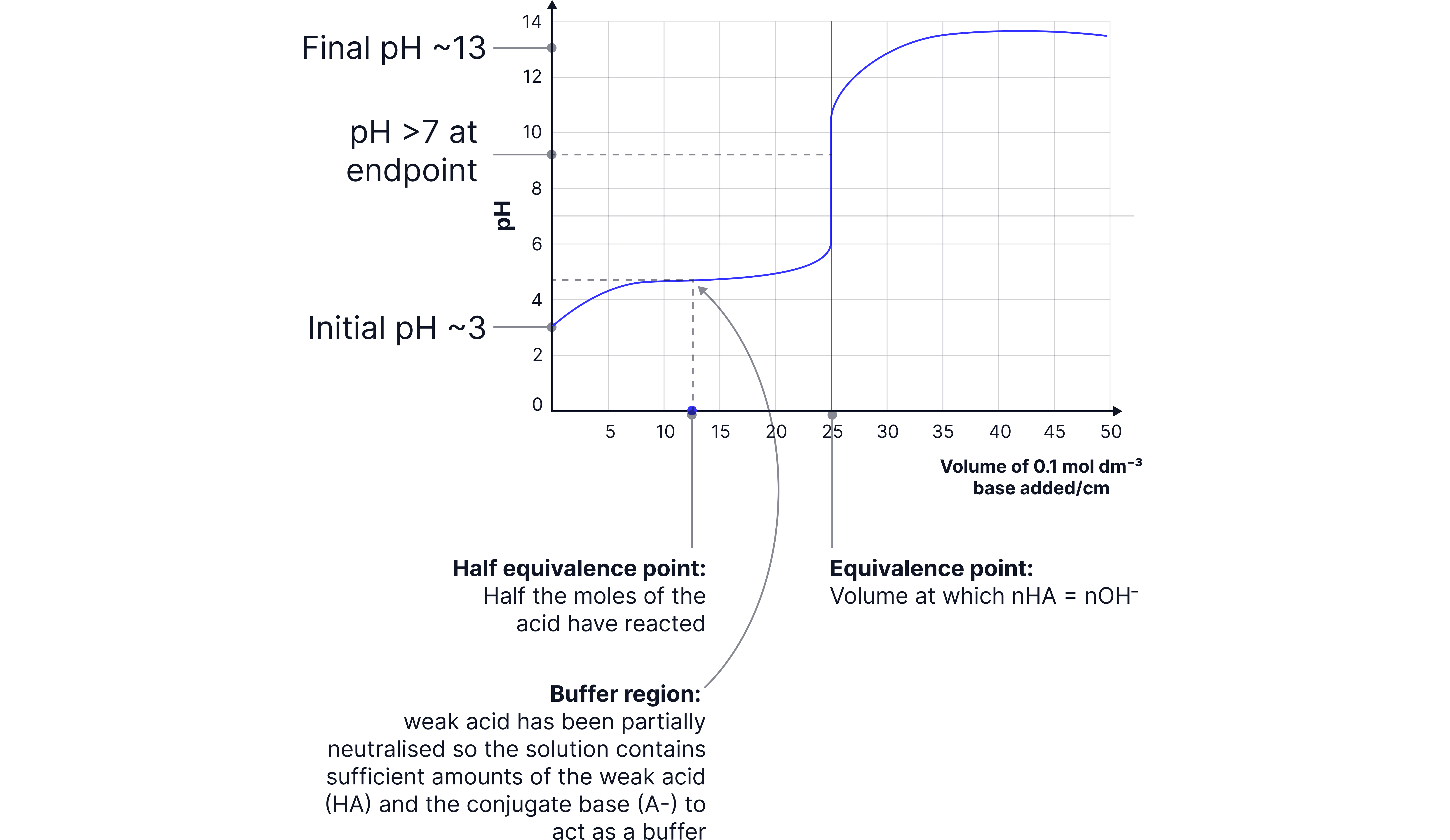
Half Equivalence Point in a Weak Acid–Strong Base Titration
The half equivalence point is the stage in the titration when half of the weak acid has been neutralised by the strong base.
At this point:
Moles of acid remaining = moles of conjugate base formed.
Why it matters:
The solution contains a buffer mixture of equal concentrations of weak acid and its conjugate base.
Using the expression:
At half equivalence, so:
How it is used:
By measuring the at the half equivalence point, you can determine the of the weak acid directly.
This provides experimental confirmation of the acid strength.
Example:
Titrating of ethanoic acid with . Equivalence (vertical section) at of NaOH added.
Therefore, half equivalence at NaOH added.
If measured at this point is found to be ,
then of ethanoic acid = .
Weak acid – weak base e.g. and
Titration Curves Practice Question Video
Choosing an Indicator
An indicator should change colour at the equivalence point (where the moles of acid and base are equal).
The end point in a titration is the point where a visible change shows that the reaction is complete.
In acid-base titrations, indicators are added and change colour at the end point.
Indicators are weak acids or bases where at least one species in the equilibrium has a colour:
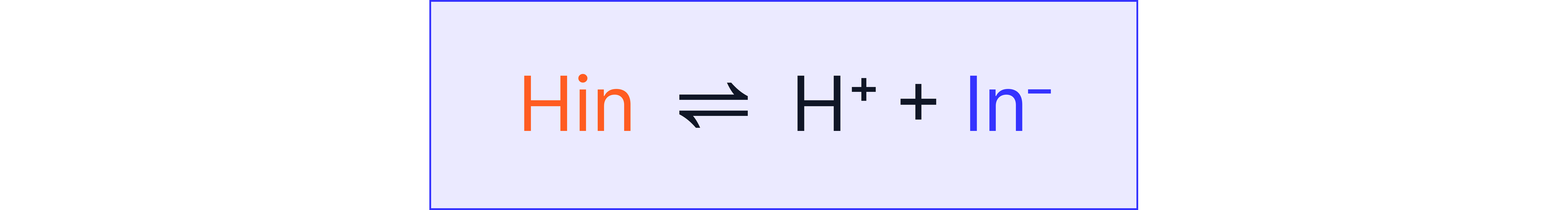
Adding acid or base shifts the position of the equilibrium changing the appearance of the solution.
Different indicators change colour at different .
An indicator is appropriate for a given acid-base combination if the at which is changes colour falls within the vertical section of a curve.
Indicator | Acidic Colour | Alkaline Colour | pH range |
Methyl Orange | Red | Yellow | 3.1-4.4 |
Methyl Red | Red | Yellow | 4.4-6.2 |
Phenolphthalein | Colourless | Pink | 8.3-10.0 |
Practice Question
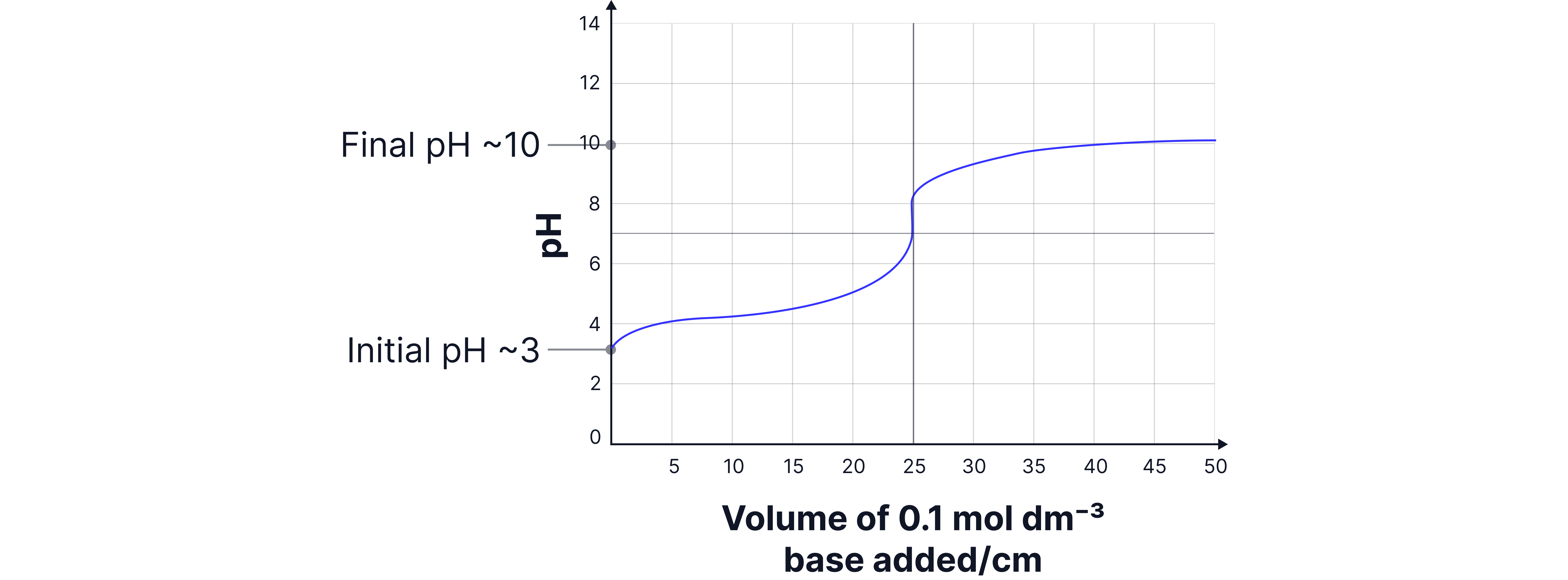
The sketch below shows the change in when a solution of sodium hydroxide is added from a burette to of a solution of the weak acid at .
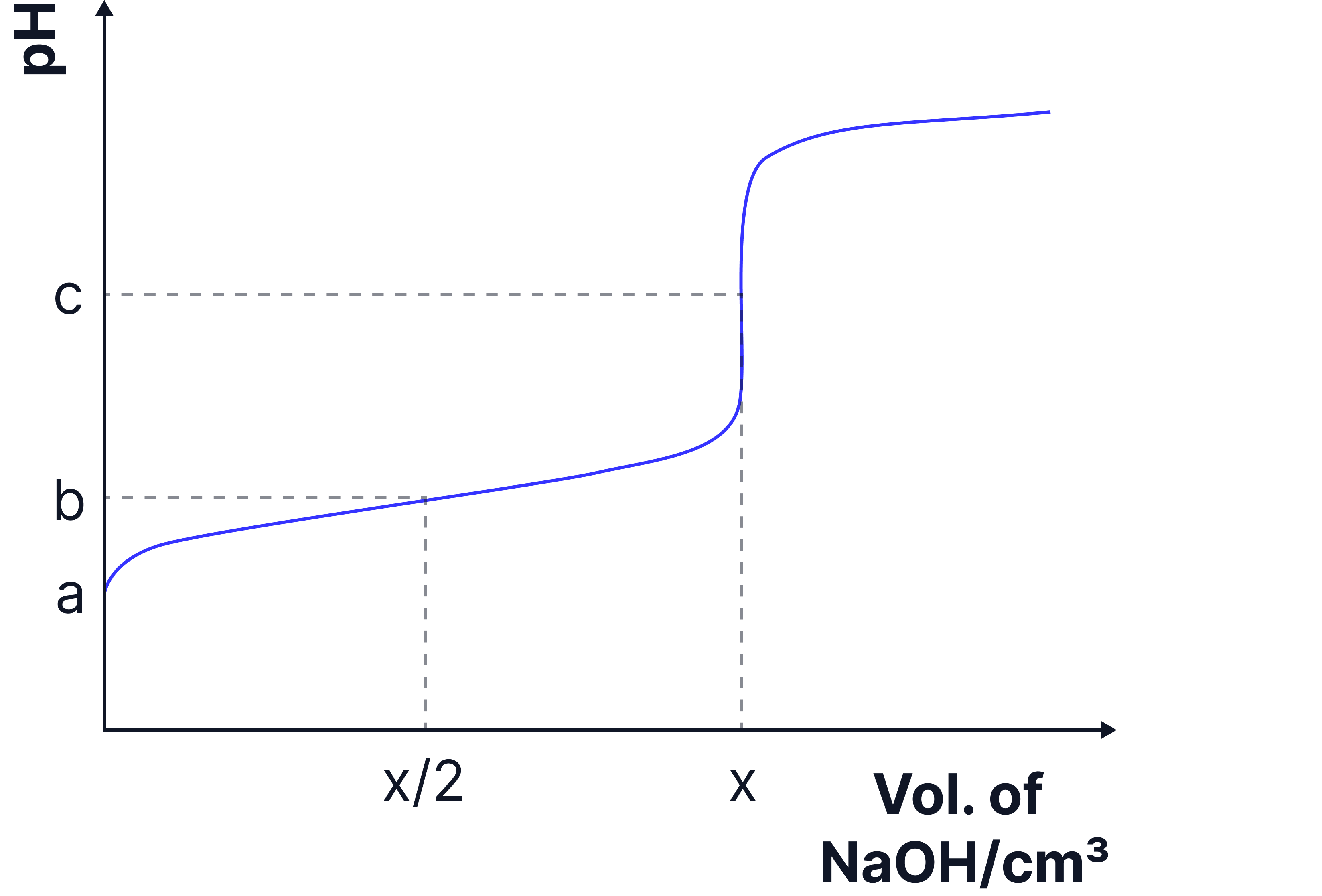
a) Calculate the value of in .
b) Deduce algebraically for the weak acid .
Answer:
a) Calculate the value of in .
equivalence point volume at which moles of acid and base are equal.
b) Deduce algebraically for the weak acid .
at half equivalence point b