Calorimetry
Lajoy Tucker
Teacher

Contents
Introduction to Calorimetry
Calorimetry is the measurement of heat energy transferred in a chemical or physical process. It allows us to determine the enthalpy change of reactions by measuring a temperature change and applying:
q = m c ΔT
Understanding the Equation
q = m c ΔT
q = heat energy transferred (J)
m = mass of the substance heated (g)
c = specific heat capacity (J g⁻¹ K⁻¹), for water = 4.18 J g⁻¹ K⁻¹
ΔT = temperature change (°C or K) = final temperature – initial temperature
Important:
For aqueous solutions, m = total mass of solution (1 cm³ ≈ 1 g).
The sign of ΔH is added at the end:
Negative (–) for exothermic
Positive (+) for endothermic
This tells us the total energy transferred for the actual amount of substance burned or reacted in the experiment. Now we have to converting to molar enthalpy. Enthalpy change (ΔH) is defined per mole of reactant. To get this, divide q by the number of moles, n, of the substance that reacted:
ΔH = q / n
If the reaction is exothermic, ΔH is negative.
If endothermic, ΔH is positive.
If confident combine the equations to go straight from experimental measurements to molar enthalpy:
ΔH = (m c ΔT) / n
Polystyrene Cup Calorimetry (Solution Reactions)
Apparatus & Setup
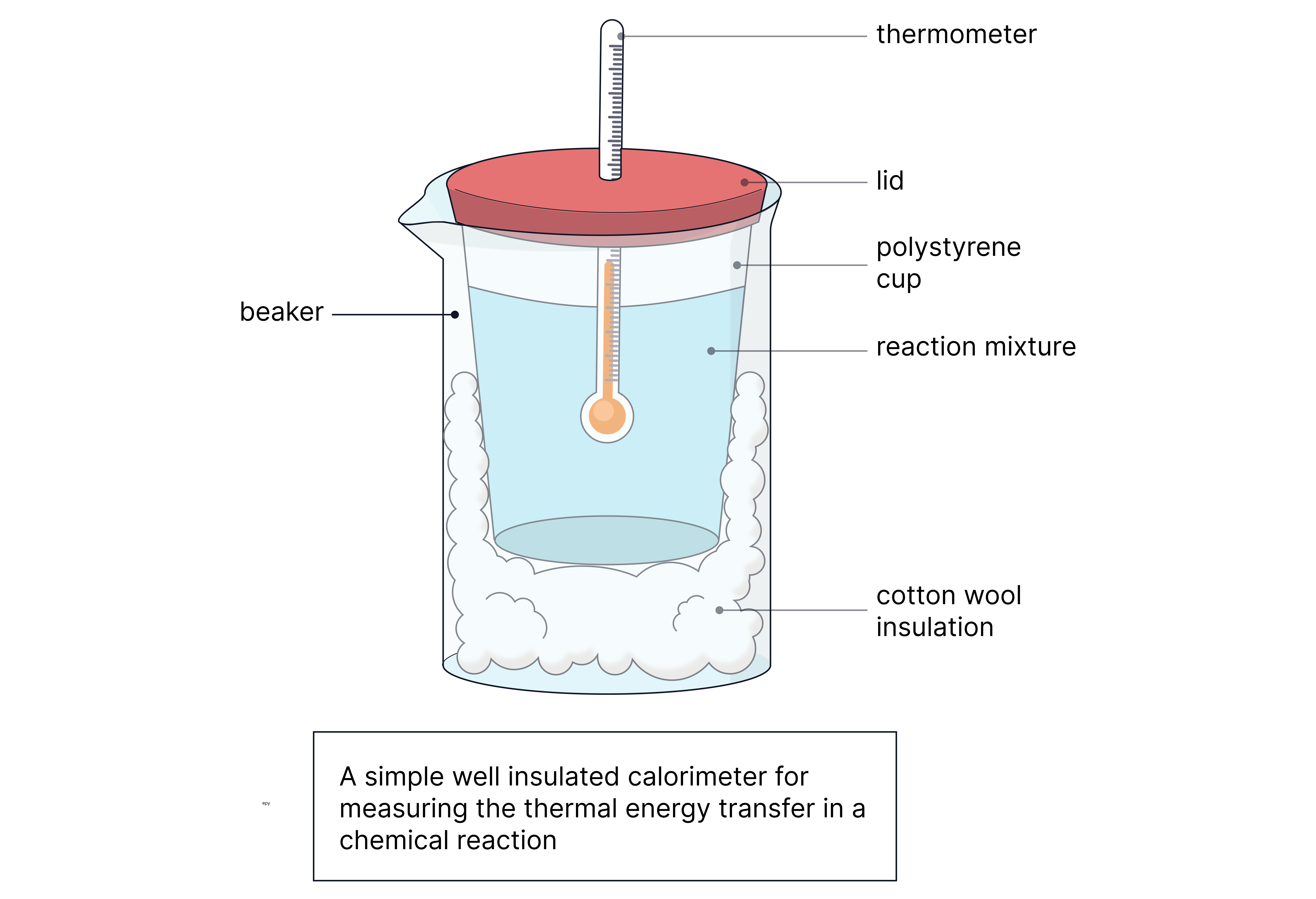
Polystyrene cup with lid to reduce heat loss
Thermometer or temperature probe
Reactants measured with measuring cylinders/pipettes
Stirrer for even mixing
Why Polystyrene?
Polystyrene is a poor conductor (good insulator) → so reduces heat exchange with surroundings.
Procedure
Measure volume(s) of reactant(s) accurately.
Place one reactant into the polystyrene cup and record the initial temperature.
Add the second reactant quickly, stir immediately.
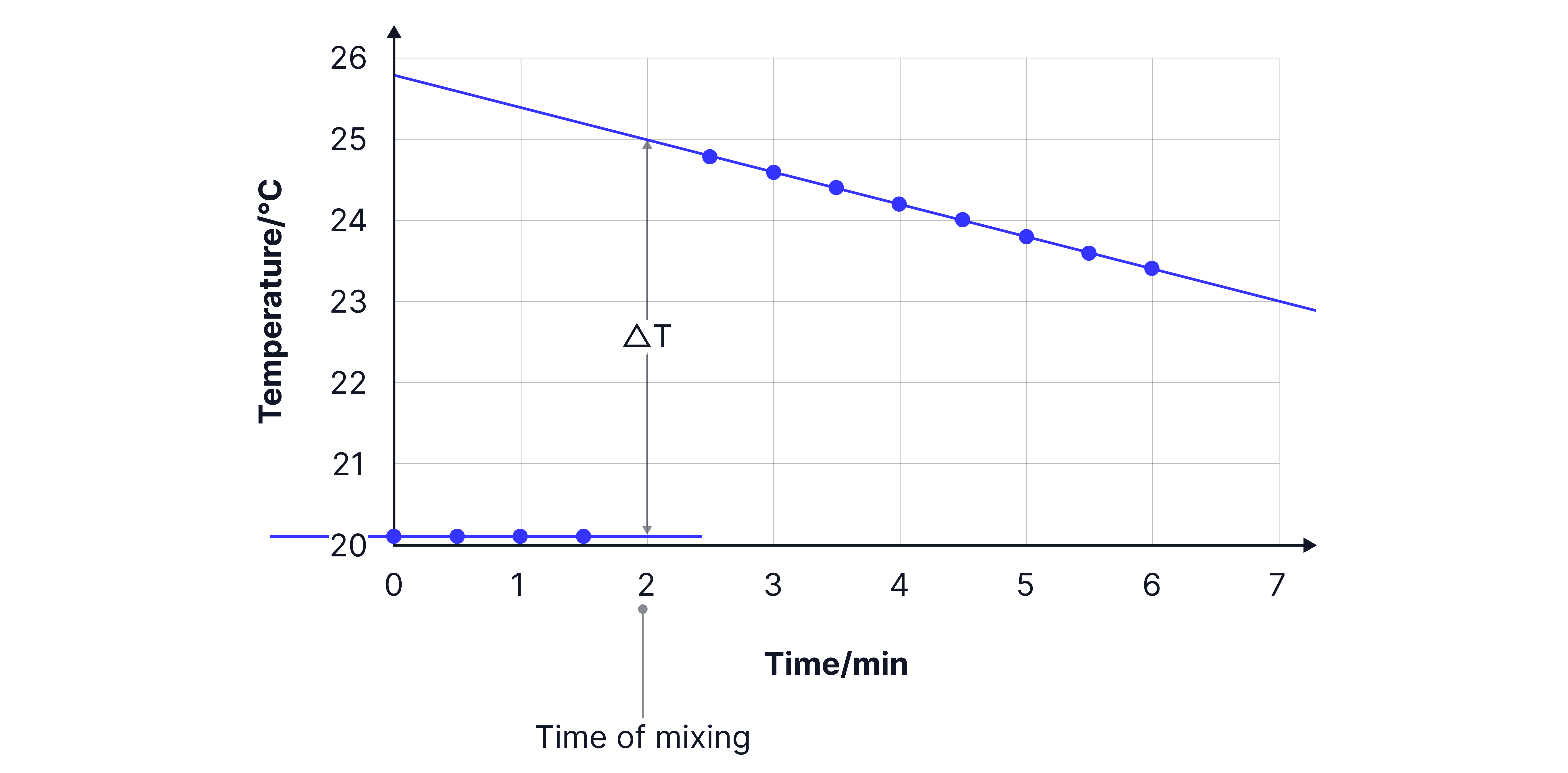
Record temperature every 30 seconds for 5 minutes.
Use a cooling curve to estimate the temperature at the exact moment of mixing.
Common Errors & Improvements
Heat loss – use a lid and insulating jacket around the cup.
Inaccurate mixing – stir continuously.
Evaporation – minimise by covering the reaction.
Measurement error – use a digital temperature probe for higher accuracy.
Cooling Curve – Why & How
When a reaction occurs, heat starts to escape to the surroundings almost immediately. By plotting a cooling curve and extrapolating back, we can estimate what the temperature would have been if no heat loss had occurred.
Easy – Neutralisation (exothermic)
Question:
25.0 cm3 of 1.00 mol dm-3 HCl at 20.0 °C is mixed with 25.0 cm3 of 1.00 mol dm-3 NaOH at 20.0 °C in a polystyrene cup. The maximum temperature recorded is 26.8 °C.
Assume density = 1.00 g cm-3 and c(solution) = 4.18 J g-1 K-1. Find ΔH (kJ mol-1) for neutralisation.
Worked solution:
Balanced chemical equation: HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
Mass, m = (25.0 + 25.0) g = 50.0 g
ΔT = 26.8 − 20.0 = 6.8 K
q = m × c × ΔT = 50.0 × 4.18 × 6.8 = 1422 J = 1.422 kJ (released)
Moles of water formed = moles of HCl = (25.0 / 1000) × 1.00 = 0.0250 mol
ΔH (per mole H2O) = −q / n = −1.422 / 0.0250 = −56.9 kJ mol-1
Medium – Dissolution of potassium chloride (endothermic)
Question:
6.00 g of KCl is dissolved in 100.0 cm3 of water at 21.0 °C in a polystyrene cup. The final steady temperature is 18.7 °C.
Assume density = 1.00 g cm-3 and c(solution) = 4.18 J g-1 K-1. Calculate the enthalpy change of solution per mole of KCl.
Worked solution:
Dissolution equation: KCl(s) → K+(aq) + Cl−(aq)
Mass of solution ≈ 100.0 g (A-Level assumption)
ΔT = 18.7 − 21.0 = −2.3 K
|q| = m × c × |ΔT| = 100.0 × 4.18 × 2.3 = 961 J
Sign: temperature falls → process absorbs heat → q = +0.961 kJ
Moles KCl = 6.00 / 74.55 = 0.0805 mol
ΔHsoln = +0.961 / 0.0805 = +11.9 kJ mol-1
Hard – Maximum temperature reached
Question:
50.0 cm³ of 1.00 mol dm⁻³ hydrochloric acid at 20.0 °C is placed in a polystyrene cup. A 0.60 g strip of magnesium metal at 20.0 °C is added and the mixture is stirred. Assume all the magnesium reacts and all heat released warms the solution and dissolved salts only. Take density = 1.00 g cm⁻³, c(solution) = 4.18 J g⁻¹ K⁻¹, and assume the enthalpy change for
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
is ΔH = −460 kJ mol⁻¹ (per mole of Mg). Calculate the maximum temperature reached.
Worked solution:
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
1. Amounts
n(HCl) = (50.0 cm³ × 1.00 mol dm⁻³) ÷ 1000 = 0.0500 mol
n(Mg) = 0.60 g ÷ 24.3 g mol⁻¹ = 0.0247 mol
Required HCl for this Mg = 2 × 0.0247 = 0.0494 mol
HCl available = 0.0500 mol → acid in slight excess → Mg is limiting.
2. Heat released (from ΔH per mole of Mg)
q(released) = n(Mg) × 460 kJ mol⁻¹
= 0.0247 × 460 = 11.36 kJ = 11 360 J
3. Temperature rise (ignore Mg mass; heat the solution only)
m(solution) = 50.0 g
q = m c ΔT ⇒ ΔT = q ÷ (m c)
ΔT = 11 360 ÷ (50.0 × 4.18) = 11 360 ÷ 209 ≈ 54.4 K or 54.44 °C
4. Maximum temperature
Tₘₐₓ = Tᵢₙᵢₜᵢₐₗ + ΔT = 20.0 °C + 54.4 °C ≈ 74.4 °C
Combustion Calorimetry
Definition & Purpose
This method measures the enthalpy change of combustion (ΔHc) by burning a fuel and using the released heat to warm a known mass of water.
Apparatus & Setup
Glass calorimeter filled with known volume of water
Could use a copper calorimeter (good conductor) filled with measured water. Copper is a better conductor of heat than glass so better heat transfer to the water.
Spirit burner with known mass of fuel
Draught shield to reduce heat loss
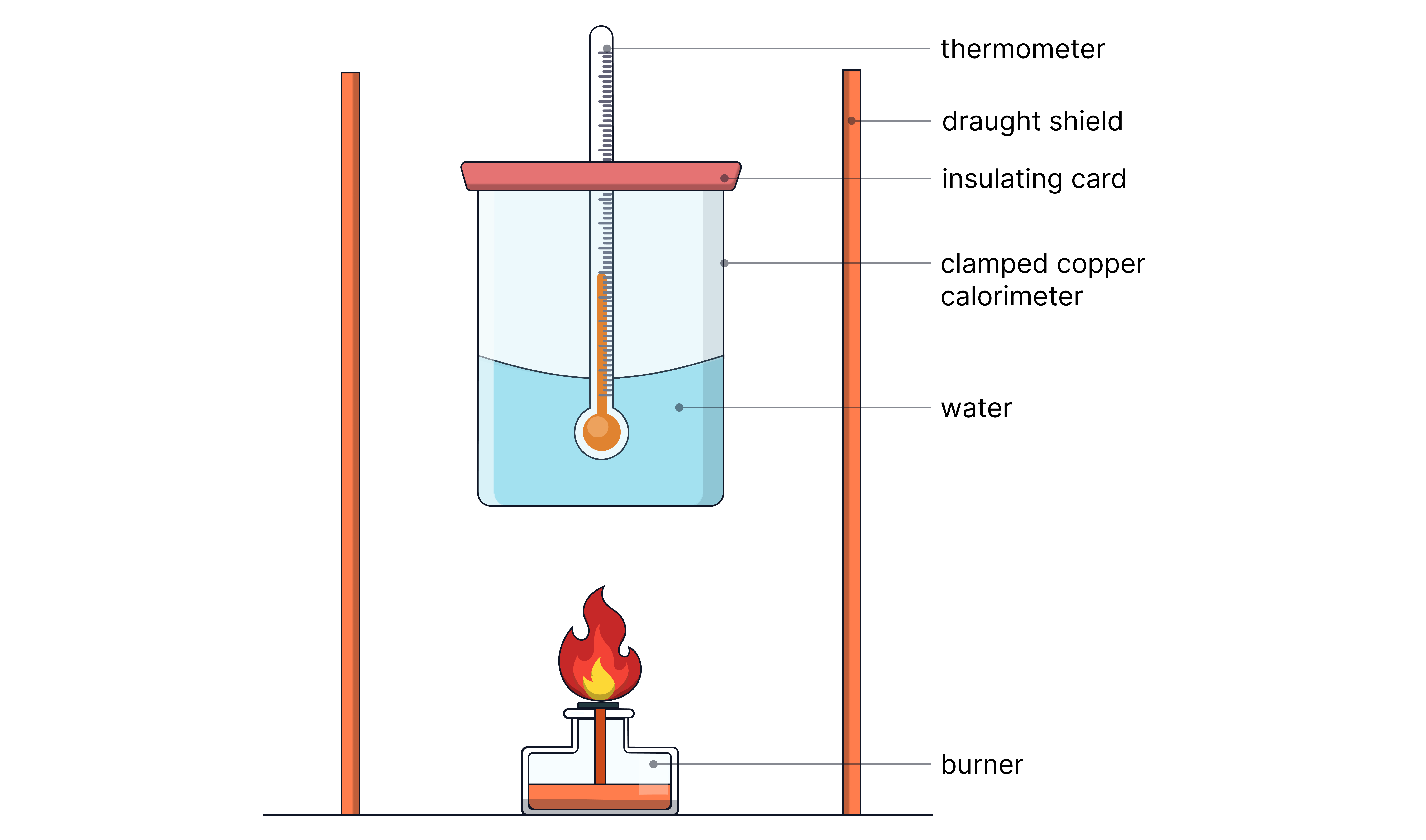
Procedure
1. Measure mass of fuel + burner before starting.
2. Measure known volume of water into calorimeter.
3. Record initial temperature of water.
4. Light burner and stir water continuously.
5. Extinguish flame after temperature rise of ~10–20°C.
6. Record final temperature and re-weigh burner.
Sources of Error
Heat lost to surroundings and calorimeter body.
Incomplete combustion of fuel.
Evaporation of fuel during setup.
Soot formation (due to incomplete combustion) → reduces heat transfer.
Improvements:
Use lid on calorimeter.
Enclose flame and calorimeter in draught shield.
Use larger temperature rise to reduce % error.
EASY – Combustion of Methanol
Question:
When 1.50 g of methanol (CH₃OH) is burned in a spirit burner, the heat released raises the temperature of 200 g of water from 20.0 °C to 36.5 °C.
c(water) = 4.18 J g⁻¹ K⁻¹.
Calculate:
a) The heat released (q)
b) The molar enthalpy of combustion of methanol in kJ mol⁻¹.
M(CH₃OH) = 32.0 g mol⁻¹.
Worked solution:
a) ΔT = 36.5 − 20.0 = 16.5 °C
q = m c ΔT = (200)(4.18)(16.5) = 13 794 J = 13.79 kJ
b) n(CH₃OH) = 1.50 ÷ 32.0 = 0.04688 mol
ΔH = q ÷ n = 13.79 ÷ 0.04688 = 294 kJ mol⁻¹
Exothermic, so ΔH = −294 kJ mol⁻¹.
MEDIUM – Combustion of Propan-1-ol
Question:
2.30 g of propan-1-ol (C₃H₇OH) is burned in excess oxygen, heating 250 g of water from 18.0 °C to 43.4 °C.
c(water) = 4.18 J g⁻¹ K⁻¹, M(C₃H₇OH) = 60.0 g mol⁻¹.
Calculate the molar enthalpy of combustion in kJ mol⁻¹.
Worked solution:
ΔT = 43.4 − 18.0 = 25.4 °C
q = (250)(4.18)(25.4) = 26 543 J = 26.54 kJ
n(C₃H₇OH) = 2.30 ÷ 60.0 = 0.03833 mol
ΔH = 26.54 ÷ 0.03833 = 693 kJ mol⁻¹
Exothermic, so ΔH = −693 kJ mol⁻¹.