Applying Hess’s Law
Dr. Davinder Bhachu
Teacher

Contents
Hess’s Law - An Introduction
Hess’s states that the total enthalpy change for a chemical reaction is independent of the route taken, meaning the overall enthalpy change is the same whether the reaction occurs in one step or multiple steps.
It is an application of the law of conservation of energy and Hess’s Law cycles allow for the calculation of enthalpy changes for reactions that are difficult to measure directly.
Key Definitions
Enthalpy change of formation – enthalpy change when 1 mole of the compound is produced from its elements in their standard states, under standard conditions ().
Enthalpy change of combustion - heat energy released when 1 mole of a substance is burned completely in oxygen under standard conditions (), with all reactants and products in their standard physical states.
Mean bond enthalpy - enthalpy required to completely separate a covalent bond, producing individual gaseous atoms from gaseous molecules. This value is calculated as the mean taken from measurements across a variety of different molecules containing that type of bond.
Hess’s Law from first principles
Enthalpy – Climbing the Energy Mountain
Picture a vast mountainous landscape. Scattered along the slopes are three base camps:
Camp Ice – low in the valley
Camp Water – halfway up the mountain
Camp Vapour – near the summit
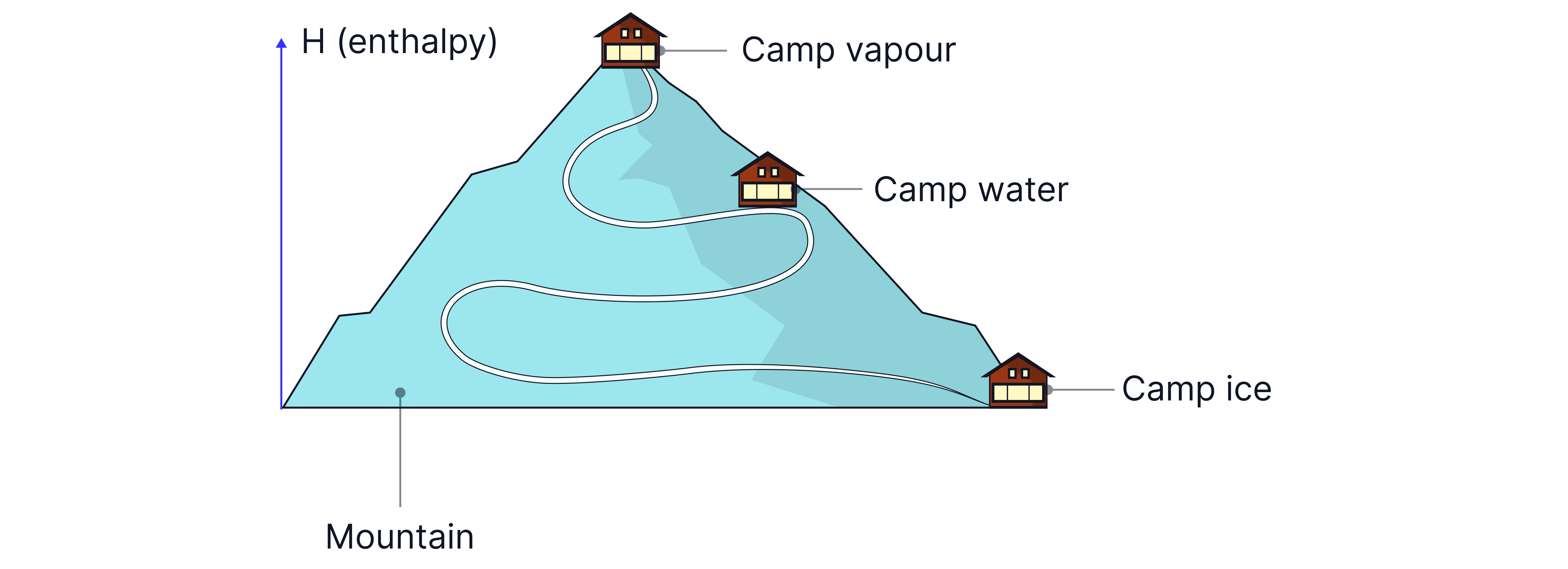
Each base camp represents a physical state of water with a different amount of stored chemical energy, called enthalpy (H). The higher the camp, the more energy is stored.
The Journey
You're a climber starting at Camp Ice, and your goal is to reach Camp Water. You can either:
Take the direct path, or
Go the scenic route (indirect path) via the summit (vapour)
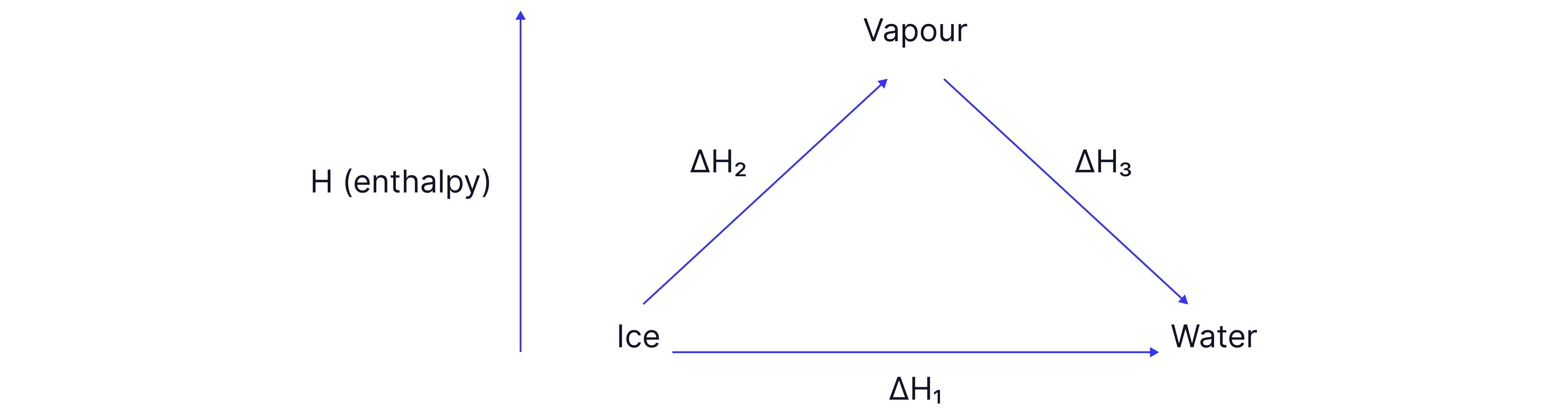
In chemistry, the enthalpy change for any transformation is simply:
It tells us how much energy is gained or lost as you move between states – just like the change in altitude on a hike up the mountain.
Route 1: Direct Path
This equation tells us how much energy is gained or lost as you move between to
Route 2: Scenic Route (Indirect path)
Add them together:
Substitute:
Simplify:
Conclusion – Hess’s Law
The overall enthalpy change from ice to water is the same whether you go directly or indirectly (via vapour):
This proves Hess’s Law – the enthalpy change depends only on the initial and final states, not on the route taken.
Applying Hess’s Law
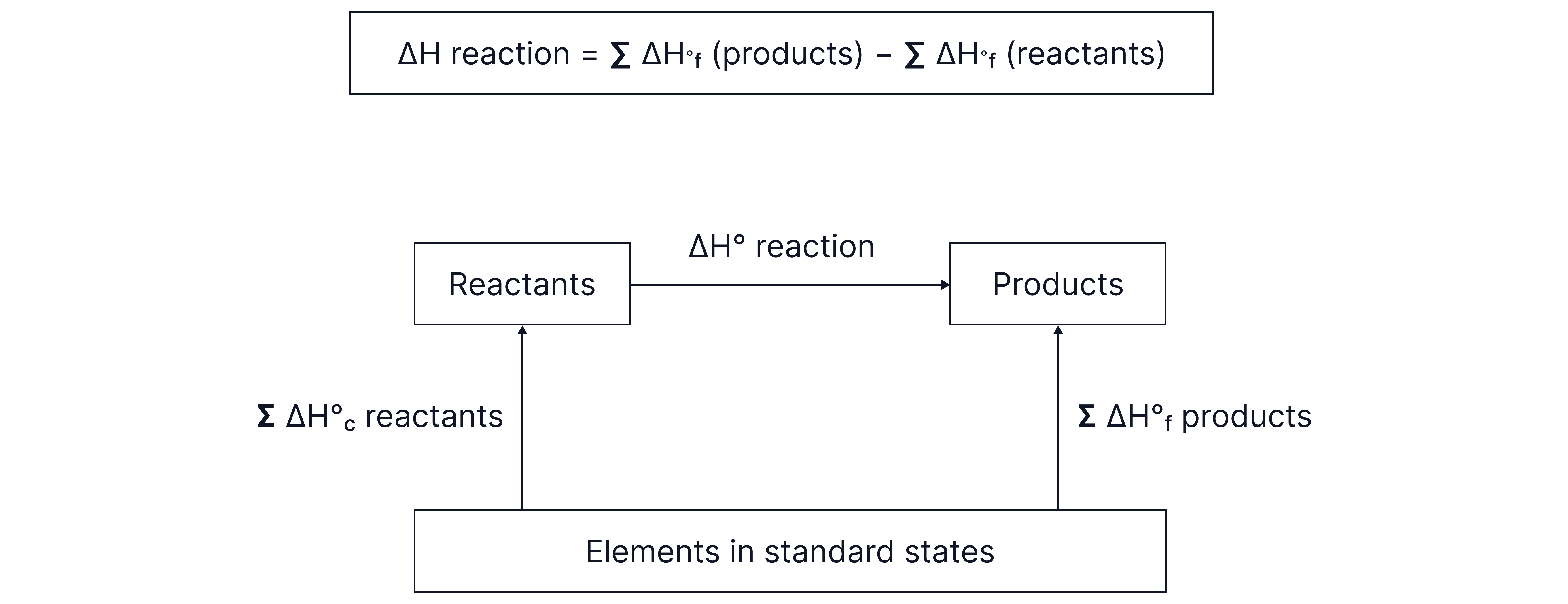
Forming Hess cycles to determine enthalpy changes from enthalpy of formation data:
Using Hess's law to determine enthalphy changes from enthalphy changes of formation.
Worked Examples
EASY- Example 1: Calculating enthalpies from formation data
Question:
Calculate the enthalpy change, , for the decomposition of calcium carbonate, , shown below:
Data ():
Answer
1. Draw the annotated Hess’s Law Cycle

2. Use Hess’s Law to calculate
Substitution and Result:
MEDIUM - Example 2: Calculating enthalpies from formation data (considering stoichiometric ratios)
Calculate the enthalpy change, , for the reduction of iron (III) oxide, , with carbon monoxide, , shown below:
Data ():
– notice how this is zero because it is already the element in its standard start, therefore there is no enthalpy change associated
Answer
1. Draw the annotated Hess’s Law Cycle
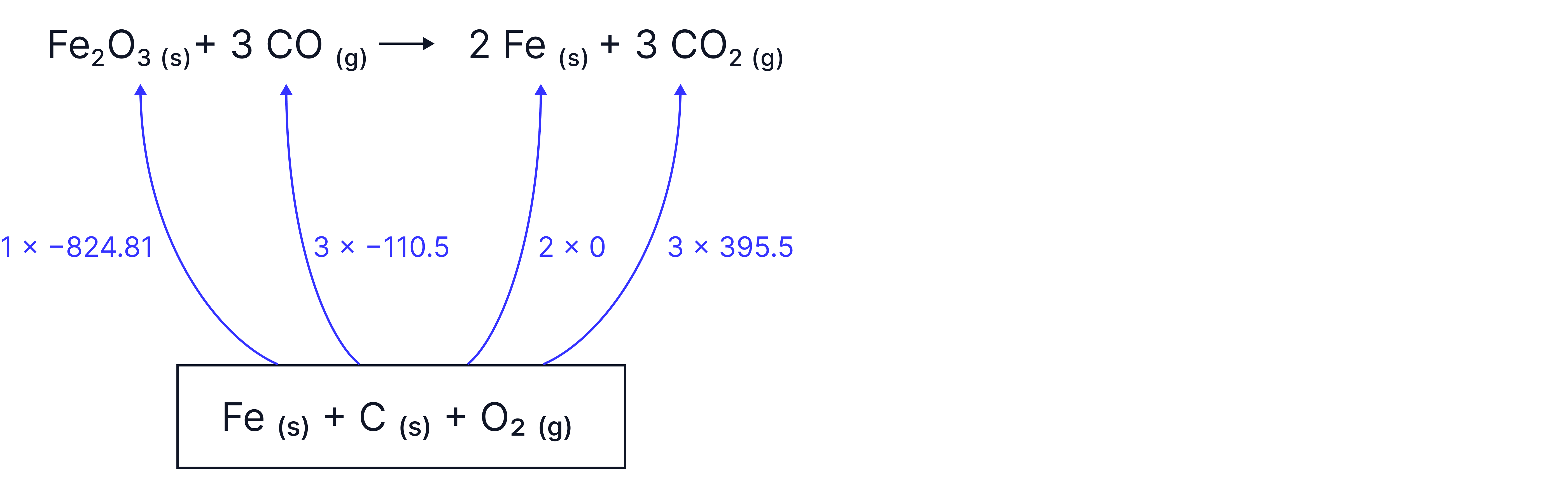
2. Use Hess’s Law to calculate
Substitution and Result:
Forming Hess cycles to determine enthalpy changes from enthalpy of combustion data:
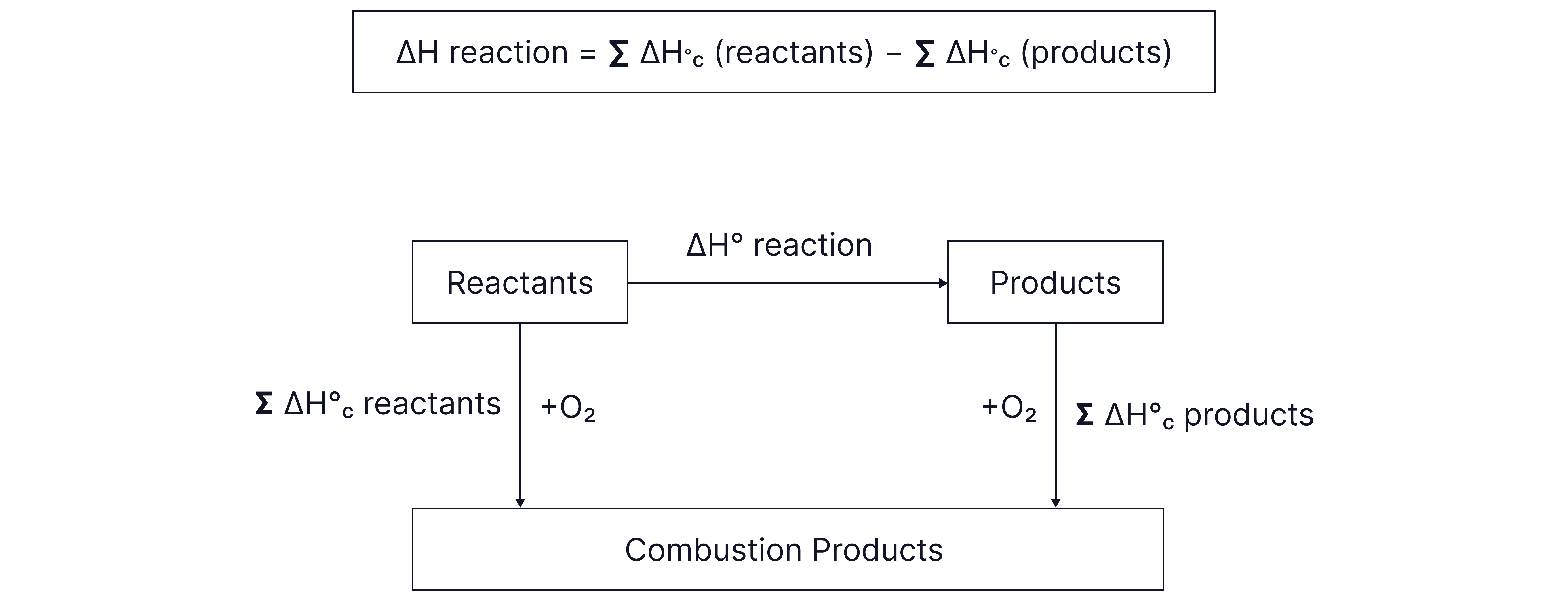
*notice how in the equation, (reactants) appears first and is positive overall, whereas (products) is negative overall. This is because of the definition of combustion, which focuses on the production of combustion products and hence, the arrows pointing the other way rouns compared to the formation examples.
MEDIUM - Example 3: Calculating enthalpies from combustion data (considering stoichiometric ratios)
Calculate the standard enthalpy of formation of ethyne, , as shown below:
Given standard enthalpies of combustion, :
*notice how in this question, the reaction shows ethyne, , being made from its elements in the standard state. As such, is the same as for this question.
Answer
1. Draw the annotated Hess’s Law Cycle

2. Use Hess’s Law to calculate from combustion data
Substitution and Result:
HARD - Example 4: Calculating missing from combustion data and given (considering stoichiometric ratios and rearranging to find missing values)
Question:
The hydrogenation of ethene to form propane is represented by:
The standard enthalpies of combustion for some species present are:
Using Hess’s Law, calculate the standard enthalpy of combustion of hydrogen gas, .
Answer
1. Draw the annotated Hess’s Law Cycle

*notice how the is now labelled and is represented as X.
2. Use Hess’s Law to calculate missing from combustion data and given
Substitution, Simplification (solve for x) and Result:
*notice that all the enthalpies for combustion, , are all negative, this is because combustion reactions are ALWAYS exothermic
Forming Hess cycles to determine enthalpy changes from enthalpy of bond enthalpy data:
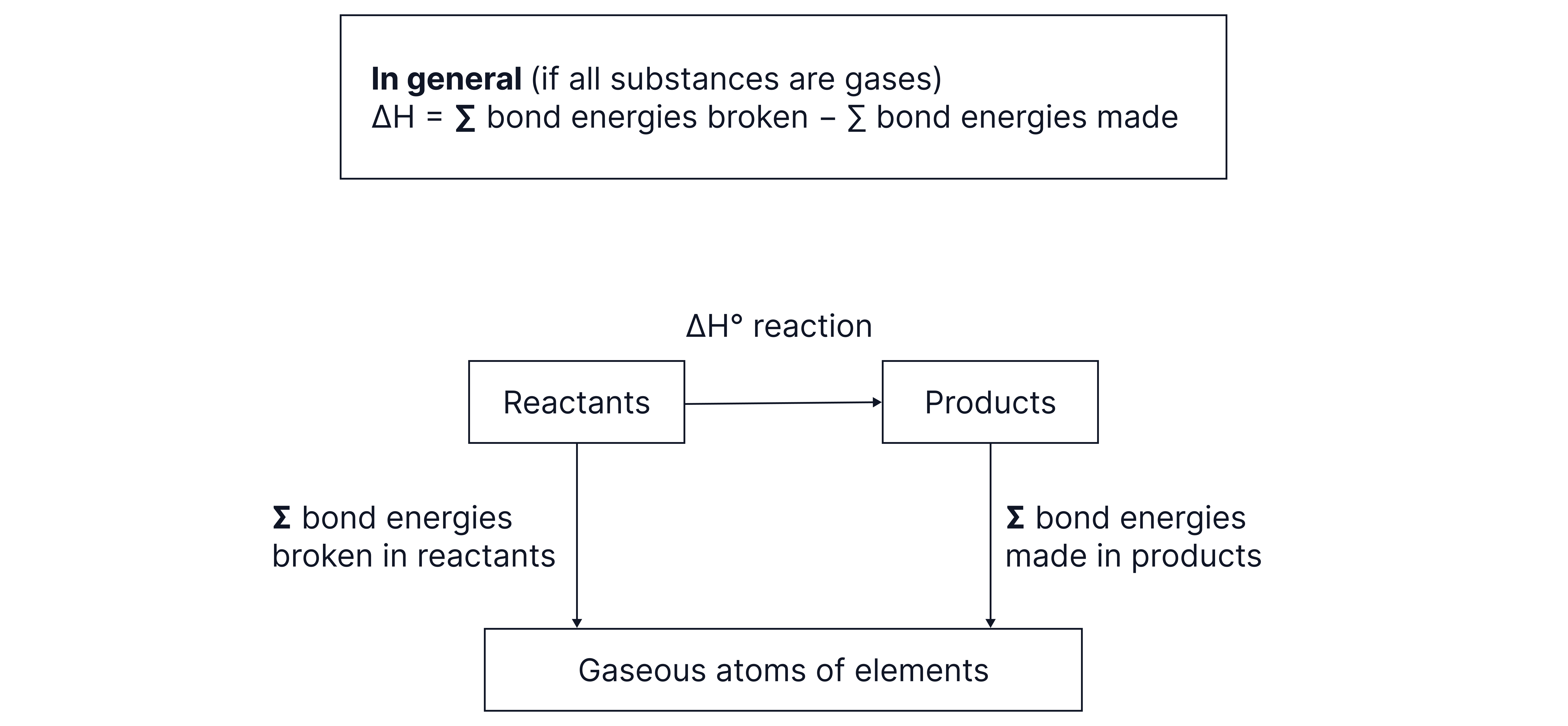
We use average (mean) bond energy values because no two bonds are ever exactly identical in their energy requirements. Even within the same molecule, each bond can differ slightly in strength depending on its environment. For instance, in a water molecule () there are two bonds, but breaking the first bond requires a slightly different amount of energy compared to breaking the second one.
Despite these variations, chemists use a single averaged bond energy value when performing calculations, applying it across all molecules containing that type of bond making these calculations less accurate than the other applications of Hess’s Law. These values will be given to you in the questions or in your data booklet.
Forming bonds, which releases energy, will always be exothermic and breaking bonds, which requires energy, will always be endothermic. Be aware of the fact that bond enthalpies are always given as positive values, the equation accounts for this, as the sum of the bond energies for bonds made has minus sign in front of it.
MEDIUM - Example 5: Calculating ΔH°r from mean bond enthalpy data
Question:
Calculate the enthalpy of combustion of ethene using the following mean bond enthalpy data below:
Mean bond enthalpies ():
Reaction:
Answer
1. Draw the displayed formula for the molecules in the reaction and count the all bonds on reactant side and product side
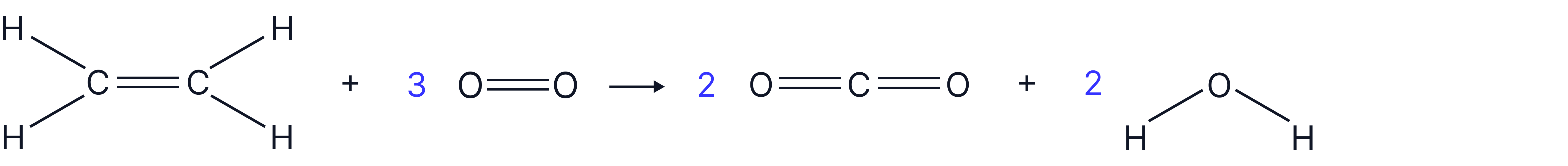
*pay close attention to the stoichiometry after counting all the bonds carefully (useful to cross them out as you count)
Reactants:
Products:
2. Use the formula, derived from Hess’ Law, to calculate
Substitution and Result:
HARD - Example 6: Calculating a missing mean bond enthalpy when given
and other bond enthalpy values
Question:
Calculate the mean bond enthalpy of the N–H bond using the enthalpy of combustion of ammonia.
Reaction:
Mean bond enthalpies ():
Answer
1. Draw the displayed formula for the molecules in the reaction and count the all bonds on reactant side and product side
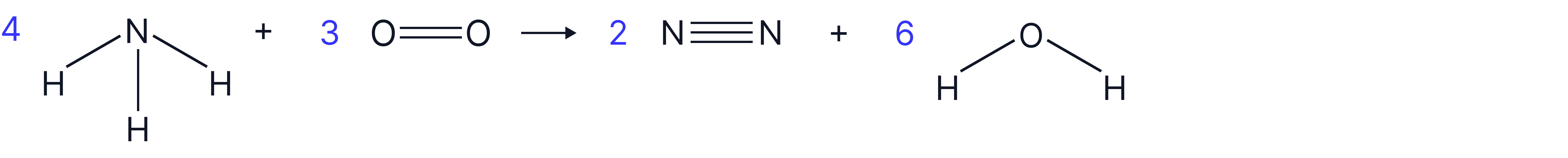
Reactants:
Products:
*as we are looking to calculate the N-H bond enthalpy, we will represent it as x
2. Use the formula, derived from Hess’s Law, and given to find the missing bond enthalpy
We are given in this question. By definition, this only applies to burning 1 mole of ammonia, . As 4 moles of ammonia are burnt here, we must multiply this number by 4 to get for this reaction.
Σ(bonds broken in reactants)
Σ(bonds made in products)
Substitution, Simplification (solve for x) and Result:
Hydrated Salts
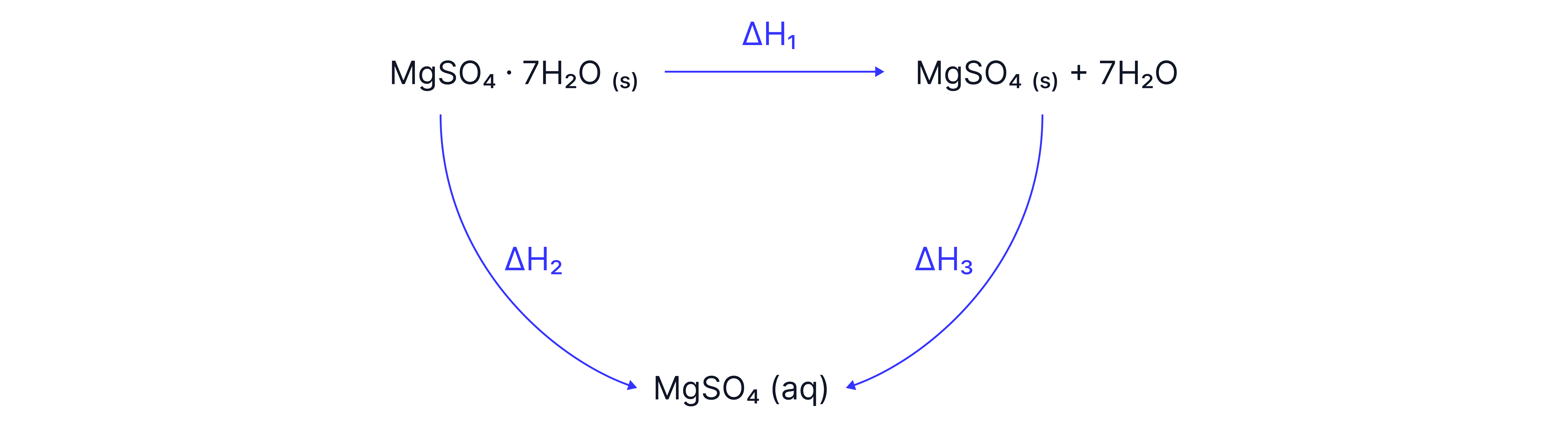
This is a Hess’s Law cycle involving magnesium sulphate heptahydrate , anhydrous magnesium sulphate , and aqueous magnesium sulphate . It’s tracking the enthalpy changes different routes to get from hydrated to aqueous .
The three enthalpy changes – More on this in Y13
: This is the enthalpy change of dehydration — the energy change when you remove 7 water molecules from hydrated to produce anhydrous (s) and 7 separate water molecules (often liquid, sometimes gas depending on the data).
Reaction:
: This is the enthalpy change of solution for hydrated magnesium sulphate — dissolving directly in water to make
: This is the enthalpy change of solution for anhydrous magnesium sulphate — dissolving into water to make
How this cycle works
Hess’s Law states that the total enthalpy change is the same no matter which route you take, provided the start and end conditions are the same. So:
1. You could dissolve hydrated directly () and measure the enthalpy change as per AQA Required Practical 2.
2. Or you could first dehydrate it (), then dissolve the anhydrous (), as per AQA Required Practical 2.
Because both routes end with , the enthalpy changes add up:
This is useful because you can rearrange to calculate enthalpy of dehydration indirectly. It is difficult to measure the enthalpy of dehydration directly because heating a hydrated salt often releases water gradually and at varying temperatures, making it hard to capture the full energy change. Some of the water may escape as steam or condense elsewhere, and the salt can partially decompose if overheated. Significant heat losses to the surroundings and the challenges of measuring a solid–gas process accurately mean that indirect methods using Hess’s Law are preferred.
EASY - Example 7: Calculating enthalpy of dehydration for a hydrated salt given its enthalpy of solution and that of the anhydrous form
Question:
Calculate the enthalpy of dehydration for magnesium sulphate heptahydrate, , given that the enthalpy of solution of magnesium sulphate heptahydrate, , and anhydrous magnesium sulphate, , are determined experimentally to be and
Worked solution:
1. Draw the annotated Hess’ Law Cycle
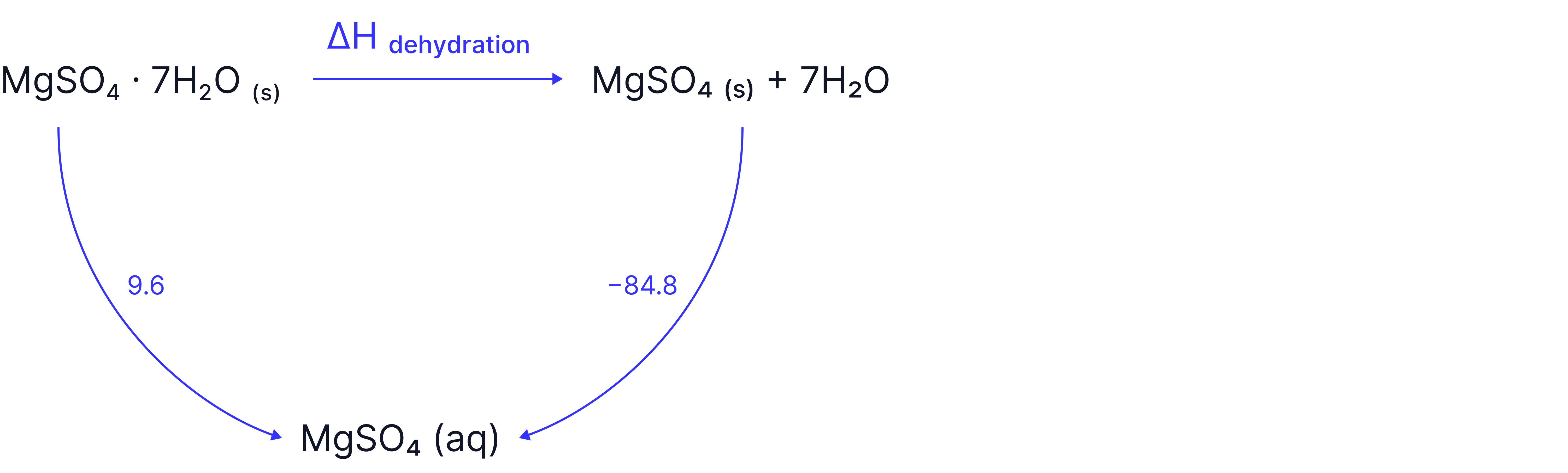
2. Use Hess’ Law to calculate missing
From our previous diagram:
Substitution and Result: