Enthalpy Changes
Lajoy Tucker
Teacher

Contents
Enthalpy – An Introduction
Enthalpy (H) is a measure of the total energy stored within a chemical system at constant pressure.
It includes:
The internal energy of the particles (their motion and interactions).
The energy needed to create space for the system by pushing against the surrounding atmosphere (pressure × volume).
In most chemistry experiments, we don’t measure H directly — instead, we focus on the change in enthalpy (ΔH) when a reaction takes place.
Why Enthalpy Is Useful
Most lab reactions happen at constant atmospheric pressure. Under these conditions, the heat transferred to or from the surroundings is equal to (ΔH).
This makes it practical to link heat changes measured in experiments (e.g. calorimetry) directly to enthalpy changes.
Enthalpy Profile Diagrams
These diagrams plot enthalpy (y-axis) against progress of reaction (x-axis) to show how energy changes as reactants turn into products.
Exothermic reaction:
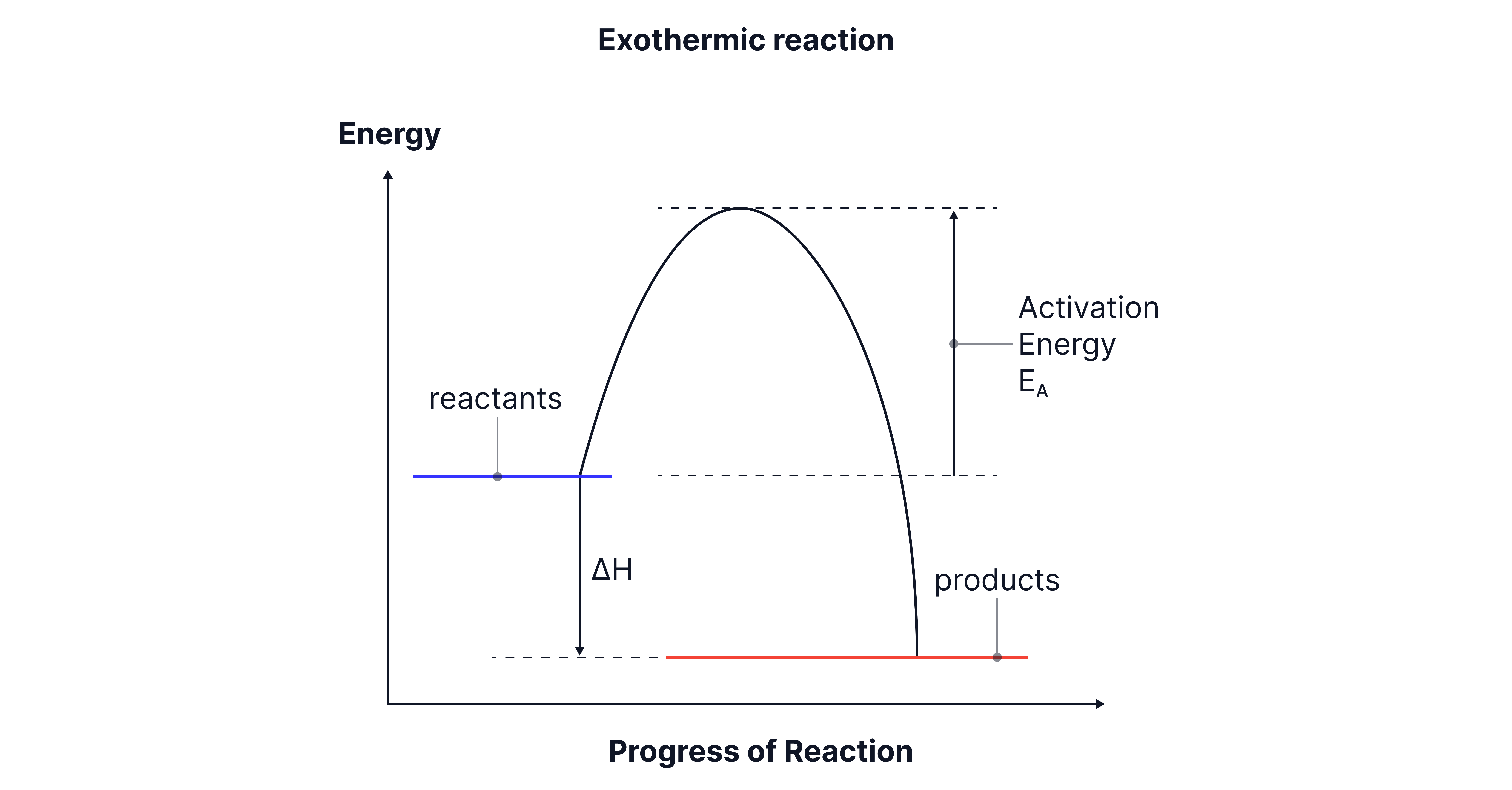
Products have lower enthalpy than reactants.
ΔH is negative.
The diagram slopes down overall, showing energy is released.
Example: combustion of methane.
Endothermic reaction:
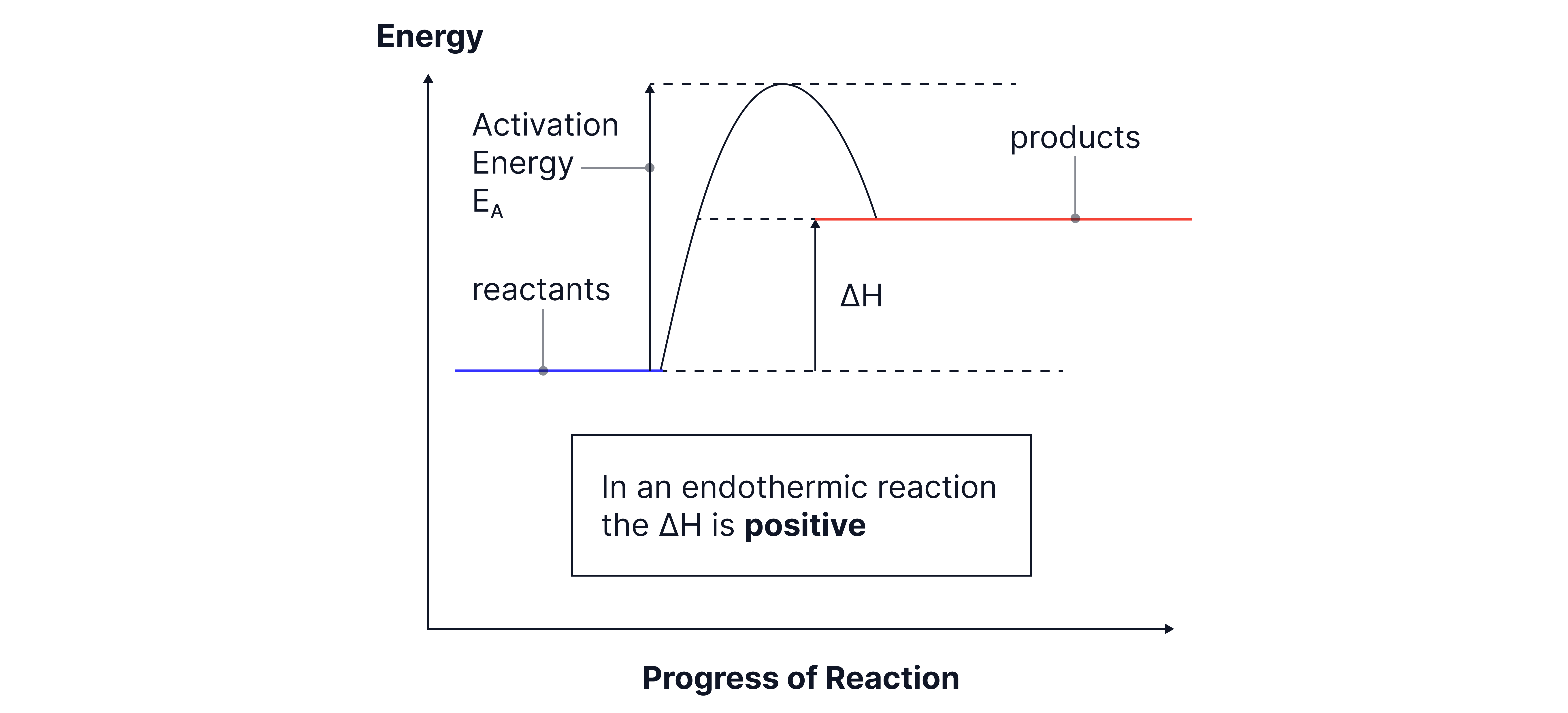
Products have higher enthalpy than reactants.
ΔH is positive.
The diagram slopes upwards overall, showing energy is absorbed.
Example: thermal decomposition of calcium carbonate.
Activation energy is shown as the “hump” between reactants and products, representing the minimum energy needed for the reaction to start. Even in exothermic reactions, you still need this initial input.
Key Types of Enthalpy Change
1. Enthalpy change of reaction – The heat change when a reaction takes place, in the quantities shown in the balanced equation, under stated conditions.
2. Standard enthalpy change of formation – The heat change when 1 mole of a compound forms from its elements in their standard states, under standard conditions .
3. Standard enthalpy change of combustion – The heat change when 1 mole of a substance burns completely in oxygen, under standard conditions.
Standard Conditions for Enthalpy Changes
Enthalpy changes are usually reported under standard conditions, which are:
Pressure of
Temperature of (25°C)
Solutions at a concentration of
All substances present in their standard physical state at
When an enthalpy change is measured under these standard conditions, the symbol is used.
Example: or
Some definitions that you need to know:
Enthalpy Change of Reaction
The enthalpy change of reaction is the heat energy change when the number of moles of reactants, as shown in the balanced chemical equation, react together under standard conditions.
Standard Enthalpy Change of Formation
The standard enthalpy change of formation of a compound is the enthalpy change when 1 mole of the compound is produced from its elements in their standard states, under standard conditions .
Examples:
Note: The standard enthalpy of formation of an element in its standard state is
Standard Enthalpy Change of Combustion
The standard enthalpy change of combustion is the heat energy released when 1 mole of a substance is burned completely in oxygen under standard conditions , with all reactants and products in their standard physical states.
Example:
Note: If combustion is incomplete, products may include carbon (soot), carbon monoxide, and water. Incomplete combustion releases less energy (less exothermic) compared to complete combustion.
Standard Enthalpy Change of Neutralisation
The standard enthalpy change of neutralisation is the heat energy change when an acid and an alkali react together in aqueous solution under standard conditions, producing exactly 1 mole of water.
Enthalpy and PV Work – Extension for A-Level Chemistry
In many chemical reactions, especially those involving gases, the system might expand or contract. This means that the system is doing pressure-volume work. For example, if a reaction produces a gas, the gas pushes against atmospheric pressure – that’s energy being used to do work.
To track this energy change, chemists use a quantity called enthalpy . Enthalpy is a measure of the total energy of a system, including both:
The internal energy – due to particles’ motion and interactions
The energy required to make room for the system by displacing the surrounding atmosphere
This is why enthalpy is defined as:
Enthalpy is particularly useful because at constant pressure, the heat change in a chemical reaction is equal to the enthalpy change . This is the key result we’ll now derive step-by-step.
Step-by-Step Derivation:
Step 1: First Law of Thermodynamics
Start with:
→ Total change in internal energy = heat + work done on the system.
Step 2: Define Pressure–Volume Work
In chemistry, the most common work is work. For expansion/compression at constant pressure:
(Negative because expansion means system loses energy.)
Substitute into Step 1:
Step 3: Define Enthalpy
Enthalpy is defined as:
Differentiate both sides:
Use product rule on :
So:
Step 4: Assume Constant Pressure
At constant pressure, so
Therefore:
Step 5: Substitute
From Step 2:
Substitute:
Conclusion
At constant pressure, the enthalpy change is equal to the heat transferred:
This is why is used in thermochemistry:
If a reaction gives off heat (exothermic),
If a reaction absorbs heat (endothermic),
Summary
We use enthalpy because:
and can’t be measured easily or directly.
is a state function (only depends on start and end points).
At constant pressure, heat exchanged, which is practical to measure.