Changes in State: Energy Changes
Lajoy Tucker
Teacher

Introduction and Definitions
State Change: Physical transformation involving energy changes (e.g. solid → liquid).
Melting point: The temperature at which a substance changes state from solid to liquid.
Boiling point: The temperature at which a substance changes state from liquid to gas.
Basic Principles
Changes of state are physical changes that involve breaking/forming attractive forces between particles.
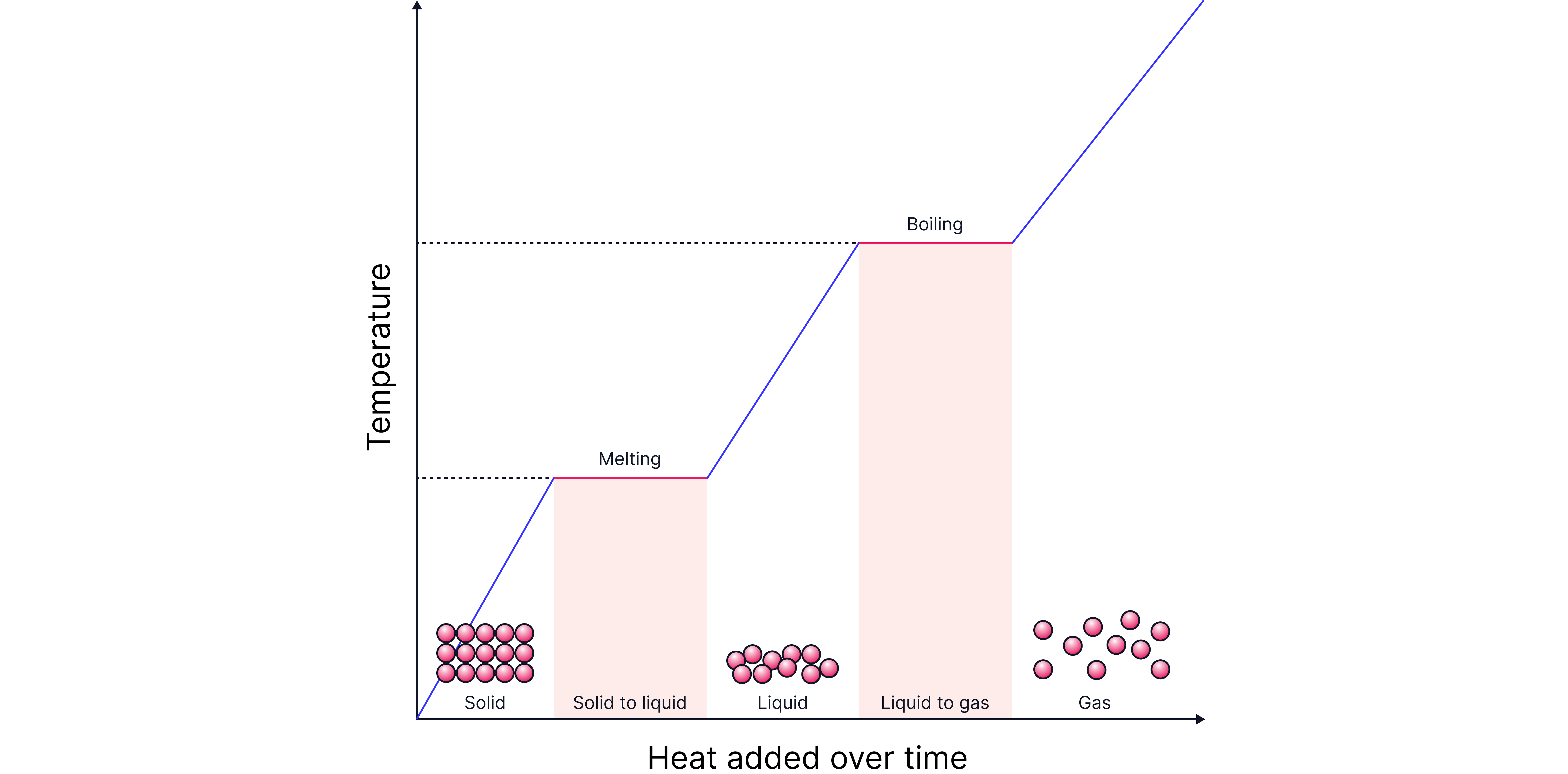
As heat energy is added, the average energy of the particles (temperature) increases.
During state changes (horizontal sections), all added heat energy is used to overcome the attractive forces between particles and so the temperature does not increase.
Melting and Boiling Points
The melting and boiling points depend on how much energy is required to overcome the attractive forces.
The stronger the forces of attraction, the more energy required to overcome these forces, and the higher the melting/boiling point.
Structure | For example… | Held together by… | Melting point/Boiling Point |
Giant ionic lattice | Many strong ionic bonds between oppositely charged ions | High | |
Simple molecular | Weak intermolecular forces | Low | |
Macromolecular/Giant covalent lattice | Diamond/graphite | Many strong covalent bonds between atoms | High |
Giant metallic lattice | Many strong metallic bonds between metal cations and delocalised electrons | High |
In general, substances with giant structure have high melting and boiling points, and simple molecules have low melting and boiling points.
Worked Example – Bonding Comparison
Question:
Why does NaCl have a higher melting point than ?
Answer:
is held together by many ionic bonds in a giant lattice which are stronger and require more energy to overcome than the forces between water molecules (intermolecular forces).
Practice Questions
Question 1
Explain why iodine (I₂) has a low melting point despite being a solid at room temperature.
Answer:
Weak van der Waals forces between molecules require little energy to overcome.
Question 2
Describe the energy changes that occur during the boiling of a liquid.
Answer:
Energy is absorbed (endothermic) to overcome intermolecular forces between particles, giving the particles enough kinetic energy to separate far enough for boiling
Question 3
Predict and explain the melting point order: NaCl, Ice, I₂
Answer:
> Ice >
: Strong ionic bonds
Ice: Hydrogen bonds
: Van der Waals forces
Ionic bonds stronger than H bonds stronger than VDWs
Key Tips:
Make sure you can spot the type of structure from the name or the formula. Further details of each bonding and structure type can be detailed in their respective revision notes.
Talk about forces between particles involved for the structure in question. Are bonds being broken or intermolecular forces?.