Structures of Covalent Substances
Lajoy Tucker
Teacher

Definition
Covalent substances consist of atoms bonded by shared pairs of electrons.
The structure of covalent substances affects their physical properties.
Types of Covalent Structures
1. Simple Molecular Substances
Simple molecular substances involve discrete molecules with weak forces between them (intermolecular forces).
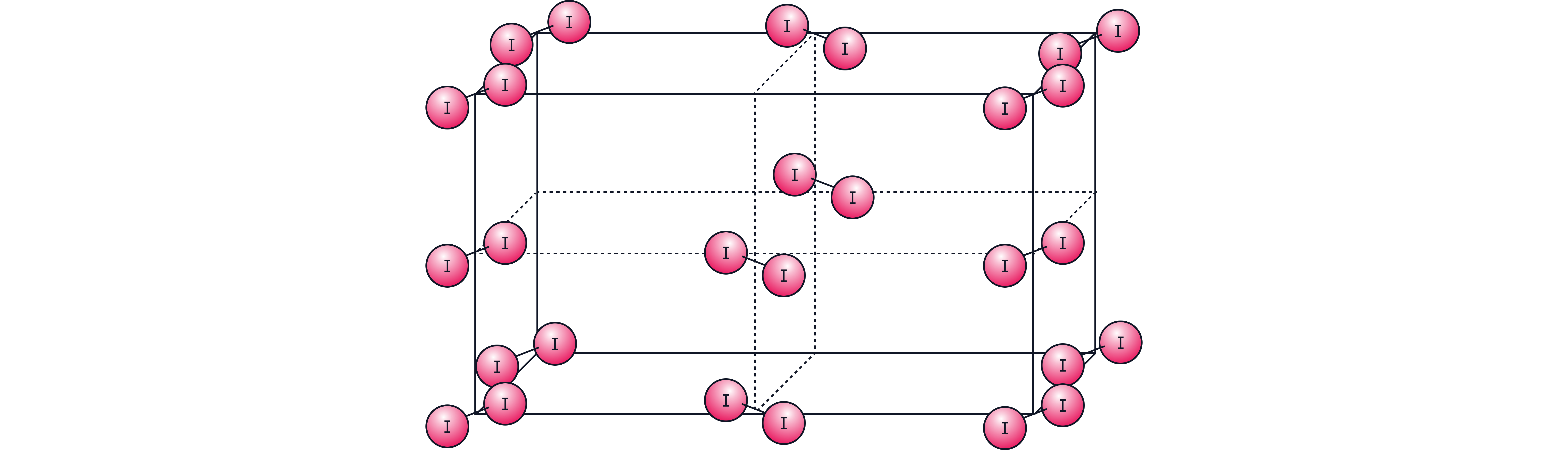
Examples include water (), the halogens ( to ), ammonia ()
These weak intermolecular forces are:
• van der Waal’s (London) forces
• Permanent dipole-dipole Interactions
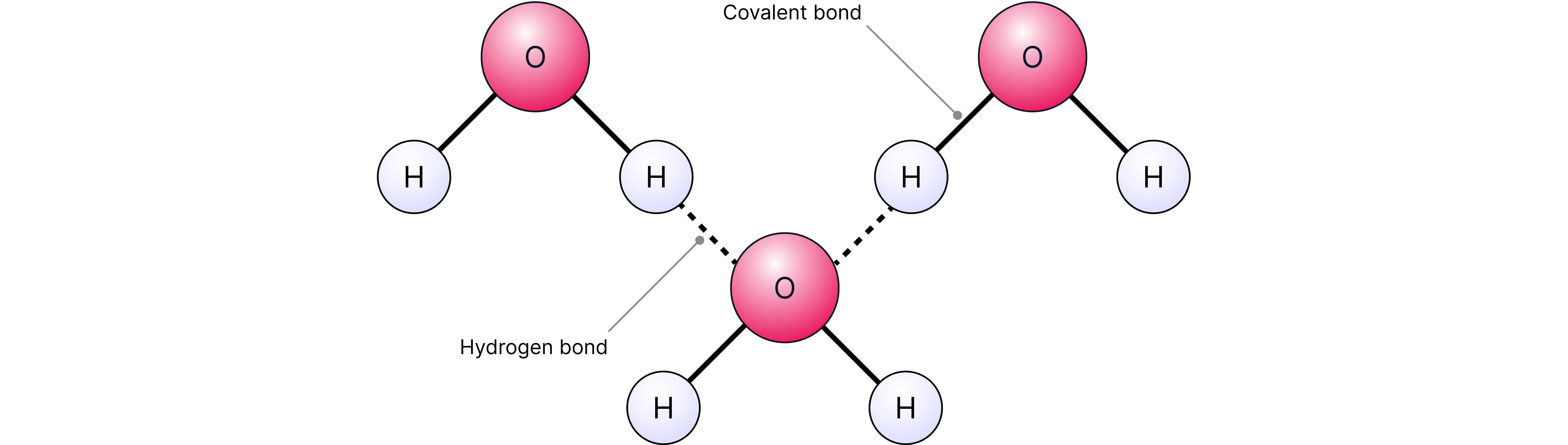
• Hydrogen bonding
Details of these intermolecular forces and how they arise can be found in the ‘Intermolecular Forces’ revision note
Properties of simple molecular structures
Low melting and boiling points:
Intermolecular forces are weaker than formal ionic, covalent, and metallic bonds. They therefore require little energy to overcome.
Examples:
is a solid at room temperature with a low melting point
exhibits hydrogen bonding, a stronger type of intermolecular force resulting in a higher boiling point compared to other molecules of similar size.
Do not conduct electricity
• There are no free ions or electrons to carry a charge.
2. Macromolecular Structures
• Also known as giant covalent structures
• These consist of a vast network of atoms connected by covalent bonds throughout the material.
Macromolecular structures have high melting and boiling points because breaking the extensive covalent network requires significant energy.
Other properties such as electrical conductivity depend on the substance in question.
Examples of Macromolecular structures:
Diamond:
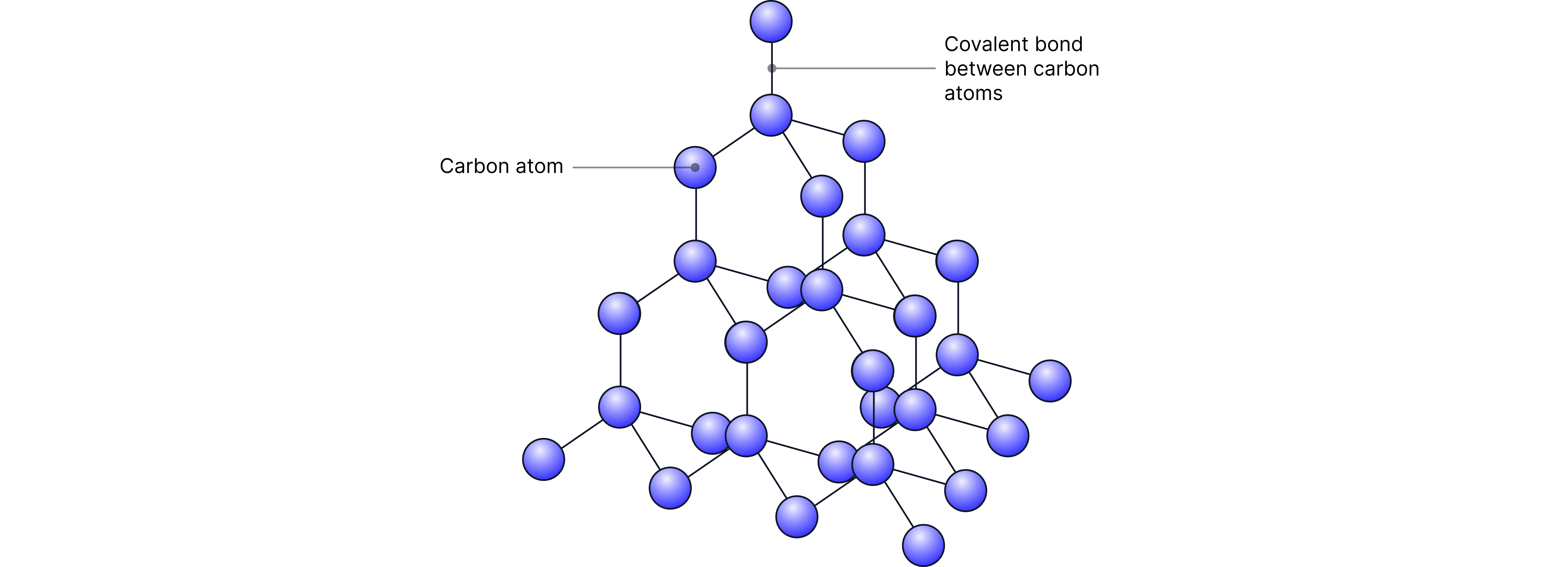
• Each carbon atom forms 4 covalent bonds resulting in 3D lattice with tetrahedral geometry around each carbon atom.
• Does not conduct electricity as there are no free charged particles (ions or delocalised electrons) to carry a charge.
Graphite:
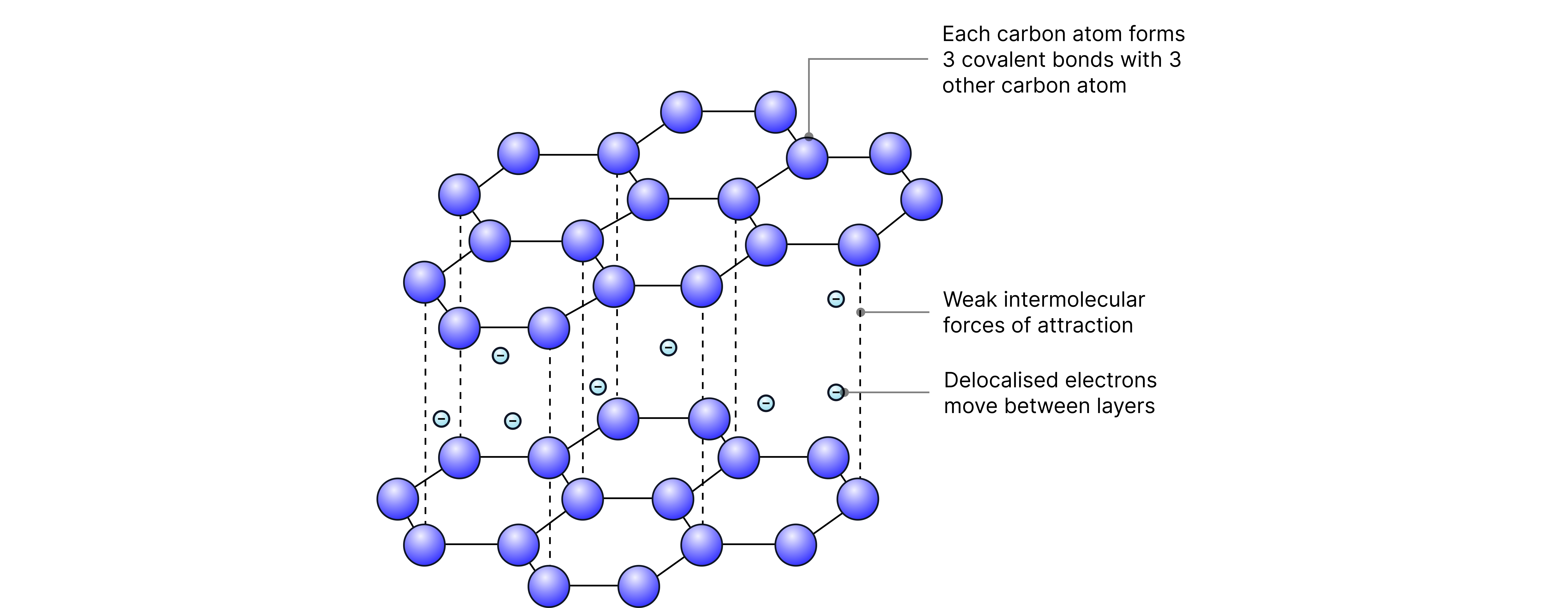
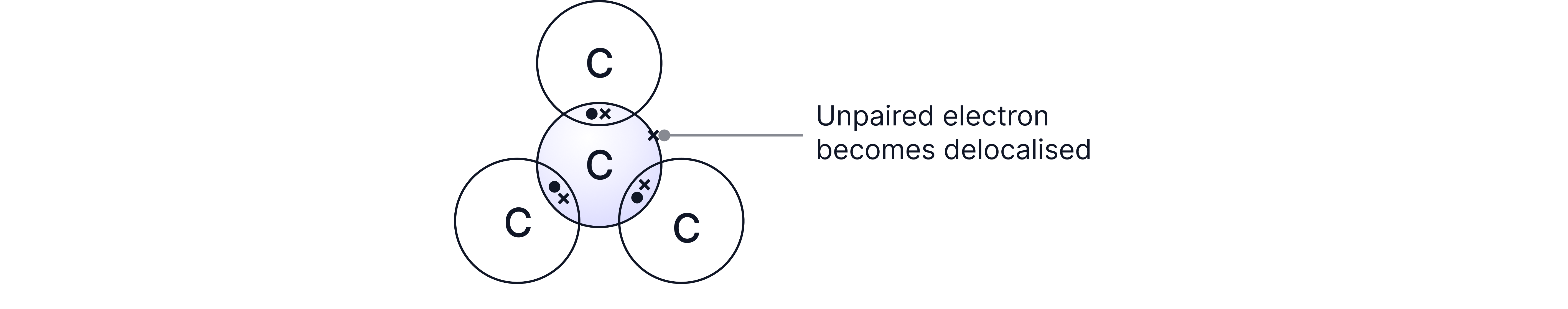
• Carbon atoms each form three covalent bonds, resulting in layers of hexagonal rings. These layers can slide over each other due to weak forces between them, making graphite slippery and useful as a lubricant.
• There is one electron per carbon atom not involved in bonding which becomes delocalised(free)
• Conducts electricity due to delocalised electrons that can move through the structure (between layers) and carry charge.
Silicon Dioxide ():
![]()
•Each silicon atom bonded to four oxygen atoms, and each oxygen to two silicon atoms.
•Similar structure to a diamond.
•Hard, high melting point, and does not conduct electricity.
Practice Question
Question
Which of the solids below has the lowest melting point?
A. Iodine
B. Diamond
C. Graphite
D. Silicon dioxide
Answer:
A. (Iodine)
Iodine has a simple molecular structure held together by intermolecular forces which are much weaker than the covalent bonds between the atoms in B-D’s macromolecular structures thus require less energy to overcome.