Structure and Bonding - Metals
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
What is Metallic Bonding?
Metallic bonding is the strong electrostatic attraction between a lattice of positive metal ions and a sea of delocalised electrons.
There is a very strong electrostatic force of attraction between these oppositely charged particles.
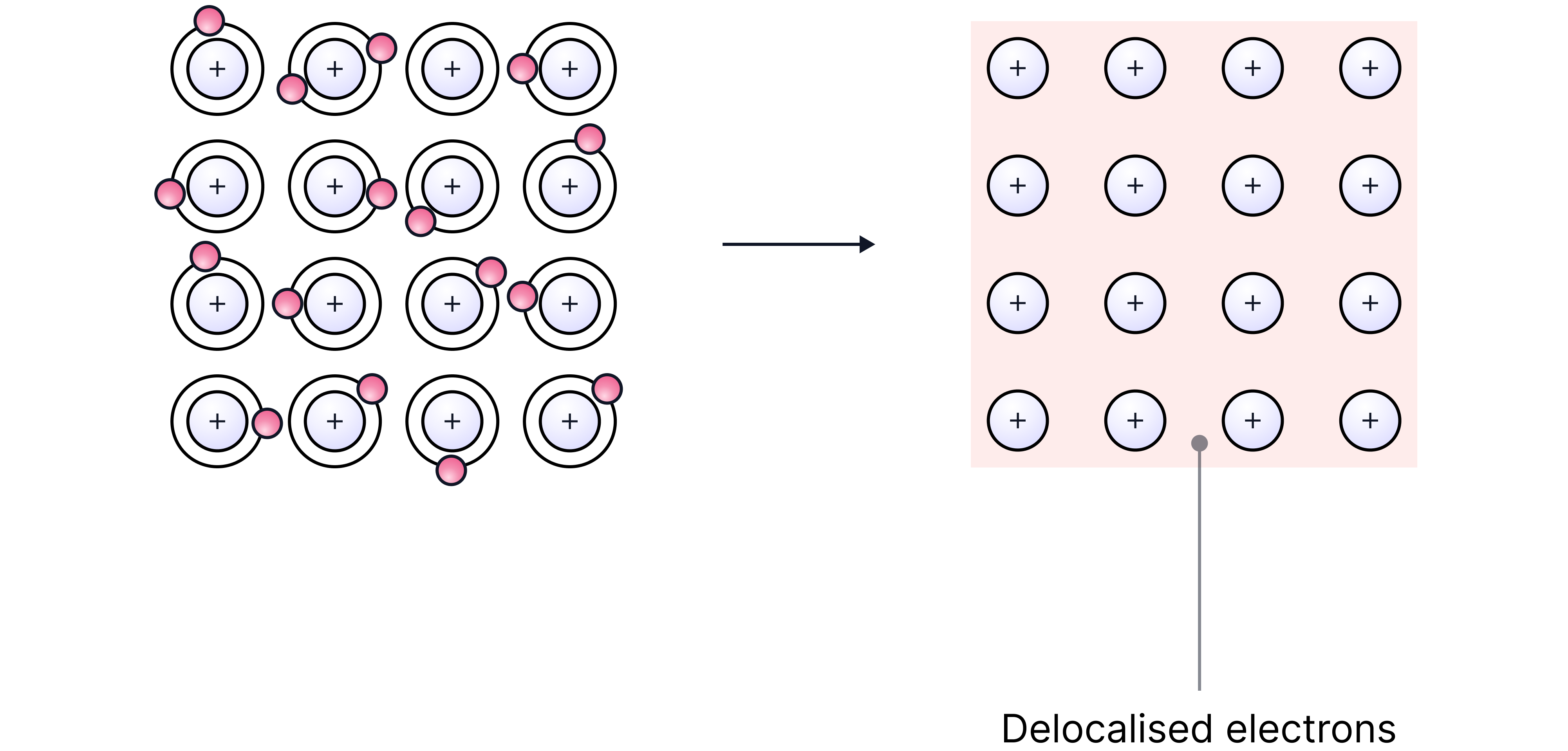
Structure of Metals
Giant Metallic Lattice:
Metals consist of layers of positive metal ions arranged in a regular pattern which can be described as a lattice.
The positive ions within the lattice are surrounded by a sea of delocalised electrons.
Outer shell electrons are free to move throughout the lattice.
They are not associated with a particular atom, which gives metals many of their properties.
Feature | Explanation |
Electrostatic Attraction | Between positive ions and delocalised electrons |
High Melting/Boiling Points | Strong metallic bonds require a lot of energy to break |
Conducts Electricity | Delocalised electrons can move and carry charge |
Malleable and Ductile | Layers of metal ions can slide over each other without breaking the bond due to the presence of delocalised electrons |
Lustrous/Shiny Appearance | Free electrons reflect light easily |
Factors Affecting the Strength of Metallic Bonding
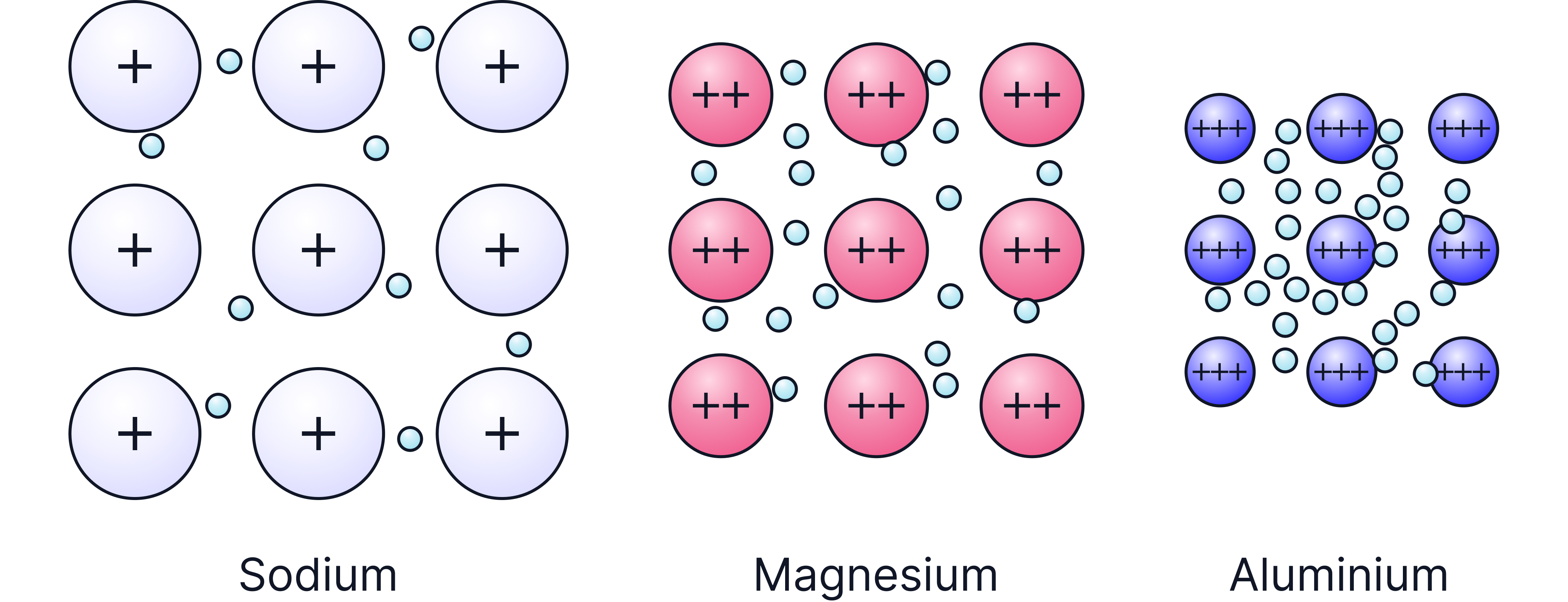
1. Number of Delocalised Electrons per Atom
The more delocalised electrons there are the stronger the attraction between them and the metal cations.
For example, the metallic bonding in magnesium is stronger than that in sodium as there are 2 delocalised electrons per magnesium atom (group 2) compared to the 1 delocalised electron for sodium (group 1).
2. Charge on Metal Ion
The higher the positive charge on the metal atom the stronger the attraction between the delocalised electron and the positive ion.
This explains why the metallic bonding in Aluminium is stronger than that in Magnesium:
3. Size of Metal Ion (Ionic Radius)
The smaller the metal ions the stronger the attraction between the nucleus and delocalised electrons.
This is because the distance between the nucleus and the outer delocalised electron is shorter and thus the electron experiences a stronger force of attraction.
This explains why the strength of metallic bonds (and as a result melting points) decrease down group 2
Question:
Which of the metals below has the weakest metallic bonds and therefore the lowest melting point?
A.
B.
C.
D.
Answer
B.
Potassium ions have a lower charge than Mg and Ba and a larger ion size than sodium ions.