Covalent and Dative Covalent Bonding
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Covalent Bonding
A covalent bond is the electrostatic attraction between a shared pair of electrons and the nuclei of two non-metal atoms.
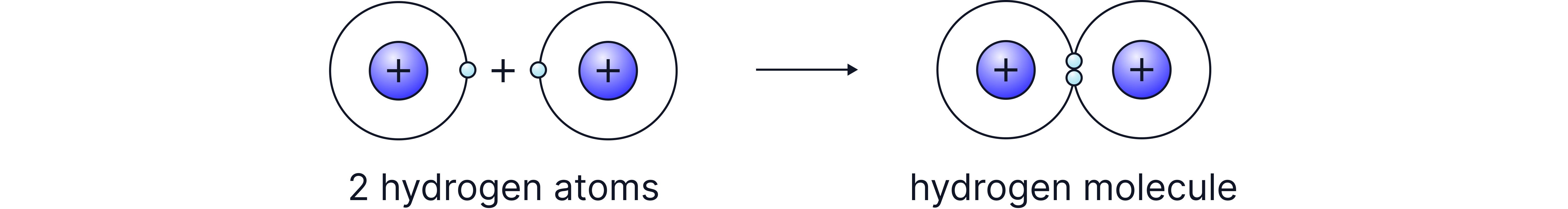
This type of bonding is typically found between non-metal atoms that need additional electrons to achieve a stable full outer shell.
Each electron pair shared forms a bond and multiple pairs can be shared to form multiple bonds.
Examples
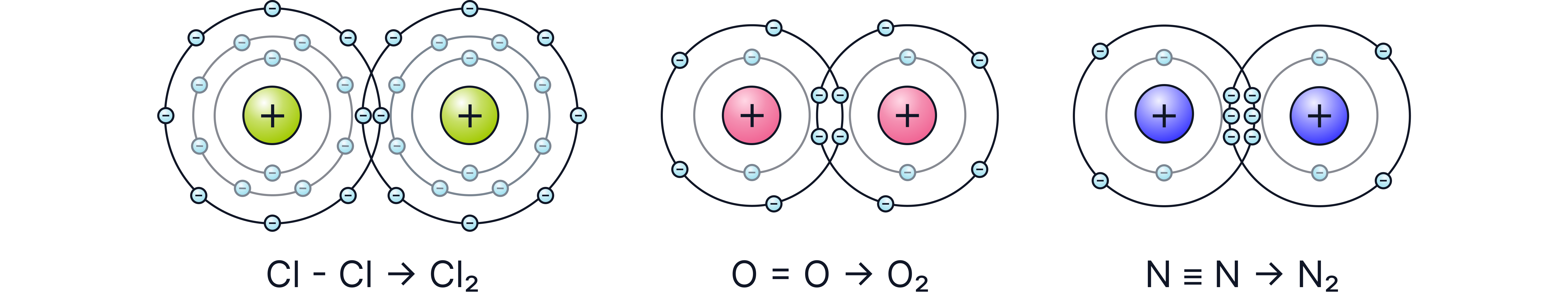
• (Chlorine): Each atom contributes one electron to the bond
• The electrons are shared not transferred. A pair of electrons are shared single covalent bond.
• (Oxygen): Double bond (two shared pairs).
• (Nitrogen): Triple bond (three shared pairs).
Dative Covalent (Coordinate) Bonding
A dative covalent bond is a covalent bond where both electrons in the shared pair come from the same atom.
Dative covalent bonds are also called coordinate bonds.
They are often formed between an atom with a lone pair and an atom with an incomplete outer electron shell.
Example 1 - Ammonium ion
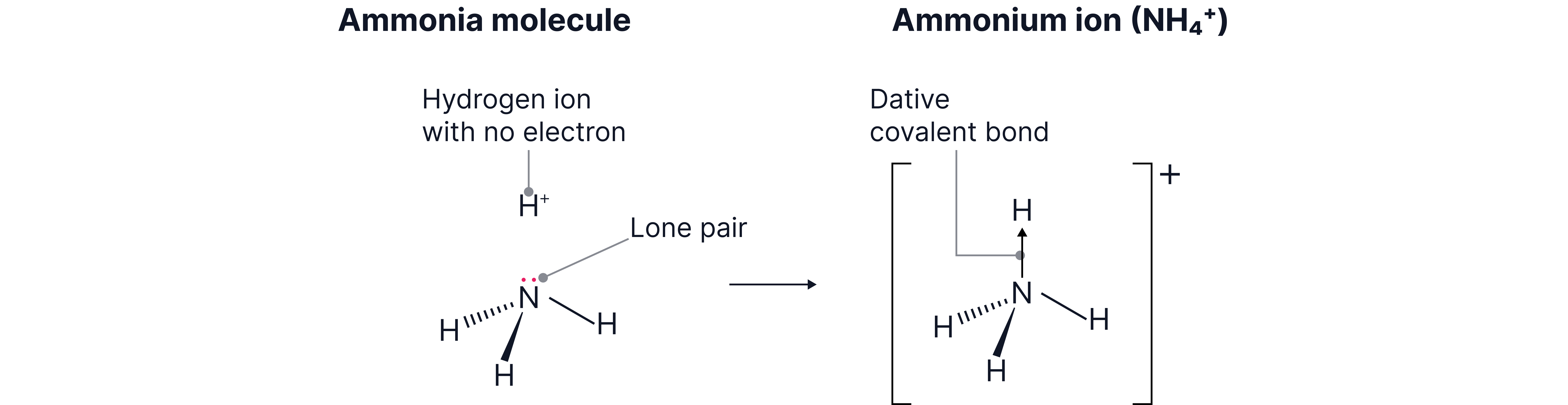
In the ammonium ion, the nitrogen atom of the ammonia molecule has a lone pair of electrons. The ion has no electrons and so can accept two to complete its outer shell.
The donates a lone pair of electrons to the ion and the arrow represents the direction of donation of the pair of electrons.
Dative covalent bonds are represented by an arrow pointing from the donor atom (the atom supplying both electrons) to the acceptor.
Example 2 - dimers
has a central Al atom with an incomplete octet (only 6 electrons) surrounded by atoms with lone pairs of electrons.
Two molecules can interact where lone pairs from atoms on two molecules donate a pair of electrons to the neighbouring electron deficient aluminum atoms forming two dative covalent bonds.
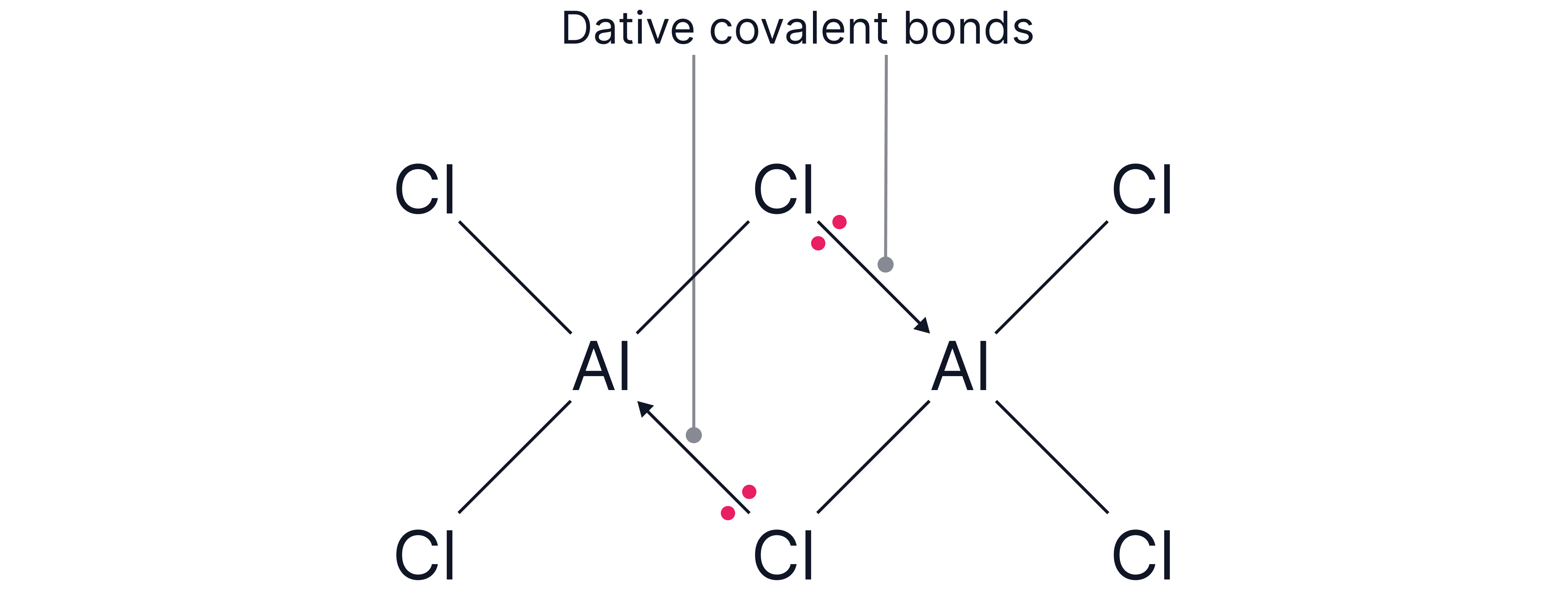
This forms the dimer
Note - The number of bonding pairs and lone pairs around the central atom of a molecule can be deduced using a dot and cross diagram or a counting method. Details of these can be found in the ‘Shapes of simple molecules and ions’ revision note.
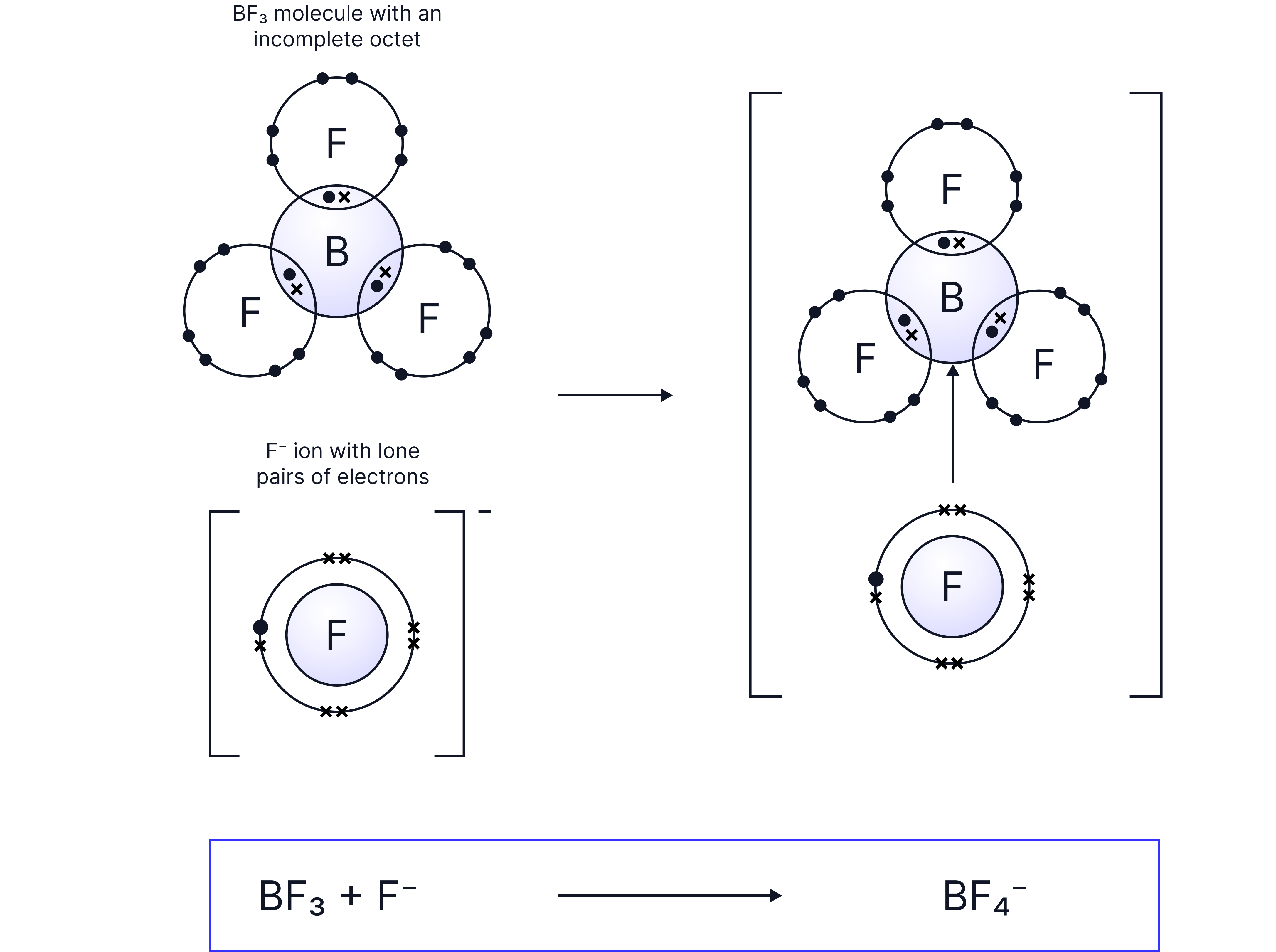
Question
molecules can form bonds with fluoride ions. Explain how this bond is formed.
Answer:
The ion donates a lone pair of electrons to the central boron atom of which only has 6 electrons on its outer shell. This forms a dative covalent bond.
Key Term Reminders
Covalent Bond – Shared pair of electrons between non‑metal atoms
Dative Covalent Bond – Shared pair of electrons donated by one atom
Lone Pair – Non‑bonding electron pair
Dot and Cross Diagram – Shows bonding and origin of electrons
Arrow () in bonding – Indicates coordinate bond direction