Structure and Bonding - Ionic
Lajoy Tucker
Teacher

Contents
Ionic Bonding
Ionic bonding occurs when electrons are transferred from a metal to a non-metal, forming positive and negative ions.
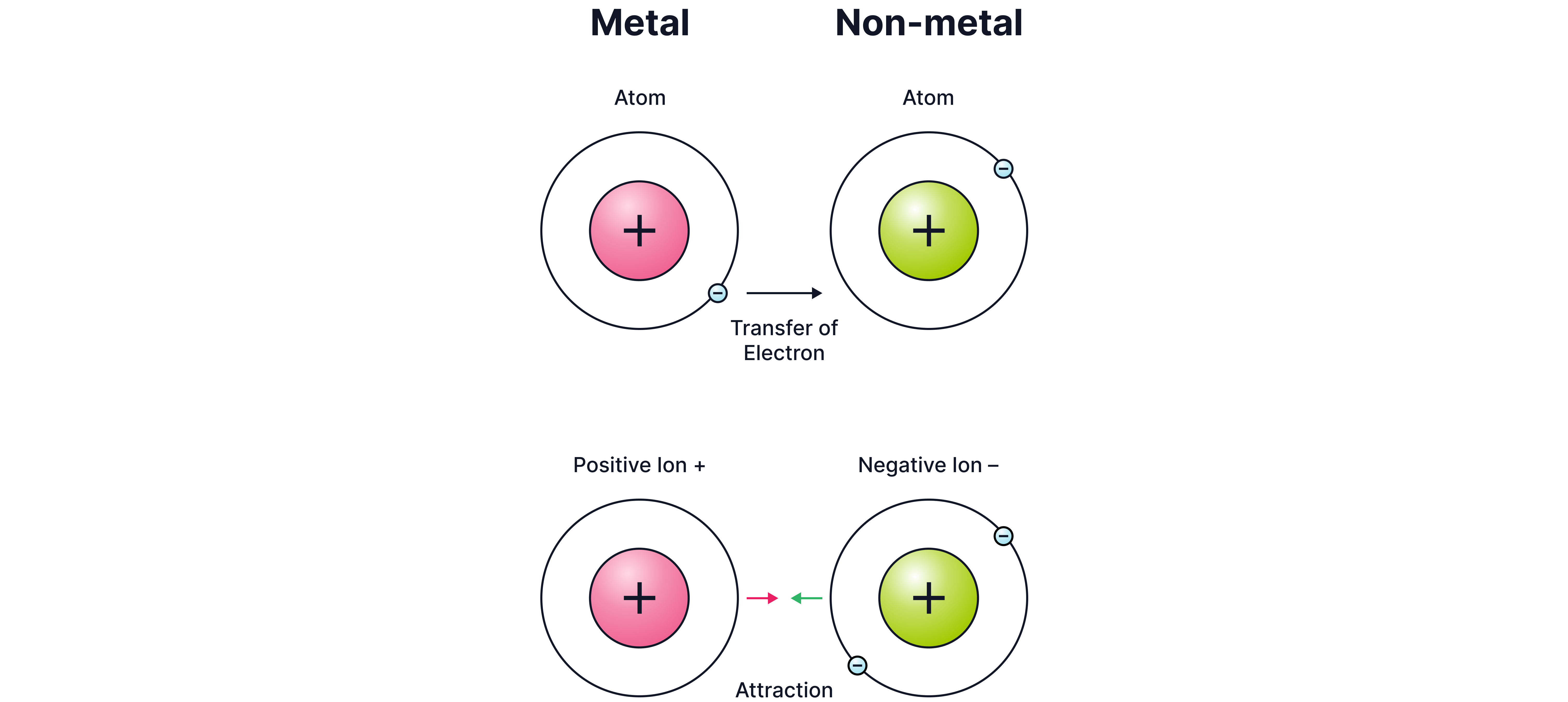
When the metal atom loses electrons, a cation is formed. Cations are positively charged ions.
When the non-metal atom gains electrons, an anion is formed. Anions are negatively charged ions.
Strong electrostatic forces of attraction hold together these oppositely charged ions.
Hence, by definition, ionic bonding is the electrostatic interaction between oppositely charged ions.
Charges of ions
Predicting the charge on a simple ion
You can predict the charge on a simple ion by using its group number in the Periodic Table. The group number gives the number of electrons on an atom’s outermost shell. In order to achieve a more stable full outer shell, atoms will lose or gain electrons.
Once the atoms become ions, their electronic configurations are the same as a noble gas, which is the most stable configuration.

| Group Number | e' lost/gained? | Charge of the ion | Example |
| 1 | Lose 1 | 1+ | |
| 2 | Lose 2 | 2+ | |
| 3 | Lose 3 | 3+ | |
| 5 | Gain 3 | 3- | |
| 6 | Gain 2 | 2- | |
| 7 | Gain 1 | 1- |
- Group 4 elements can form 2+,4+, or 4- ions.
Transition metal ions
Transition metals form ions with variable oxidation states. Rather than predicting the charges using the periodic table, the oxidation state is given in Roman numerals in parentheses e.g. iron
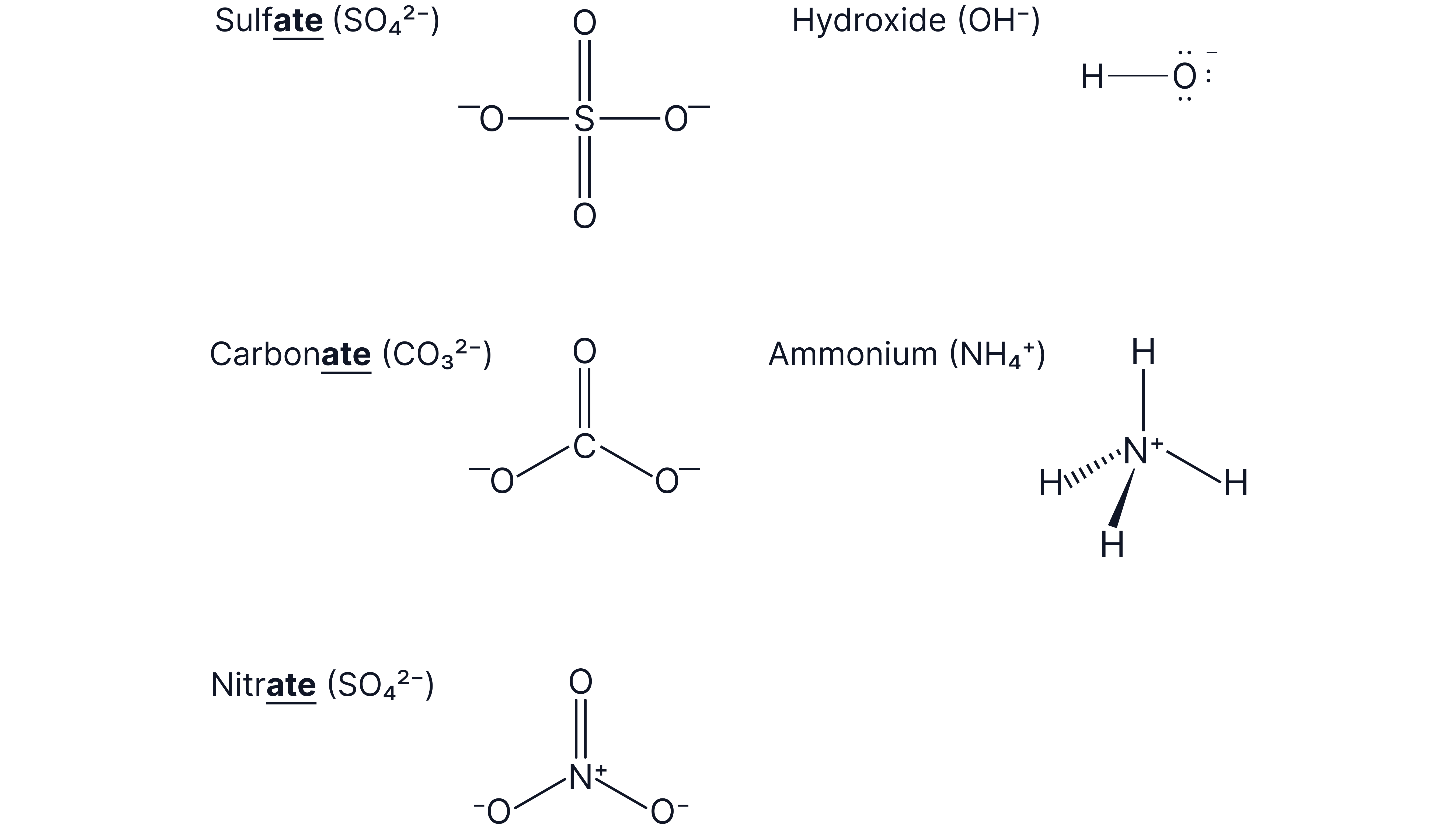
Compound ions
A compound (or polyatomic ion) consists of two or more atoms chemically bonded together containing a net charge.
Examples:
Ionic Formula
Oppositely charged ions combine in a ratio that leads to a compound with no overall charge.
e.g. sodium chloride
and give
e.g. potassium oxide
and gives
Two ions are required to cancel the charge of the ion
e.g. aluminium sulfate
and gives
Two ions with three ions result in equal and opposite charges and
Note a ‘swap and drop’ shortcut can be used to quickly deduce the formula of ionic compounds.
e.g. Magnesium nitride

This works as swapping the numbers allows cross-multiplication of the values cancelling out the charges (2x3 = 3x2)
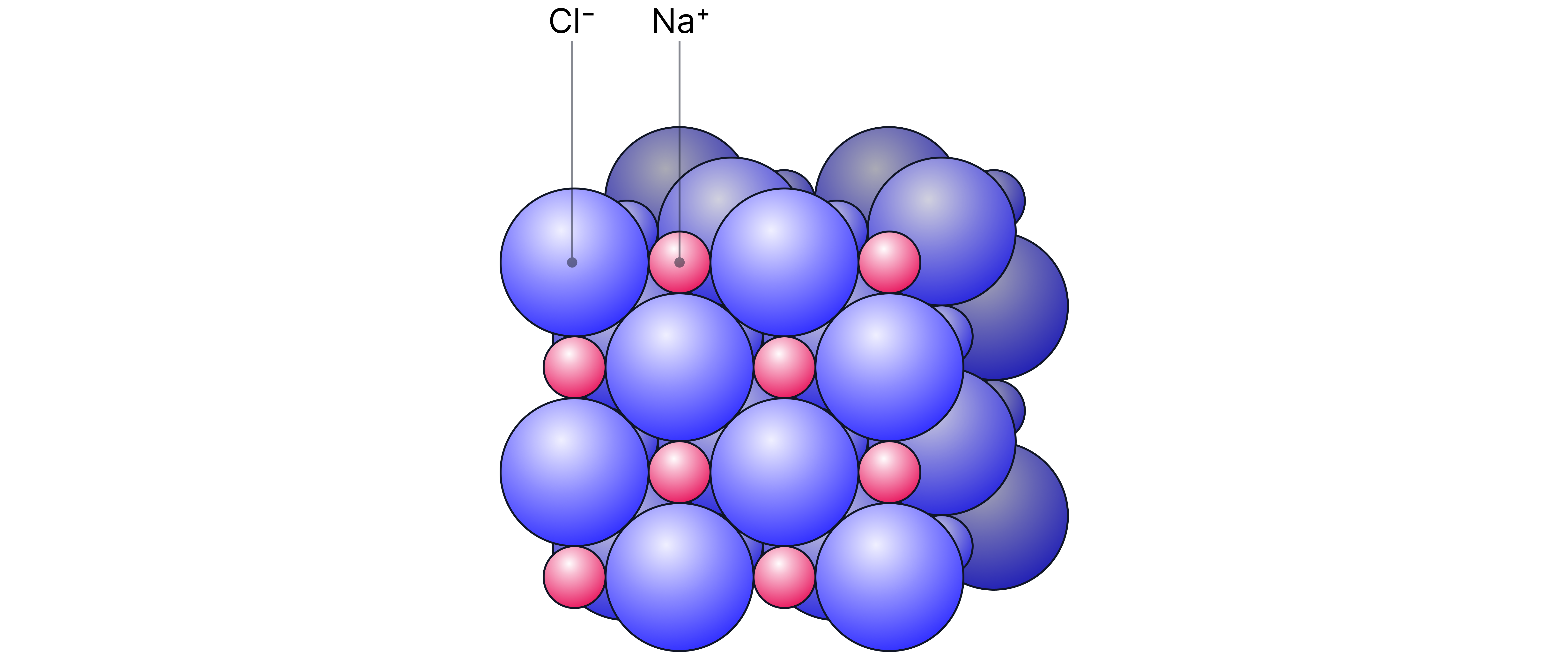
Giant Ionic Lattice Structure
A giant ionic lattice is a repeating three-dimensional structure of ions held by ionic bonds.
Compounds that are bonded ionically contain ions that are arranged in a regular 3D lattice. Each positive ion is surrounded by negative ions and vice versa.
The ions are held together by strong ionic bonds in all directions.
Physical properties of ionic compounds
Ionic Compounds have high melting and boiling points:
Lots of energy is required to overcome the many strong electrostatic forces of attraction between the oppositely charged ions in the lattice.
The greater the charge and the smaller the size of the ion the stronger the electrostatic forces of attraction are and the higher the melting point.
e.g. has a higher melting point than as the and have higher charges and smaller ionic radii than and resulting in stronger ionic bonds.
Electrical conductivity:
Ionic compounds do not conduct electricity in the solid state but do in the molten state.
This is because in the solid state, the ions are in a fixed lattice whereas in the molten/ dissolved state, the ions are free to move and can carry charge.
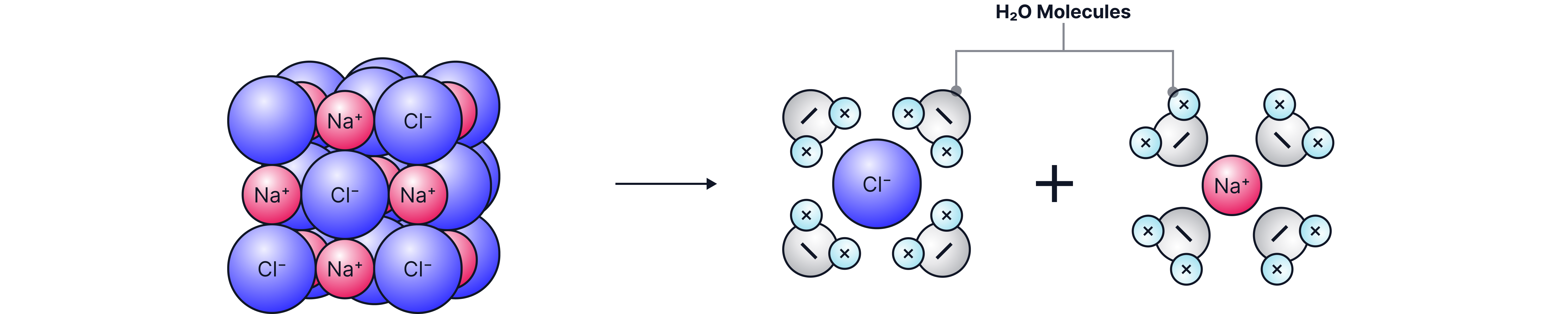
Solubility in water
• Many ionic compounds dissolve in water
• Water is a polar solvent which stabilises the separated ions
• The solubility of an ionic compound depends on the strength of the ionic bond, the stronger the ionic bond the greater the electrostatic force between the positively and negatively charged ions and more energy is required to break these bonds apart. These compounds are less soluble as it takes a significant amount of energy to break these ions apart and dissolve the compound.
e.g. is highly soluble, is less soluble.
Key Tips and Reminders
Use the periodic table to deduce the charges of main group ions.
Ions ending in -ate are oxygen-containing compound ions. Be careful not to mix these up with the simple ions ending in -ide.
Consider both the size and the charge of the ions when considering the strength of an ionic bond.