Collision Theory
Lajoy Tucker
Teacher

Contents
Reaction Profile Diagram
This diagram shows the energy change during a reaction.
Reactants start at a higher energy level.
To react, particles must climb an energy barrier – this is
The difference between the energy of the reactants and products is (enthalpy change).
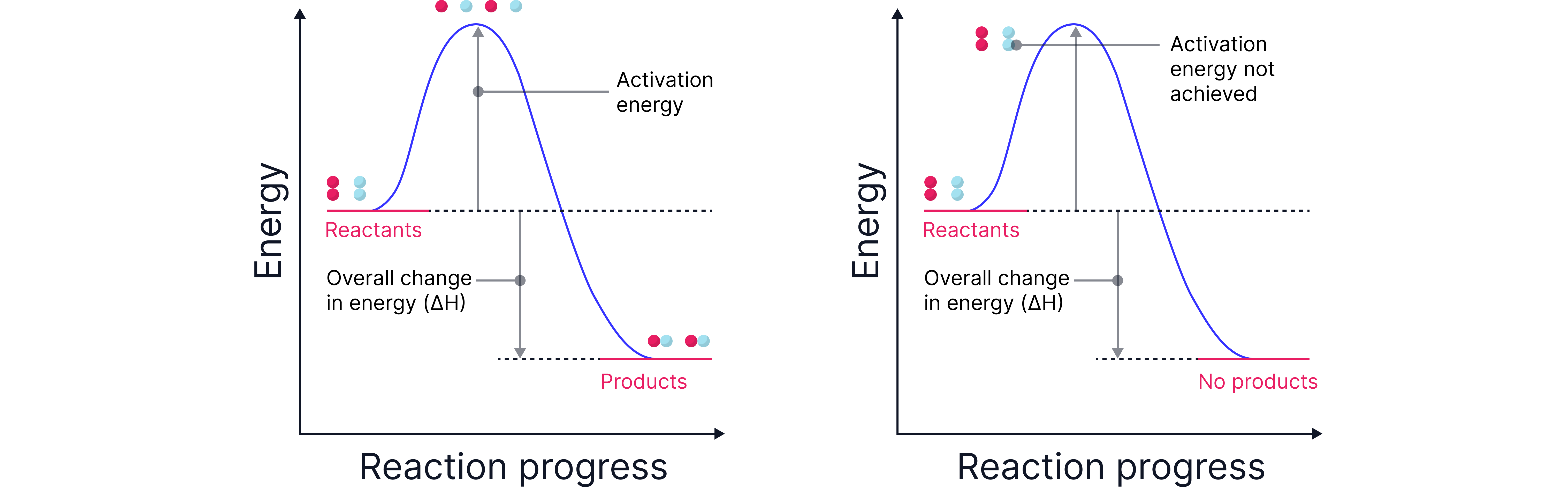
For a chemical reaction to occur, particles must collide with sufficient energy. This energy must be enough to break bonds in the reactants.
The minimum amount of energy required for a successful collision is known as the activation energy ().
If particles collide with less than this energy, they simply bounce off each other.
Collision Theory of Chemical Reactions
The rate of a chemical reaction refers to how quickly reactants are converted into products. Reactions occur when particles collide successfully – but not all collisions lead to a reaction.
For a collision to be successful, two conditions must be met:
Particles must collide with sufficient kinetic energy to overcome the activation energy barrier.
Particles must collide with the correct orientation to allow bonds to break and form effectively.
Activation Energy :
This is the minimum amount of energy required for a collision to result in a reaction. It’s the energy needed to break bonds in the reactants.
Rate of reaction increases when the number of successful collisions per second increases.
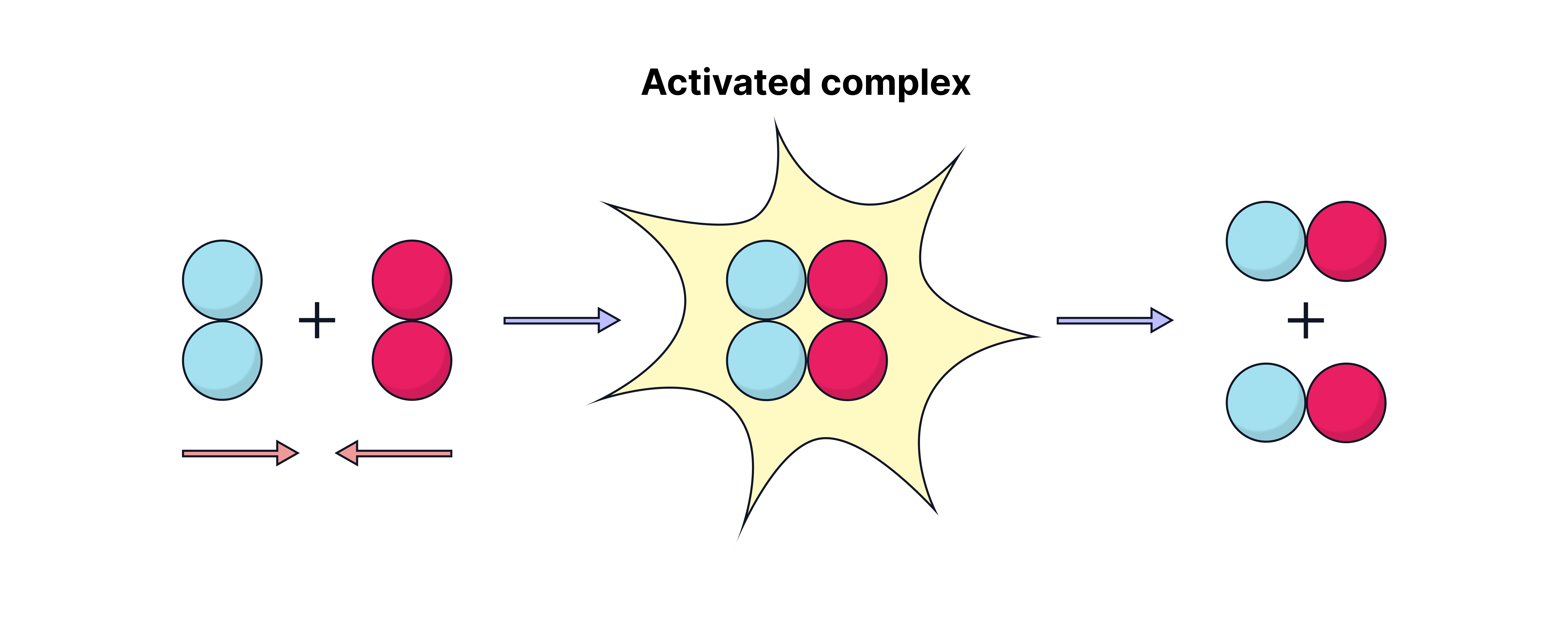
What happens during a successful collision?
Formation of the Activated Complex (Transition State):
Reactant particles collide with enough kinetic energy
An activated complex forms – a high-energy, unstable intermediate
Bonds in the reactants break, and new bonds in the products start to form
If the collision also has correct orientation, the reaction proceeds
Products are formed, and the system loses energy as it stabilises.
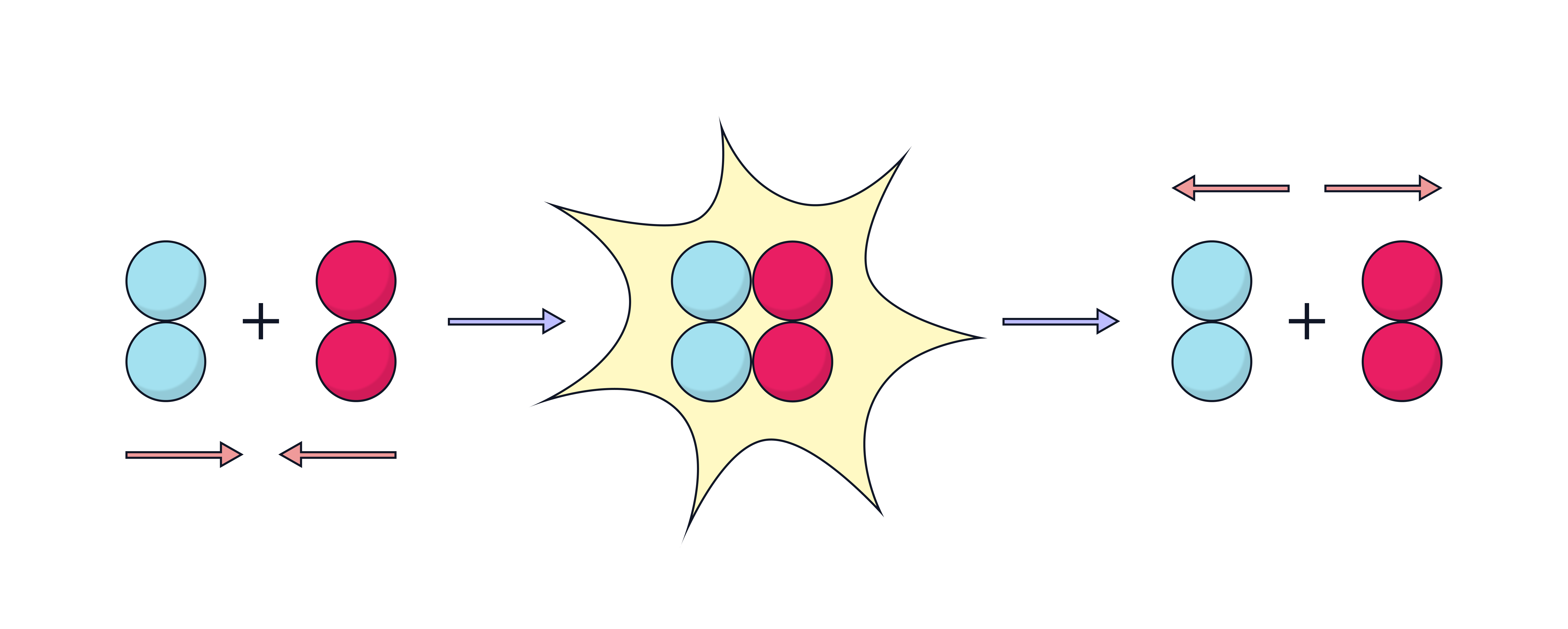
What causes an unsuccessful collision?
Particles collide with too little energy
They do not overcome the activation energy barrier
No bonds break, so no new bonds form
Particles simply bounce apart with no chemical change
Even if the orientation is correct, the reaction will not proceed without enough energy.
Orientation matters too!
Even if particles have enough kinetic energy, they must approach each other in a suitable spatial arrangement for bonds to form
If the orientation is incorrect, no reaction takes place – the collision is unsuccessful
Visual analogy: Imagine two puzzle pieces colliding. They must be aligned correctly to fit (form a product). If they’re misaligned, they bounce off.
Summary Table
|
Collision Type |
Energy ≥ Eₐ |
Correct Orientation |
Reaction Occurs? |
|
Successful |
✅ |
✅ |
✅ |
|
Energy too low |
❌ |
✅ or ❌ |
❌ |
|
Wrong orientation |
✅ |
❌ |
❌ |
|
Wrong energy + angle |
❌ |
❌ |
❌ |
Effect of Increasing Concentration and Pressure on Reaction Rate
Core Idea: When you increase the concentration of a solution or the pressure of a gas, you increase the number of particles per unit volume.
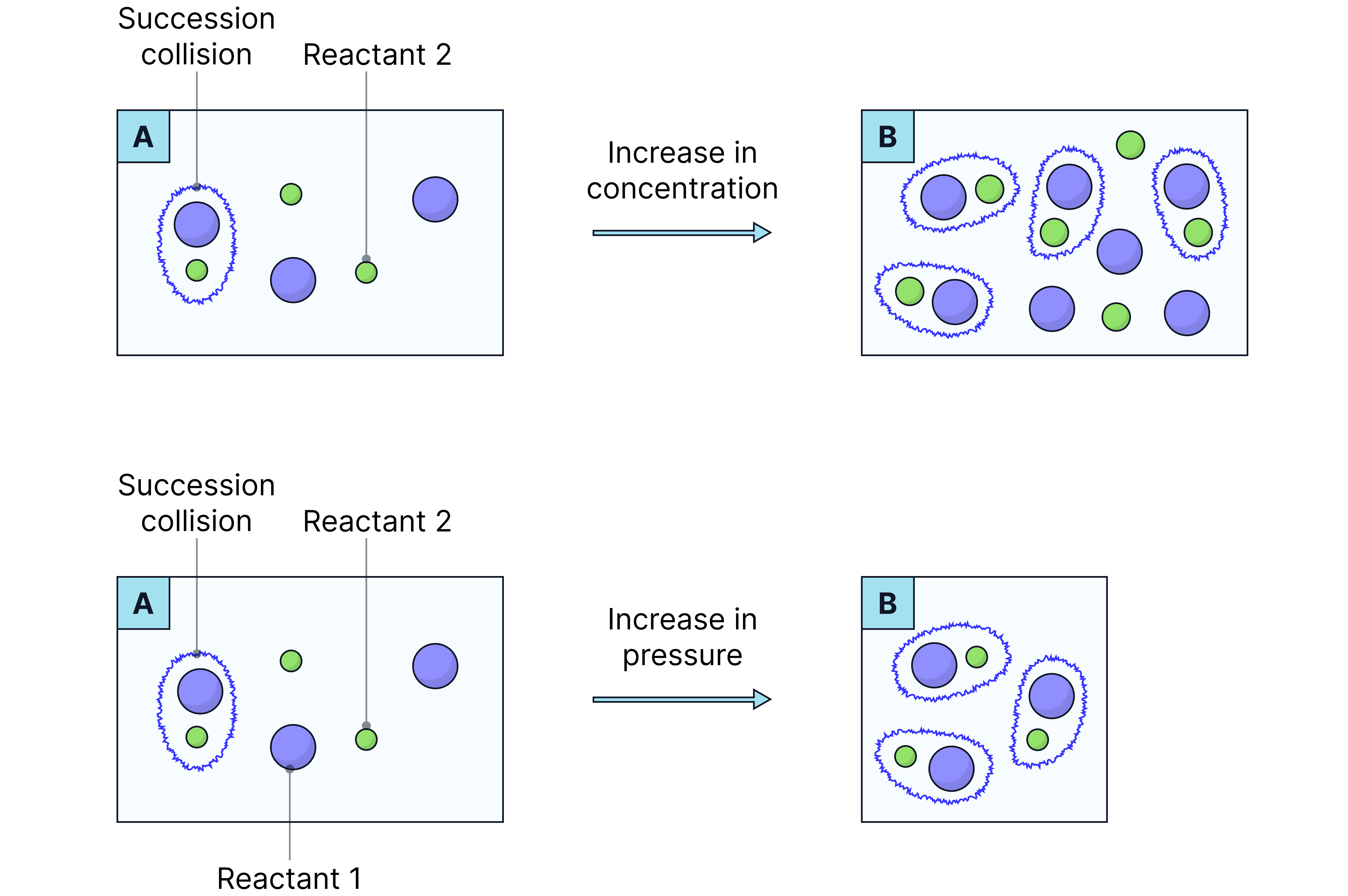
This leads to:
More frequent collisions between particles
A higher rate of successful (effective) collisions
Therefore, an overall faster rate of reaction
Exam Tip – Use Precise Language:
If a question refers to doubling concentration or pressure, make sure you say:
"Doubling the number of particles per unit volume"
"Doubles the frequency of successful collisions"
Avoid vague terms like "more collisions" – be quantitative and specific.
Energy Distribution Doesn’t Shift with Concentration
Increasing concentration does not change the shape of the Maxwell–Boltzmann distribution:
The most probable energy (Eₘₚ) and mean energy stay the same
But the curve becomes taller, and the area under the curve increases
This is because there are more total particles, not because individual particles are more energetic
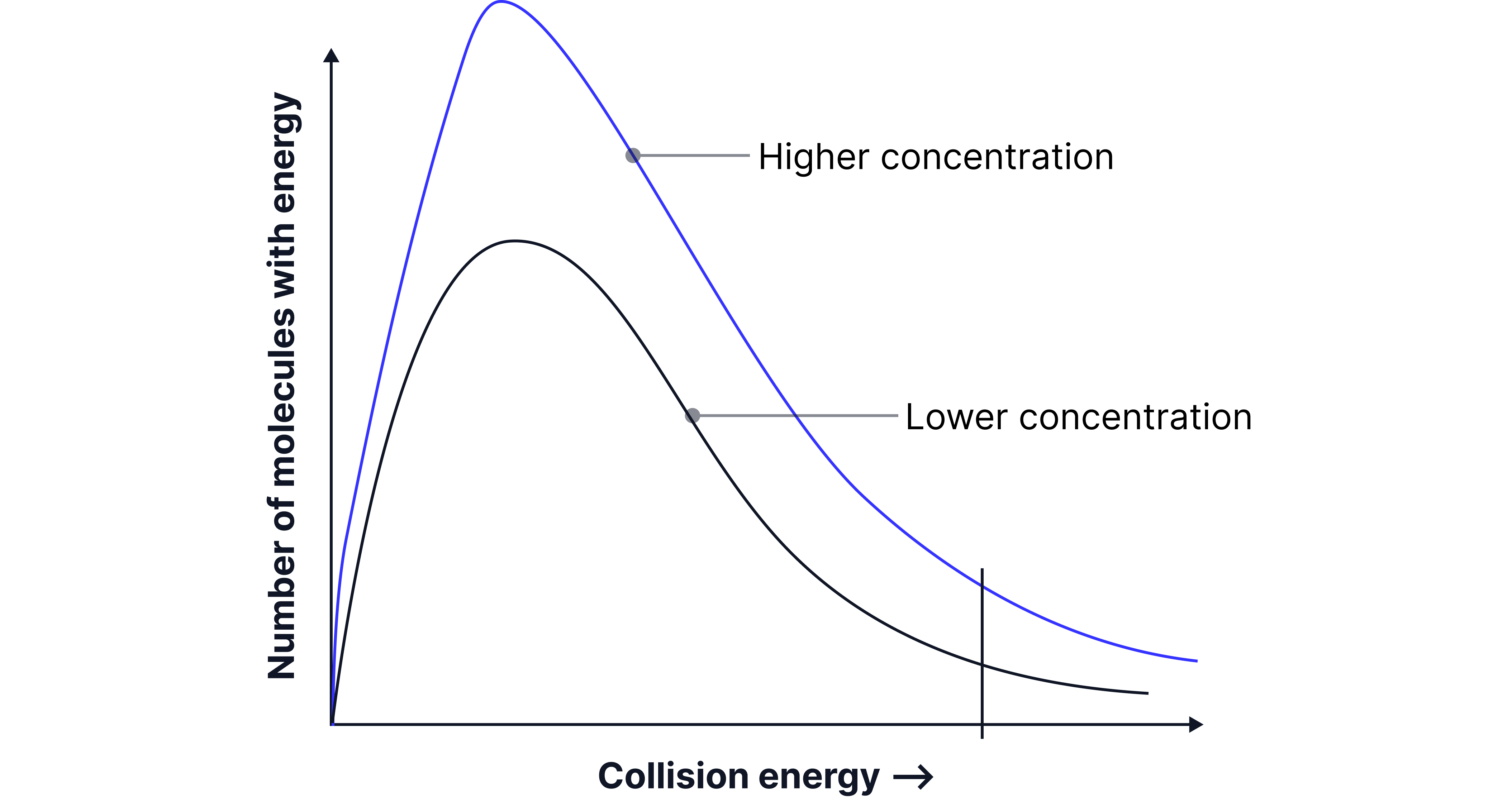
Result: There are more particles with energy ≥ Eₐ, even if the proportion doesn’t increase.
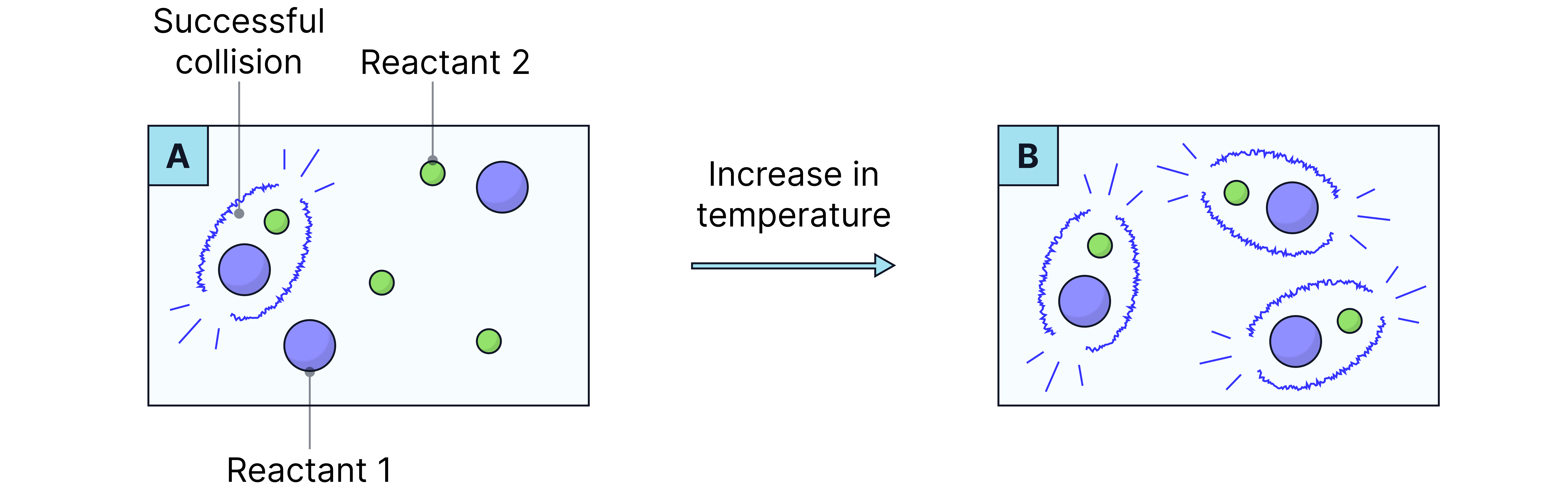
Effect of Increasing Temperature on Reaction Rate
When temperature increases, particles gain more kinetic energy, so:
They move faster, increasing the frequency of collisions per second.
A greater proportion of particles have energy ≥ activation energy (Eₐ).
This leads to a higher frequency of successful collisions, so the rate of reaction increases.
On the Maxwell–Boltzmann distribution:
The curve becomes broader and lower.
The peak shifts to the right, indicating a higher average energy.
The area beyond Eₐ increases significantly, meaning many more particles now have enough energy to react.
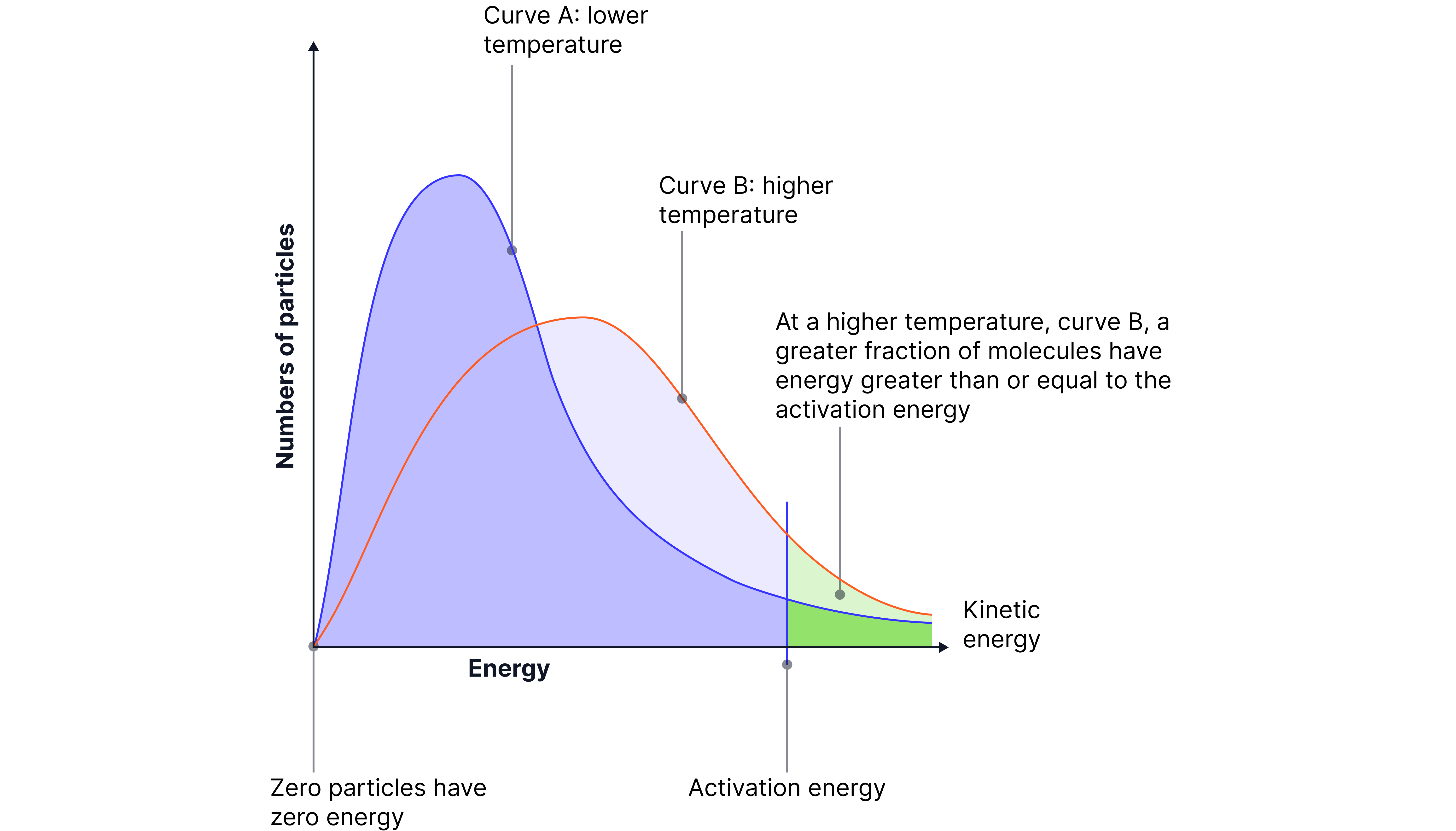
Key phrasing for exams:
“A significantly greater proportion of particles have energy equal to or greater than the activation energy, increasing the rate of successful collisions.”
Effect of Increasing Surface Area
When a solid reactant is broken into smaller pieces (e.g. powdered), its surface area increases, exposing more particles to potential collisions.
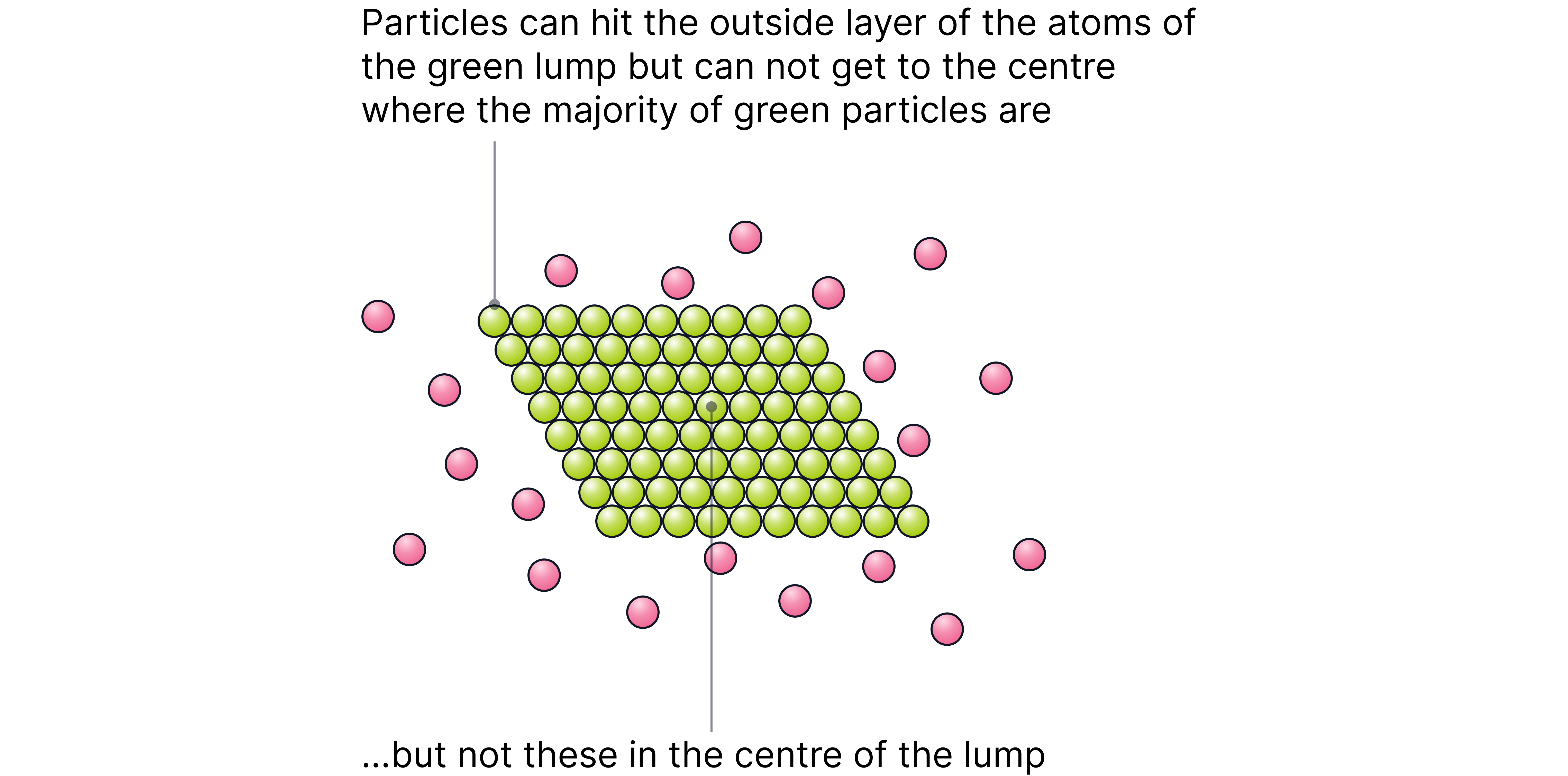
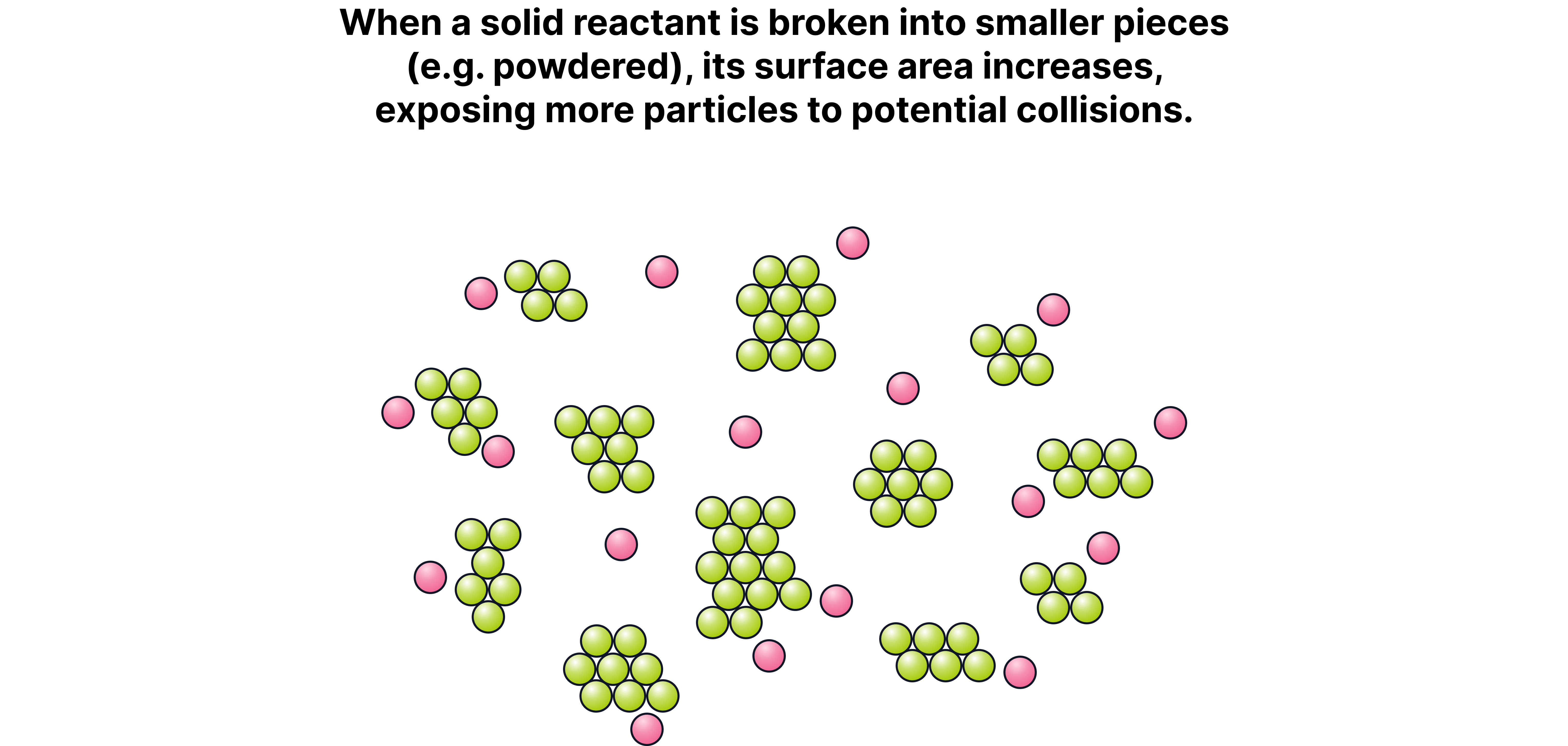
This leads to more collision sites per second.
The frequency of successful collisions between solid and liquid/gas reactants increases.
As a result, the rate of reaction is higher, though the activation energy and energy distribution stay the same.
Use in responses:
“Larger surface area → more frequent collisions → faster reaction.”
Effect of Catalysts on Reaction Rate
A catalyst is a substance that increases the rate of a chemical reaction without being chemically changed or used up in the process.
How it works:
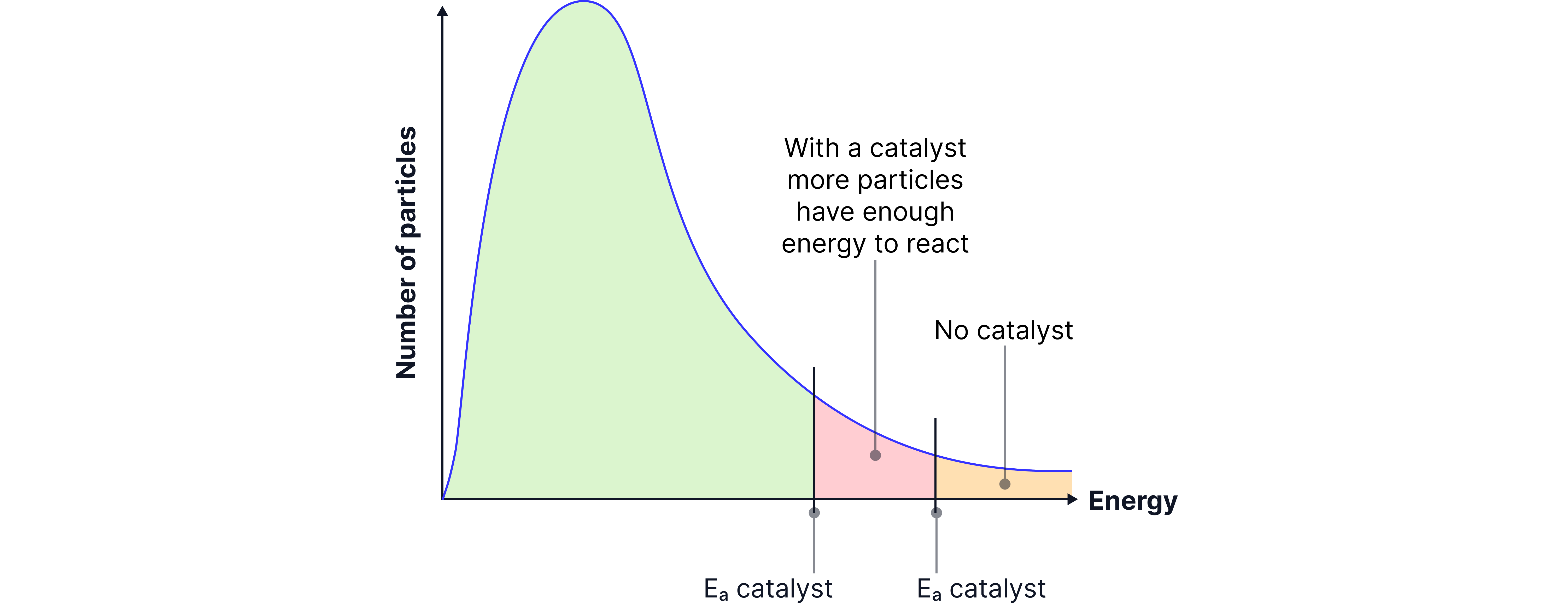
It offers an alternative reaction pathway with a lower activation energy (Eₐ).
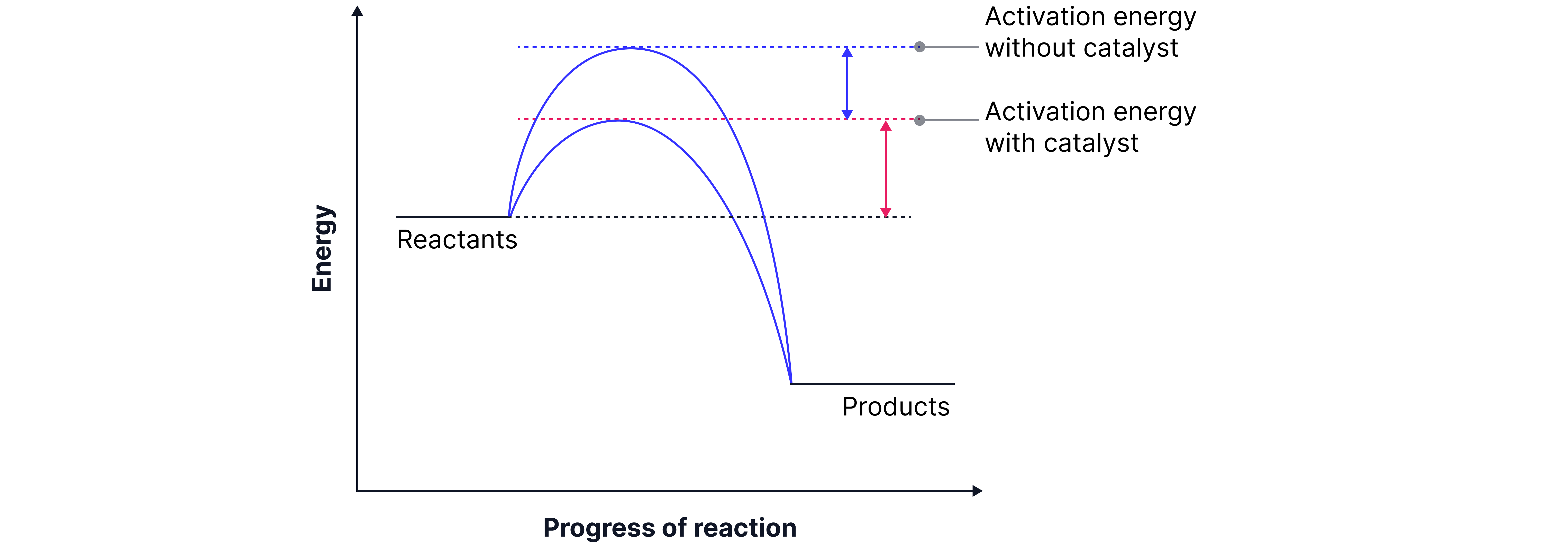
This means that more particles now have energy ≥ new, lower Eₐ.
Hence, the frequency of successful collisions increases, even though the overall energy distribution doesn’t change.
Worked Examples
Question: The rate of reaction of sodium thiosulfate with hydrochloric acid increases when the temperature is raised. Calculate the effect of increasing temperature by 10°C on the rate of reaction, given the following data:
Rate constant at 20°C:
Rate constant at 30°C:
Answer:
Temperature increases the collision energy and frequency, making the reaction occur faster.
The rate of reaction roughly doubles when the temperature is increased by 10°C, as seen in the increase of the rate constant from 0.02 to 0.05.
Question: Explain why increasing the temperature of a reaction generally increases the rate of reaction, according to collision theory.
Answer:
According to collision theory, for a reaction to occur, particles must collide with sufficient energy (activation energy) and proper orientation.
Increased temperature increases the kinetic energy of particles, meaning that they move faster.
As particles move faster, they collide more frequently and with greater energy.
More frequent collisions increase the likelihood of successful collisions, where the particles have enough energy to overcome the activation energy barrier.
Higher energy collisions increase the proportion of collisions that have the necessary activation energy for the reaction to occur.
As a result, increasing temperature increases the rate of reaction by increases the frequency and energy of particle collisions.