Commercial Cells including the Lithium Cell
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Introduction to Commercial Cells
Commercial electrochemical cells convert chemical energy into electrical energy to power our devices. They fall into two main categories:
1. Non-rechargeable cells (Primary cells) - use once and dispose
2. Rechargeable cells (Secondary cells) - can be recharged and reused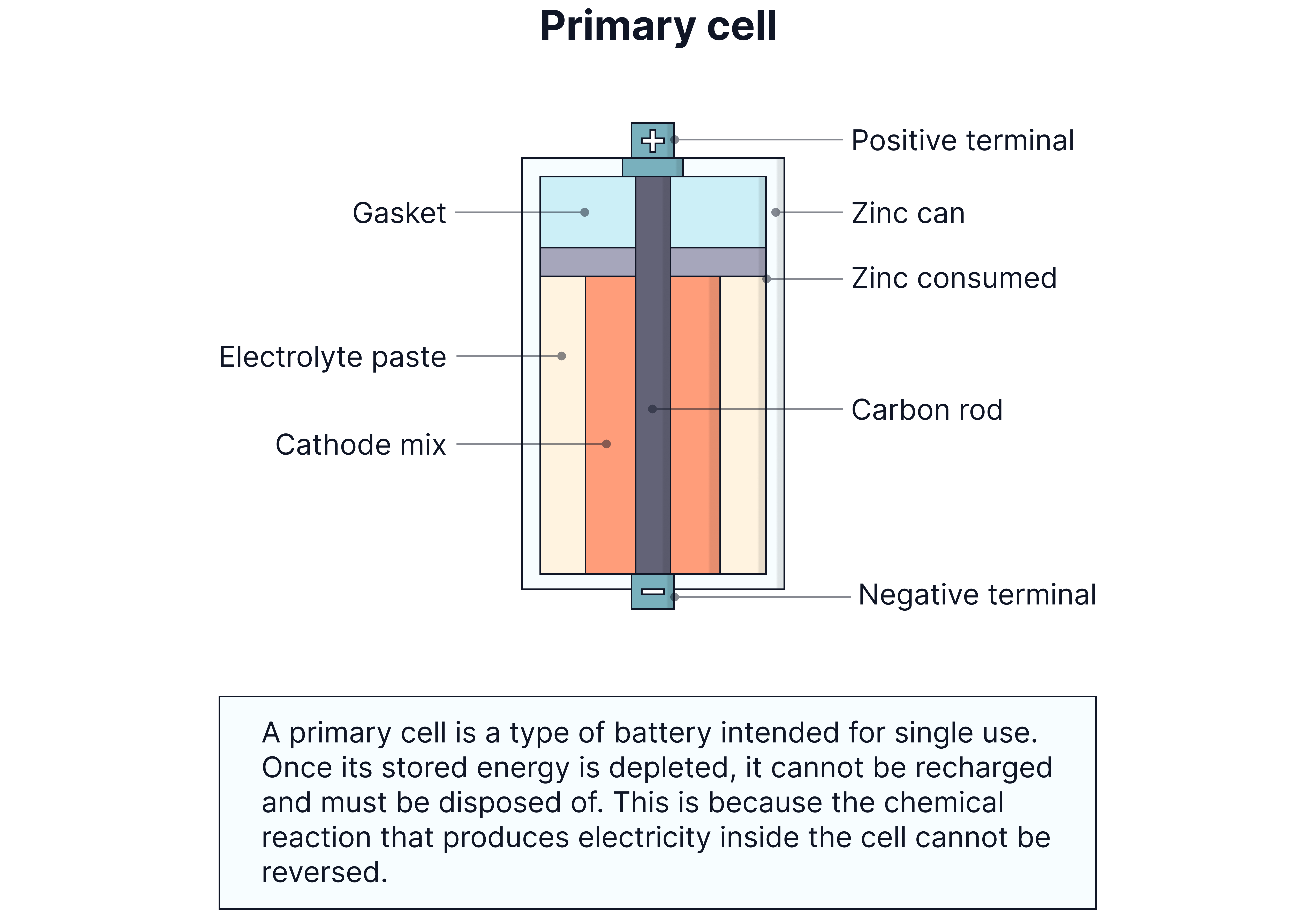
Non-Rechargeable Cells (Primary Cells)
How They Work
Non-rechargeable cells have irreversible chemical reactions. Once the reactants are used up, the cell is "dead" and must be thrown away.
Key characteristics:
Single use only - cannot be recharged
Reactions are irreversible - products cannot be converted back to reactants
Voltage drops to zero when chemicals are used up
Usually cheaper initially but more expensive long-term
Zinc-Carbon Cell (Leclanché Cell)
The standard household battery found in remote controls, clocks, and flashlights.
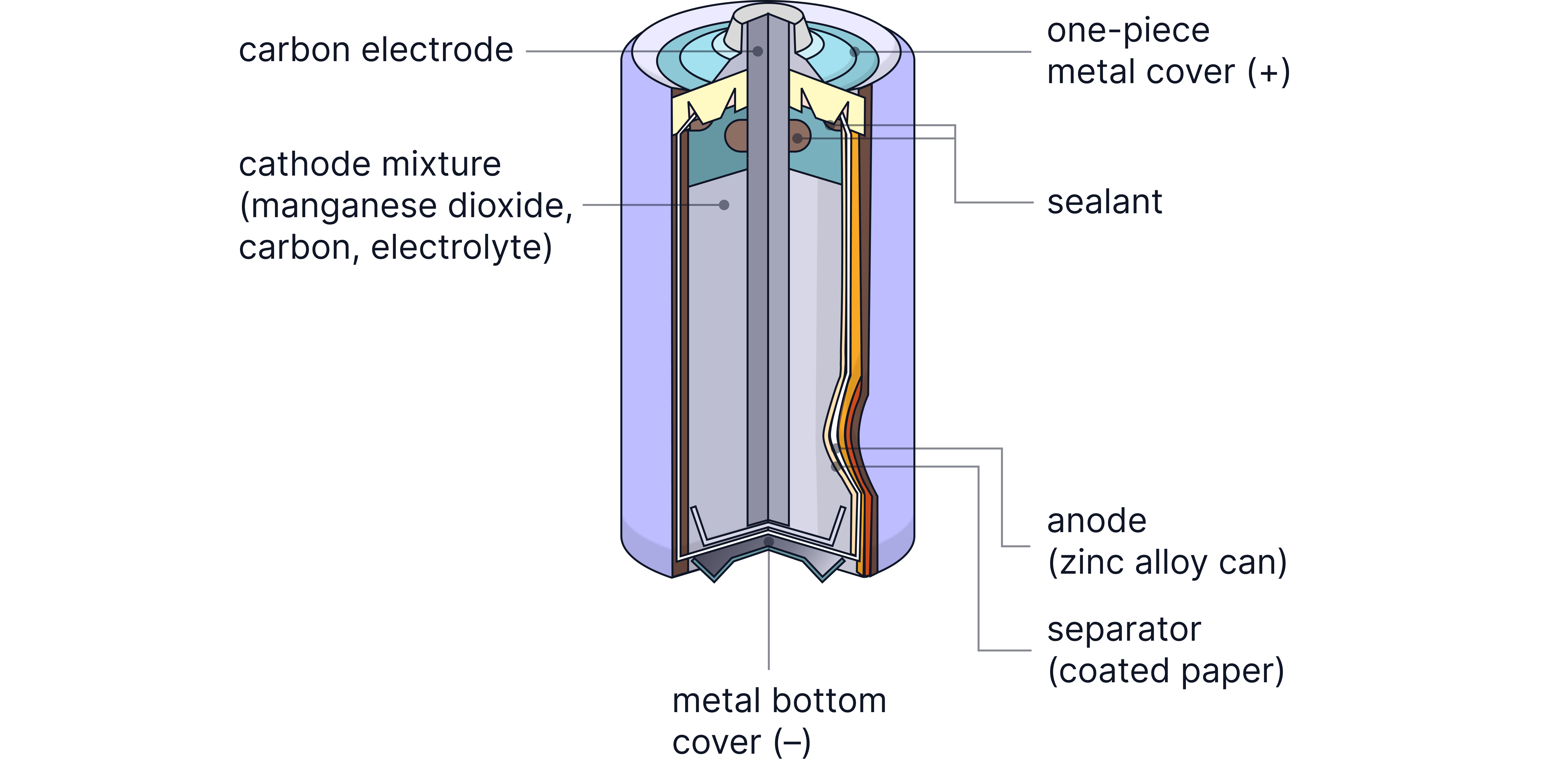
How it works:
Negative electrode (anode): Zinc case dissolves
Positive electrode (cathode): Carbon rod with manganese dioxide paste
Half-reactions:
Anode:
Cathode:
Cell voltage:
Pros: Very cheap
Cons: Short life, voltage drops during use, can leak
Alkaline Cell
The premium household battery - lasts longer and performs better than zinc-carbon.
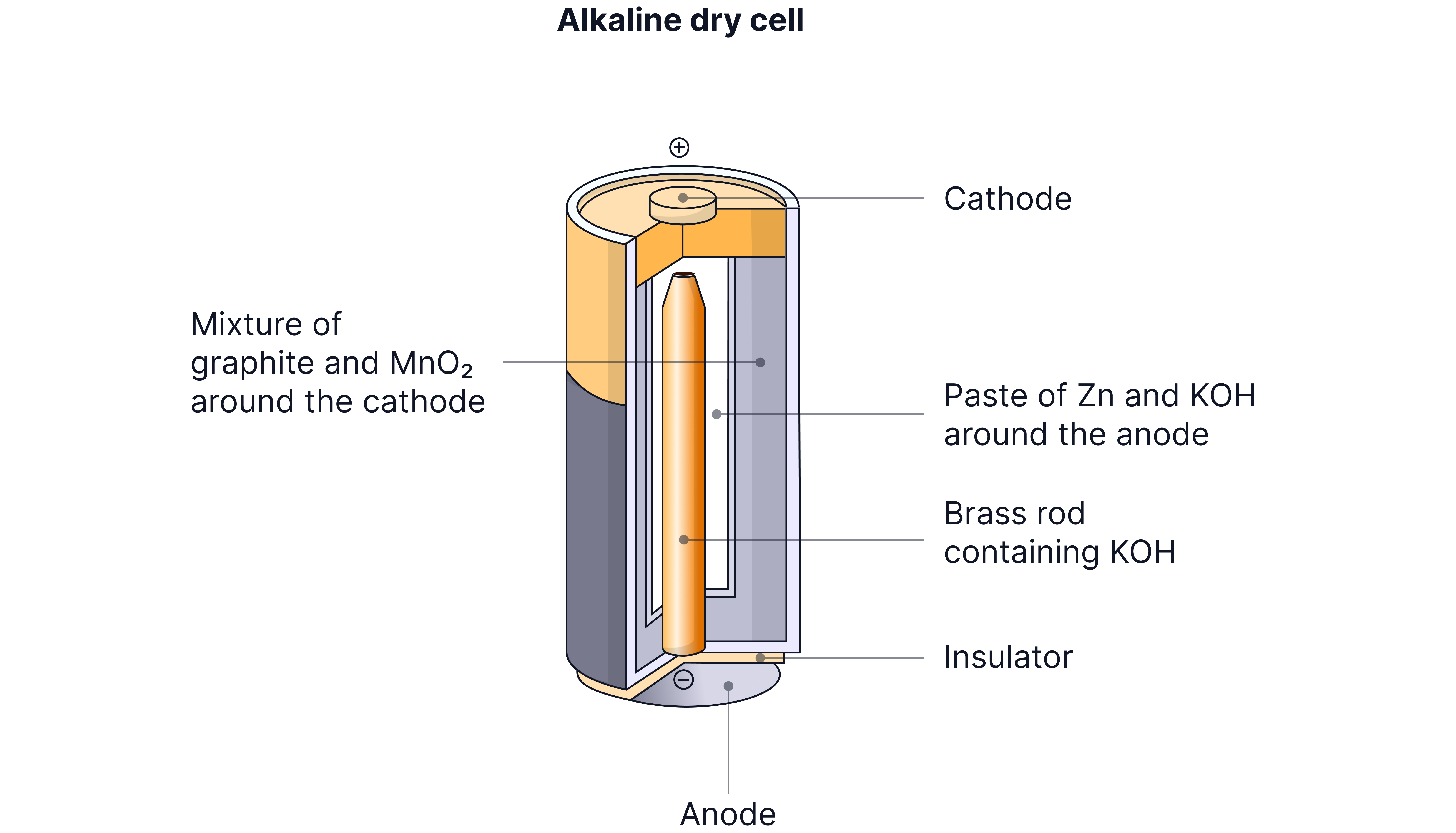
How it works:
Negative electrode: Zinc powder in alkaline solution
Positive electrode: Manganese dioxide
Half-reactions:
Anode: Zn + 2OH⁻ → ZnO + H₂O + 2e⁻ (E° = -1.22V)
Cathode: MnO₂ + H₂O + e⁻ → MnO(OH) + OH⁻ (E° = +0.15V)
Cell voltage: E°cell = (+0.15) - (-1.22) = 1.37V
Why alkaline is better than zinc-carbon:
Longer lasting - more energy stored
Better at low temperatures - works in winter
More stable voltage - doesn't drop as quickly during use
Less likely to leak
Pros: Long life, reliable, widely available
Cons: More expensive than zinc-carbon
When to Use Non-Rechargeable Cells
Best for:
Low-drain devices - clocks, remote controls, smoke detectors
Emergency equipment - flashlights, radios (long shelf life)
Devices used infrequently - don't need regular recharging
Backup power - when you need guaranteed power after long storage
Lithium Ion Batteries Explainer Video
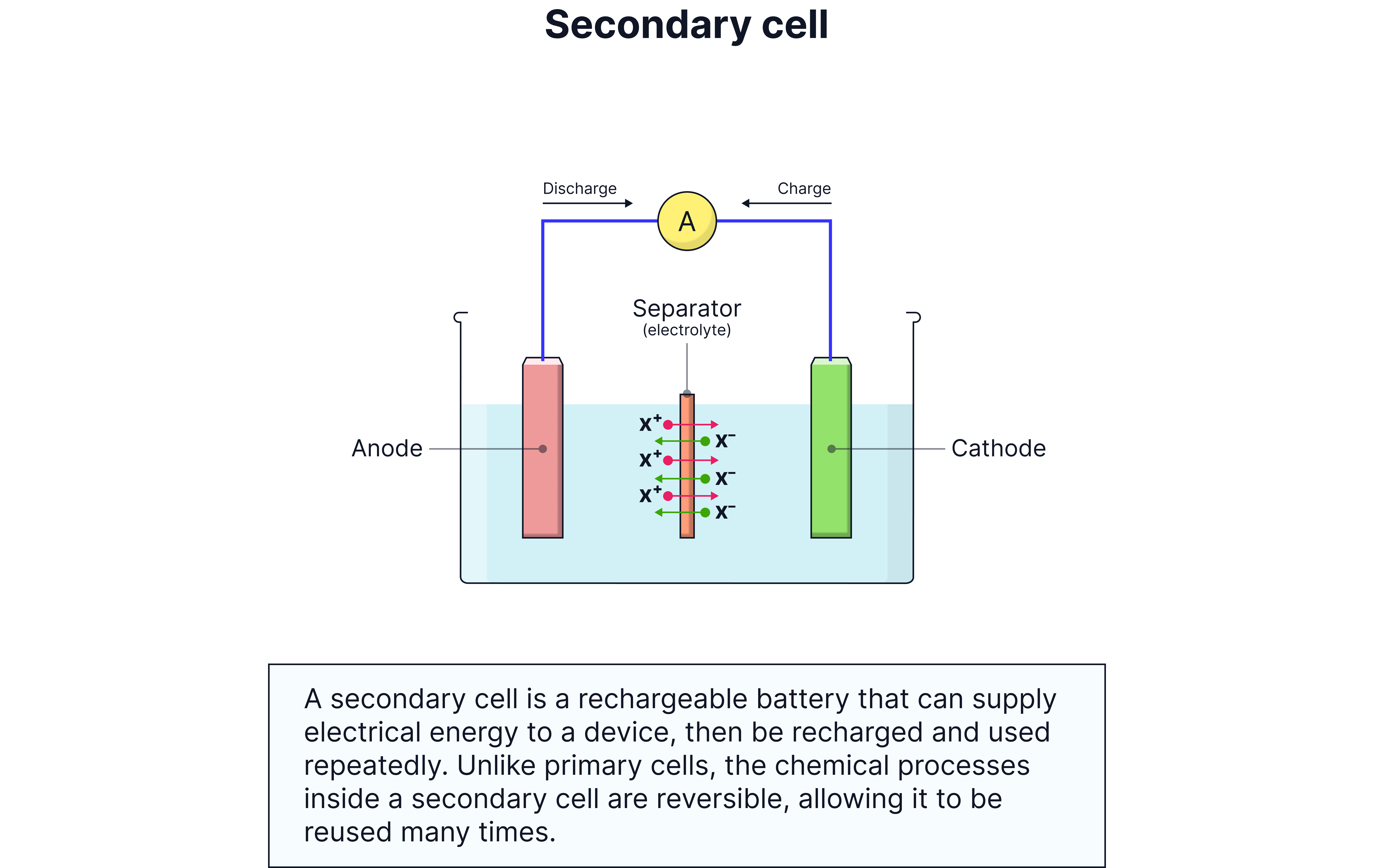
Rechargeable Cells (Secondary Cells)
How They Work
Rechargeable cells have reversible chemical reactions. You can reverse the discharge reaction by applying electrical energy (charging).
Key characteristics:
Reusable - can be charged and discharged hundreds of times
Reversible reactions - products stay near electrodes and can be converted back
Higher initial cost but cheaper long-term
Better for environment - less waste
Lead-Acid Battery
The car battery - powerful, reliable, and handles high currents.
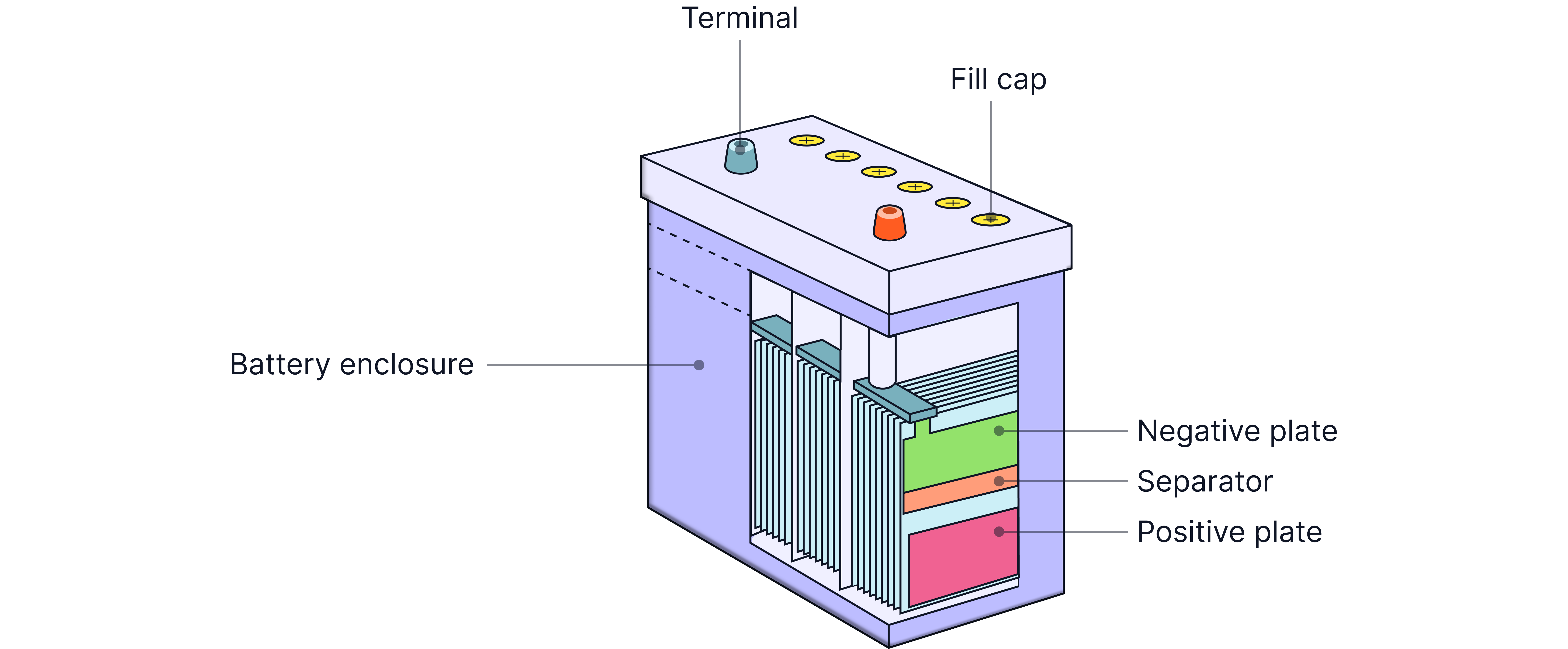
How it works:
Negative electrode: Spongy lead ()
Positive electrode: Lead dioxide ()
Electrolyte: Sulfuric acid ()
Half-reactions:
Anode:
Cathode:
Cell voltage:
Overall reactions:
Discharge:
Charge:
Why lead-acid for cars:
High current - can deliver hundreds of amps for starting
Reliable - works in hot and cold weather
Cheap - low cost for the amount of power
6 cells in series give
Pros: High power, reliable, cheap, well-recycled
Cons: Heavy, contains toxic lead, needs maintenance
Lithium-Ion Battery
The modern standard for phones, laptops, and electric cars.
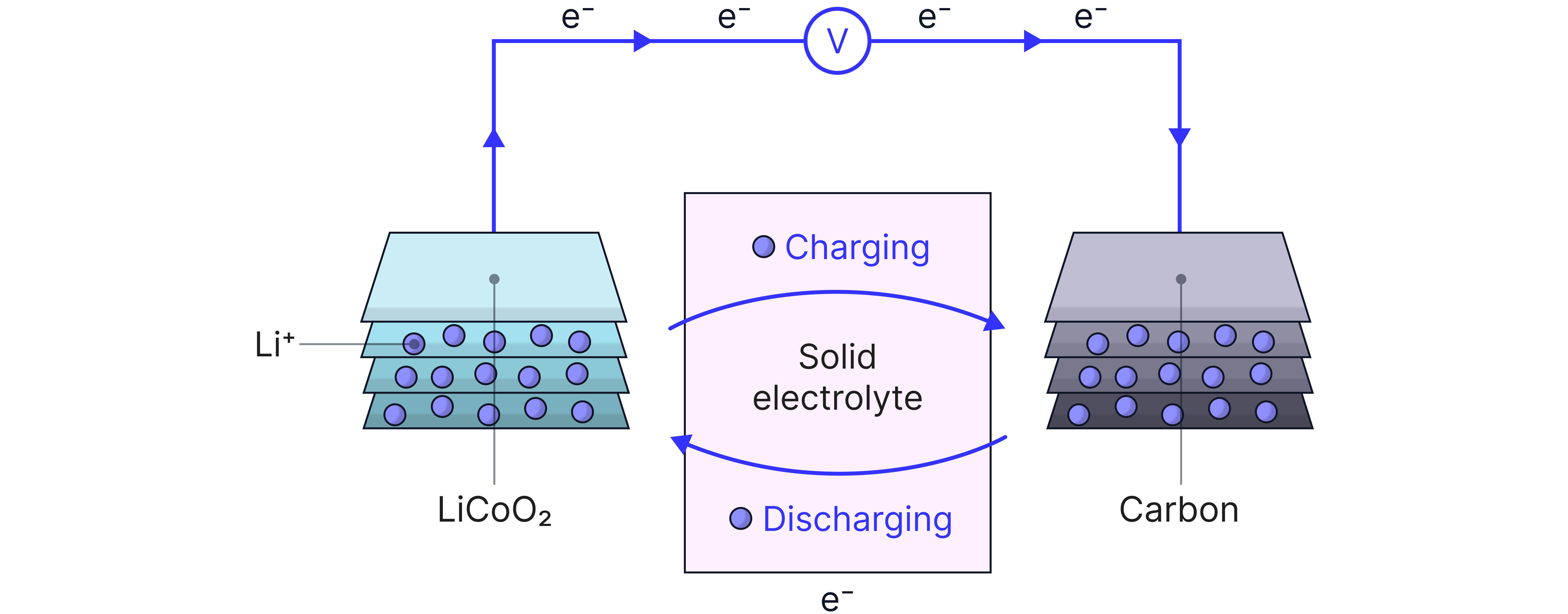
How it works:
Negative electrode: Graphite (stores lithium ions)
Positive electrode: Lithium metal oxide (like )
Lithium ions move between electrodes during charge/discharge
Half-reactions:
Anode:
Cathode:
Cell voltage: E
Overall Reactions:
Discharge:
Charge:
Why lithium-ion dominates:
High voltage - vs for older batteries
Light weight - high energy per gram
No memory effect - can charge anytime without losing capacity
Long lasting - hundreds of charge cycles
Low self-discharge - holds charge when not used
Pros: Lightweight, high energy, no memory effect, long life
Cons: Expensive, can be dangerous if damaged, needs protection circuits
Nickel-Cadmium (Ni-Cd) Battery
Older rechargeable technology - mostly replaced but still used in some applications.
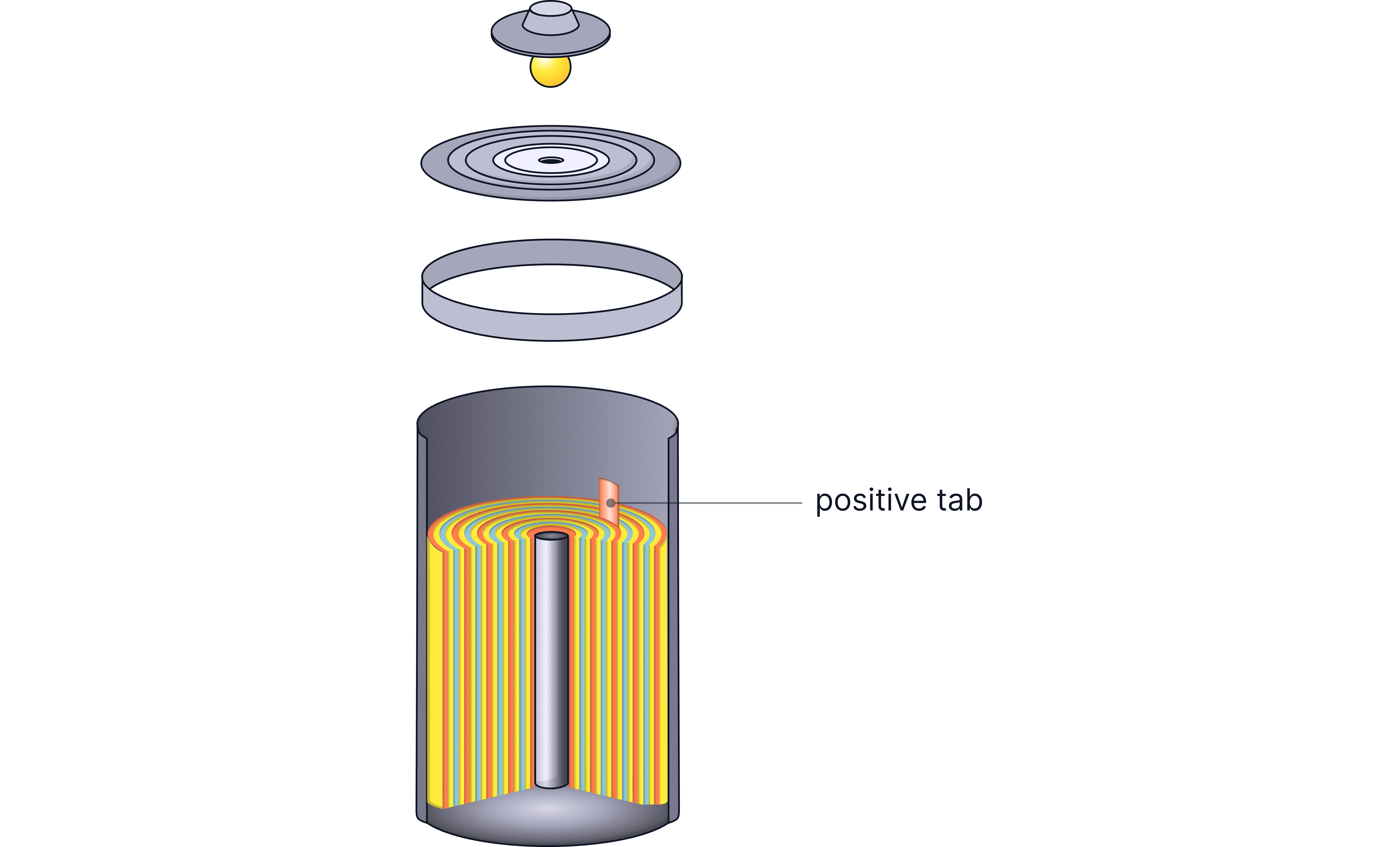
How it works:
Negative electrode: Cadmium
Positive electrode: Nickel oxyhydroxide
Half-reactions:
Anode:
Cathode:
Cell voltage:
Problems with Ni-Cd:
Memory effect - loses capacity if not fully discharged before recharging
Cadmium is toxic - environmental hazard
Self-discharge - loses charge when stored
Comparing Non-Rechargeable vs Rechargeable
Initial cost: Non-rechargeable cheaper
Long-term cost: Rechargeable much cheaper if used regularly
Example calculation:
Alkaline batteries: £2 each, last 20 hours = £0.10 per hour
Rechargeable AA: £8 each, last 500 cycles of 20 hours = £0.0008 per hour
Rechargeable is 125× cheaper per hour of use!
Environmental Impact
Non-rechargeable:
High waste - thrown away after single use
More materials needed - constant replacement
Disposal problems - millions end up in landfill
Rechargeable:
Less waste - one battery replaces hundreds of disposables
Better recycling - especially lead-acid (98% recycled)
Lower lifetime environmental impact