Fuel Cells
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
What Are Fuel Cells?
Fuel cells are fundamentally different from batteries. Instead of storing energy like rechargeable cells, they continuously convert fuel into electricity as long as fuel and oxidizer are supplied.
Key differences from batteries:
Continuous operation - don't need recharging, just refueling
Constant voltage - voltage stays steady as long as fuel flows
External reactants - fuel and oxidizer supplied from outside
Scalable - can be made tiny or huge depending on power needs
Fuel Cells Explainer Video
Acidic Hydrogen Fuel Cell
How It Works
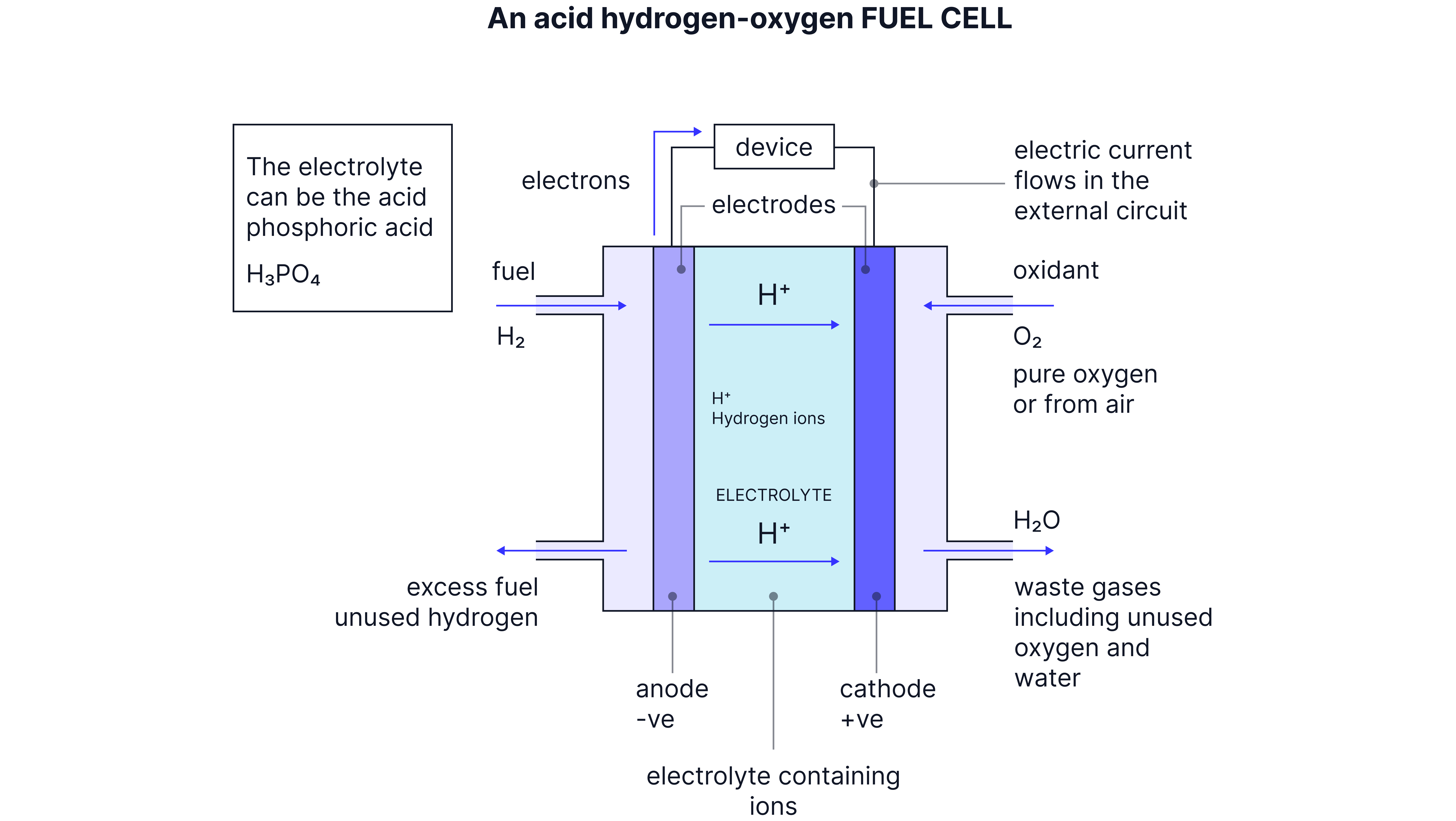
Electrode Reactions:
Anode (negative):
Cathode (positive):
Overall Reaction:
Cell EMF:
Alkaline Hydrogen Fuel Cell
How It Works
Uses potassium hydroxide (KOH) electrolyte instead of the polymer membrane.
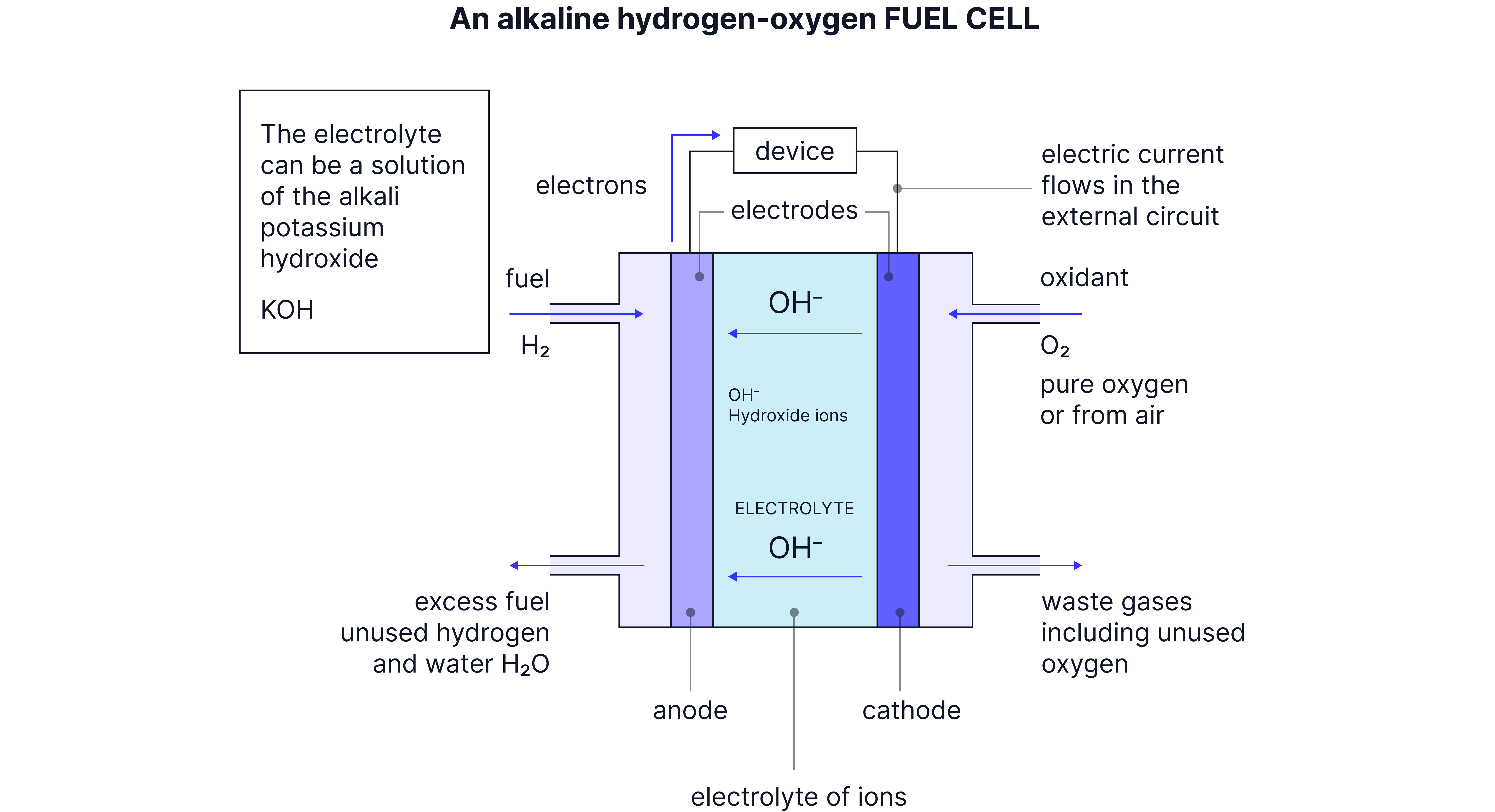
Electrode Reactions:
Anode (negative):
Cathode (positive):
Overall Reaction:
Cell EMF:
Key Differences from Acidic
Ion movement: ions move from cathode to anode (opposite direction to PEM)
Same overall reaction:
Same voltage: Both give 1.23V
Methanol Fuel Cell
Why Methanol?
Methanol is a liquid fuel that's much easier to store and handle than hydrogen gas.
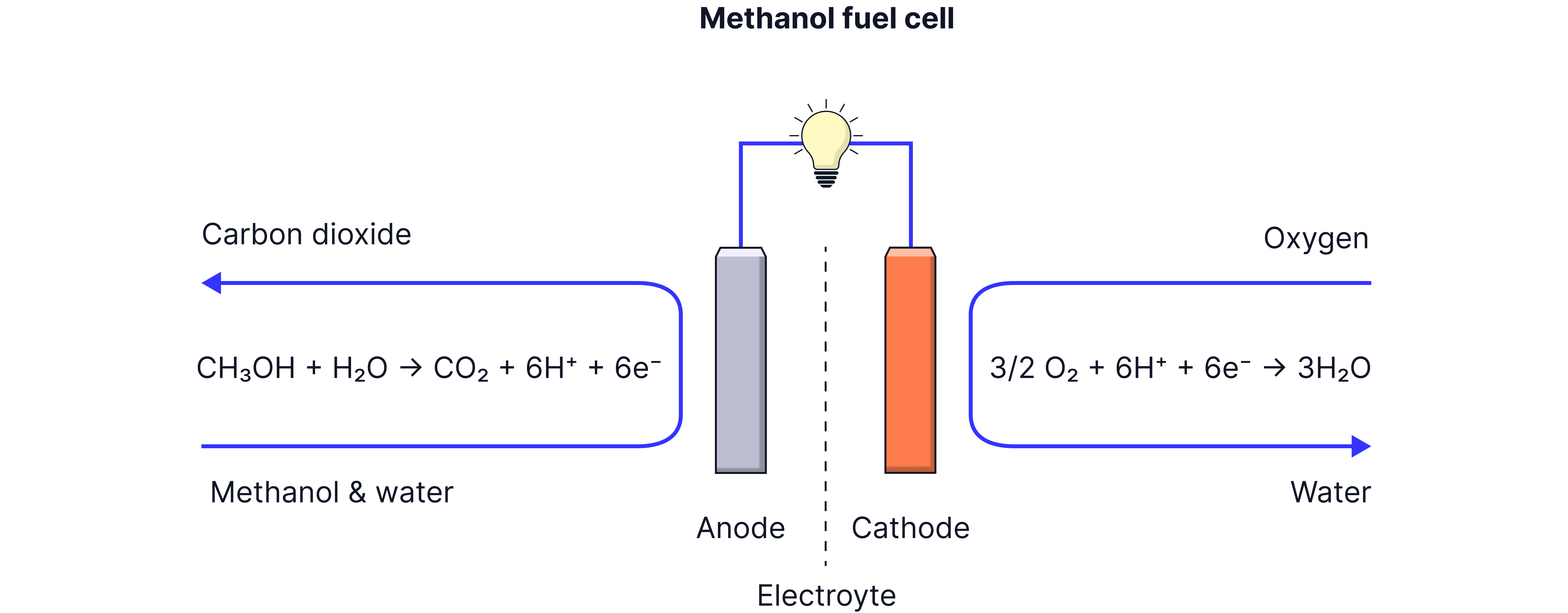
Electrode reactions:
Anode:
Cathode:
Overall reaction:
Cell EMF:
Advantages:
Liquid fuel - easy to store and transport
No hydrogen infrastructure needed
Higher energy density than hydrogen
Room temperature operation
Disadvantages:
emissions - not completely clean
Lower efficiency - slower reactions than hydrogen
Methanol toxicity - poisonous if ingested
Fuel crossover - methanol can pass through membrane
Comparison of Cell Types
Fuel Cells vs Traditional Cells: Benefits and Risks
Comparison Table
Aspect | Traditional Cells (Primary/Secondary) | Fuel Cells | Advantage |
|---|---|---|---|
Voltage Output | Voltage drops as reactants are consumed | Constant voltage as long as fuel is supplied | Fuel Cells |
Operation Time | Limited by stored chemicals | Continuous operation with fuel supply | Fuel Cells |
Refueling/Recharging | Replace (primary) or recharge for hours (secondary) | Refuel in minutes (no long recharging needed) | Fuel Cells |
Efficiency | 25–30% in applications | 40–60% efficiency | Fuel Cells |
Environmental Impact | Disposal issues, some toxic materials | Only water produced (H₂ fuel cells) | Fuel Cells |
Scalability | Limited by physical cell size | Easily scaled by stacking cells | Fuel Cells |
Initial Cost | Low to medium | Very high | Traditional Cells |
Fuel Storage | No external fuel needed | Complex/dangerous (H₂ pressurized) | Traditional Cells |
Infrastructure | Widely available | Limited (few H₂ stations) | Traditional Cells |
Safety | Generally safe | Hydrogen flammable/explosive | Traditional Cells |
Maintenance | Minimal | Complex systems require servicing | Traditional Cells |
Temperature Sensitivity | Some limitations | Water can freeze and damage cells | Traditional Cells |
Benefits of Fuel Cells
Performance Benefits
Constant voltage output - steady power as long as fuel flows
No downtime - continuous operation without recharging
High efficiency - direct chemical to electrical conversion
Quiet operation - no moving parts or combustion
Environmental Benefits
Clean emissions - only water from hydrogen fuel cells
No disposal issues - no toxic battery waste
Renewable potential - can use green hydrogen from renewable energy
Risks and Disadvantages of Fuel Cells
Safety Concerns
Hydrogen is highly flammable and explosive
High pressure storage (up to 700 bar) creates rupture risks
Specialized safety training required for handling
Infrastructure Limitations
Limited refueling stations - few hydrogen filling points
High infrastructure costs - expensive to build H₂ network
Transport challenges - difficult to move hydrogen safely
Technical Issues
Complex systems - pumps, sensors, controls can fail
Cold weather problems - water can freeze in cells
Catalyst degradation