Electron Configuration
Lajoy Tucker
Teacher

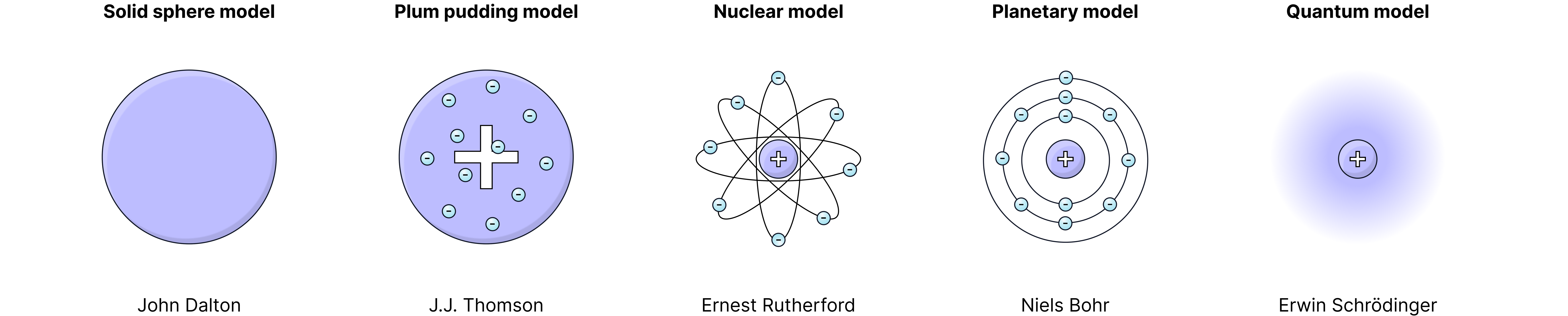
Models of the Atom
Bohr model (GCSE): electrons in fixed shells, circular orbits around the nucleus.
Quantum model (A level): shells contain subshells (s, p, d, f), which are made of orbitals.
Orbitals are regions where there is a high probability of finding an electron.
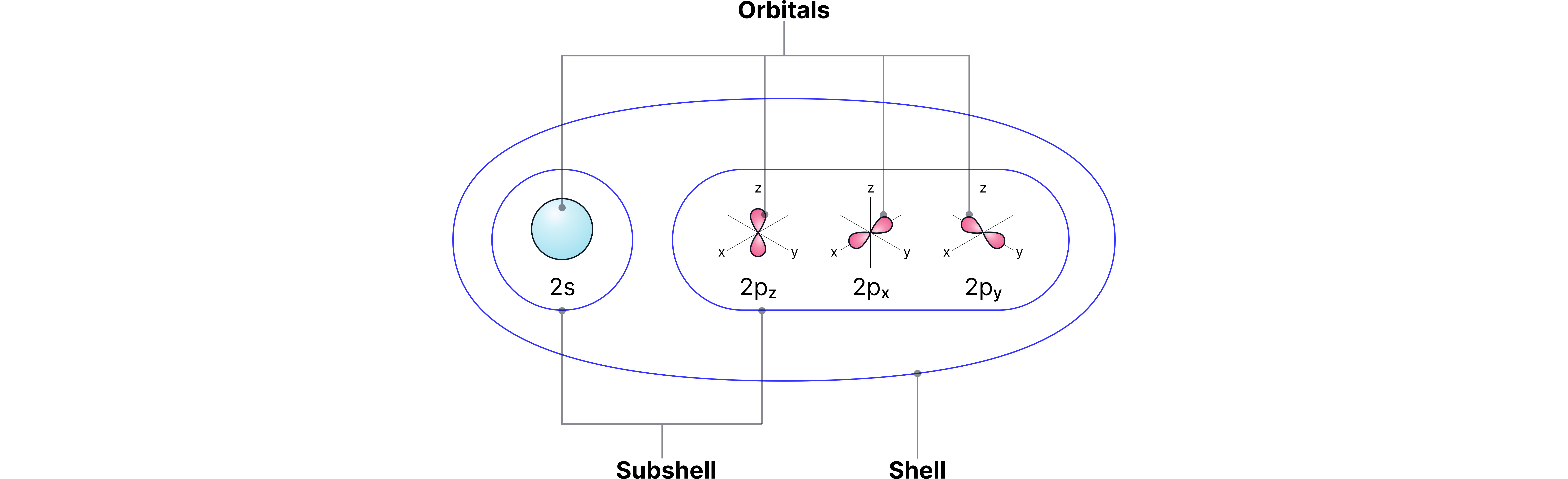
Shells and Subshells
Shells (principal energy levels): labelled n = 1, 2, 3, 4 …
Subshells: each shell is split into subshells labelled s, p, d, f.
Capacity of subshells:
s: 1 orbital, 2 electrons
p: 3 orbitals, 6 electrons
d: 5 orbitals, 10 electrons
f: 7 orbitals, 14 electrons
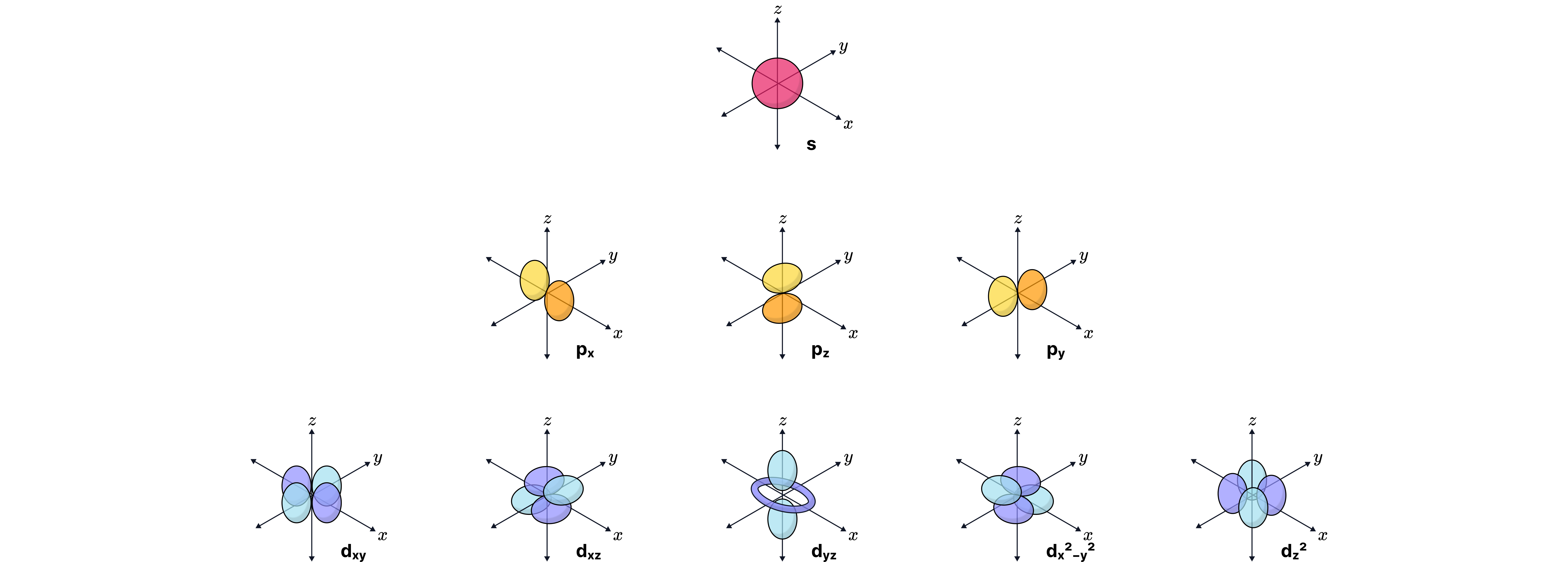
Orbitals
An orbital is a region that can hold up to 2 electrons with opposite spins.
Shapes:
s = spherical
p = dumbbell (px, py, pz)
You need to know the shape of the s and p-orbitals and be able to draw them. You do not need to be able to draw the shape of the d-orbitals.
Rules for Filling Orbitals
Aufbau principle: fill from lowest energy upwards (1s → 2s → 2p → 3s …).
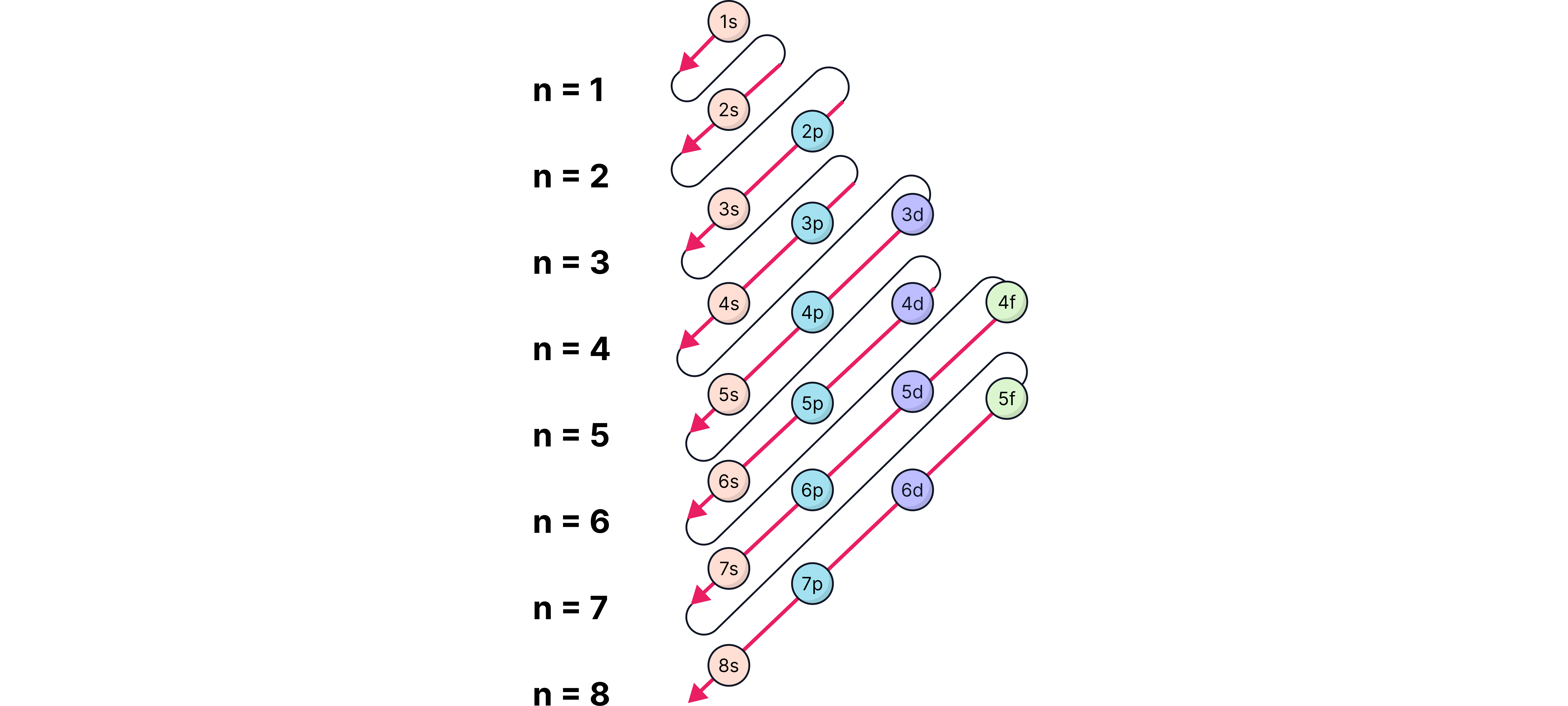
Hund’s rule: orbitals in the same subshell are filled singly before pairing.
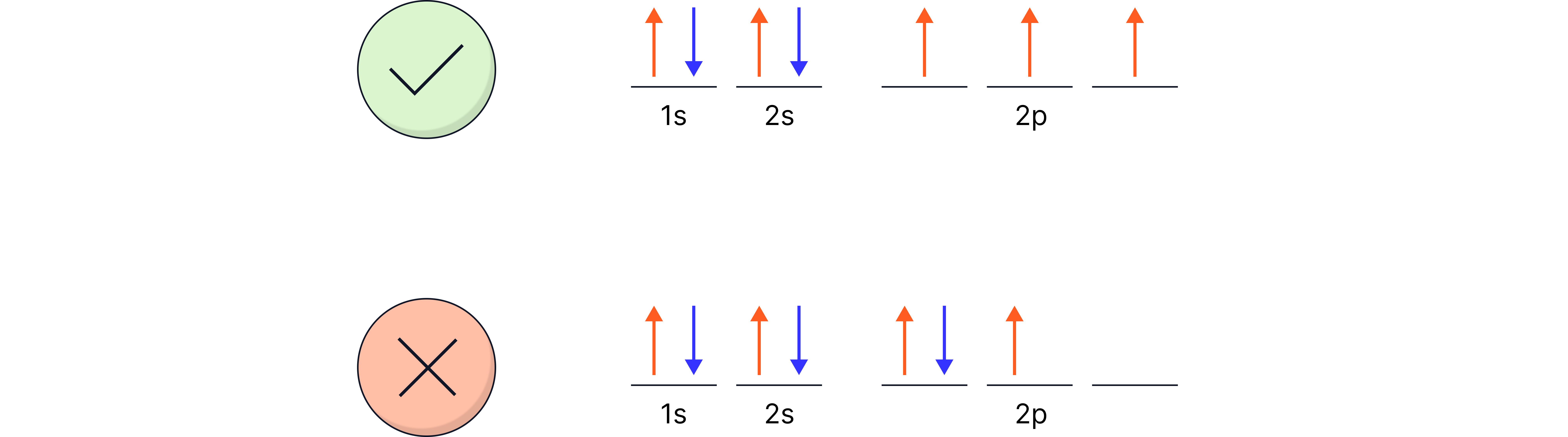
Pauli exclusion principle: no two electrons in the same orbital can have the same spin.
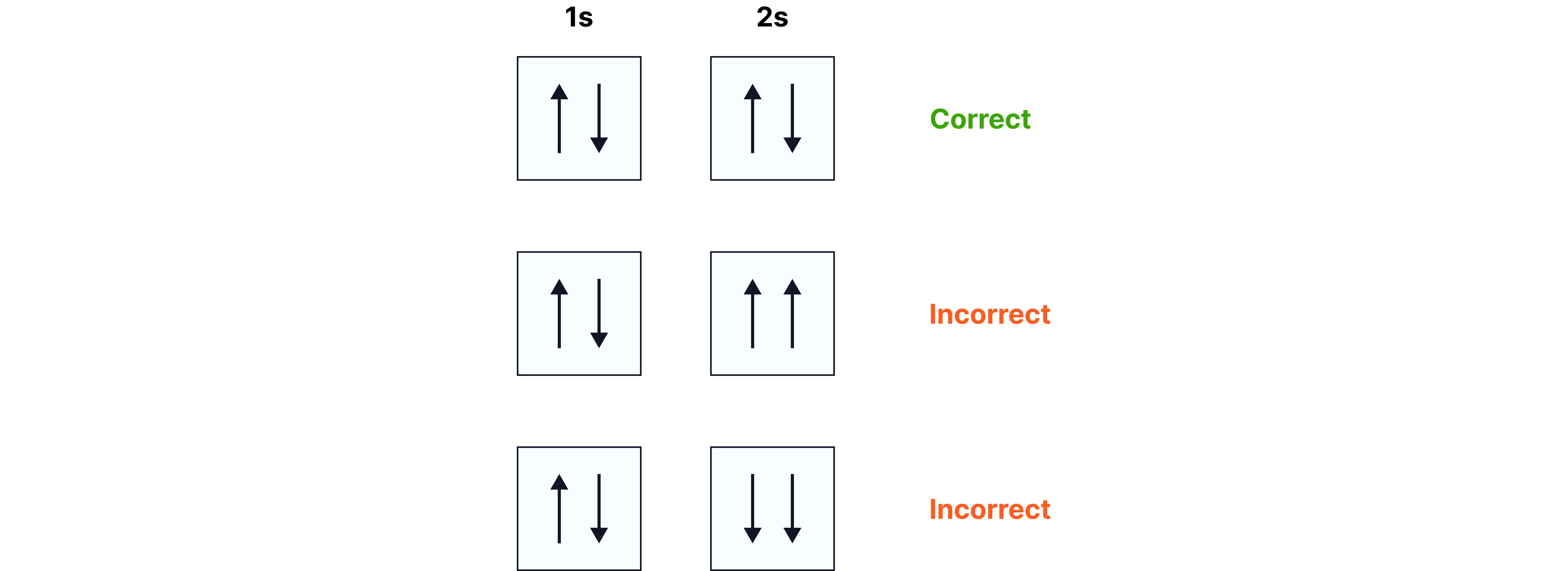
Writing Electron Configurations
Full notation: list every orbital. Sodium for example has the electron configuration:
Na (Z = 11): 1s² 2s² 2p⁶ 3s¹
This is read as “one s two” and not 1s squared etc.
The first number denotes the number of the main energy level/principal quantum number. The letter denotes the type of sub-shell. And the superscript number denotes the number of electrons in that sub-shell.
Shorthand notation: use previous noble gas in square brackets.
Examples:
O (Z = 8): 1s² 2s² 2p⁴ or [He] 2s2 2p4 where [He] has the electron configuration of 1s2
Ca (Z = 20): [Ar] 4s²
Sc (Z = 21): [Ar] 4s² 3d¹
Electron Configurations for Ions
Positive ions (cations): electrons are lost from the outermost shell first.
Example: Mg (Z = 12): [Ne] 3s² → Mg²⁺ = [Ne]
Negative ions (anions): electrons are gained into the next available orbital.
Example: O (Z = 8): [He] 2s² 2p⁴ → O²⁻ = [Ne]
The Periodic Table Blocks
s-block: outer electron in s subshell (Groups 1–2).
p-block: outer electron in p subshell (Groups 13–18).
d-block: transition metals, filling d subshell.
d-block in more detail
1. Which fills first – 4s or 3d?
The 4s orbital has slightly lower energy than 3d when both are empty.
Therefore, 4s fills before 3d.
Example: Sc (Z = 21): [Ar] 4s² 3d¹
2. What happens on ionisation?
Once both 4s and 3d contain electrons, the energy of 4s is slightly higher than 3d.
This means 4s electrons are lost first during ionisation.
Example: Fe (Z = 26):
Neutral atom: [Ar] 4s² 3d⁶
Ion: Fe²⁺ [Ar] 3d⁶ (lost the 4s electrons)
3. The Anomalies – Chromium and Copper
Cr (Z = 24): expected [Ar] 4s² 3d⁴, but actual [Ar] 4s¹ 3d⁵
– Reason: half-filled d⁵ subshell is more stable (lower repulsion, symmetrical distribution).
Cu (Z = 29): expected [Ar] 4s² 3d⁹, but actual [Ar] 4s¹ 3d¹⁰
– Reason: fully filled d¹⁰ subshell is extra stable.
Summary of d-block electron configurations
Element | Neutral Atom Configuration | Common Ion Configurations | Notes |
Sc (21) | [Ar] 4s² 3d¹ | Sc³⁺: [Ar] | 4s² and 3d¹ all lost → stable Ar core |
Ti (22) | [Ar] 4s² 3d² | Ti²⁺: [Ar] 3d²Ti³⁺: [Ar] 3d¹ | 4s electrons lost first |
V (23) | [Ar] 4s² 3d³ | V²⁺: [Ar] 3d³V³⁺: [Ar] 3d² | Multiple oxidation states |
Cr (24) | [Ar] 4s¹ 3d⁵ (anomaly) | Cr²⁺: [Ar] 3d⁴ Cr³⁺: [Ar] 3d³ | Half-filled 3d subshell more stable |
Mn (25) | [Ar] 4s² 3d⁵ | Mn²⁺: [Ar] 3d⁵ Mn³⁺: [Ar] 3d⁴ | Mn²⁺ is especially stable (half-filled d) |
Fe (26) | [Ar] 4s² 3d⁶ | Fe²⁺: [Ar] 3d⁶ Fe³⁺: [Ar] 3d⁵ | Fe³⁺ stable due to half-filled d |
Co (27) | [Ar] 4s² 3d⁷ | Co²⁺: [Ar] 3d⁷ Co³⁺: [Ar] 3d⁶ | |
Ni (28) | [Ar] 4s² 3d⁸ | Ni²⁺: [Ar] 3d⁸ | Ni²⁺ is common in complexes |
Cu (29) | [Ar] 4s¹ 3d¹⁰ (anomaly) | Cu⁺: [Ar] 3d¹⁰ Cu²⁺: [Ar] 3d⁹ | Full 3d subshell stabilises Cu⁺ |
Zn (30) | [Ar] 4s² 3d¹⁰ | Zn²⁺: [Ar] 3d¹⁰ | d¹⁰ full shell makes Zn²⁺ very stable |