Ionisation Energies
Lajoy Tucker
Teacher

Contents
Definition
The first ionisation energy is the enthalpy change when one mole of gaseous atoms loses one electron per atom to form one mole of gaseous ions with a single positive charge.
Equation (general form):
X(g) → X⁺(g) + e⁻
Always defined in the gaseous state.
Units: kJ mol⁻¹.
The attraction between the nucleus and an electron can be described using
Coulomb’s Law:
F ∝ 1 / r²
Where:
F = attractive
r = distance between nucleus and electron.
Factors Affecting Ionisation Energy
1. Nuclear Charge
More protons = stronger positive nuclear charge.
Worked Example 1:
Compare H (Z = 1) and He (Z = 2).
Both outer electrons are in the 1s orbital, so similar distance (r) and shielding.
He has double the number of protons than H.
So the attraction between the electrons and nucleus in He is stronger than that of H
Consequence: helium has a much higher ionisation energy than hydrogen.
2. Distance from the Nucleus (r in the denominator)
Electrons further away from the nucleus experience less attraction.
Because F ∝ 1/r², even a small increase in r causes a big decrease in attraction.
Worked Example 2:
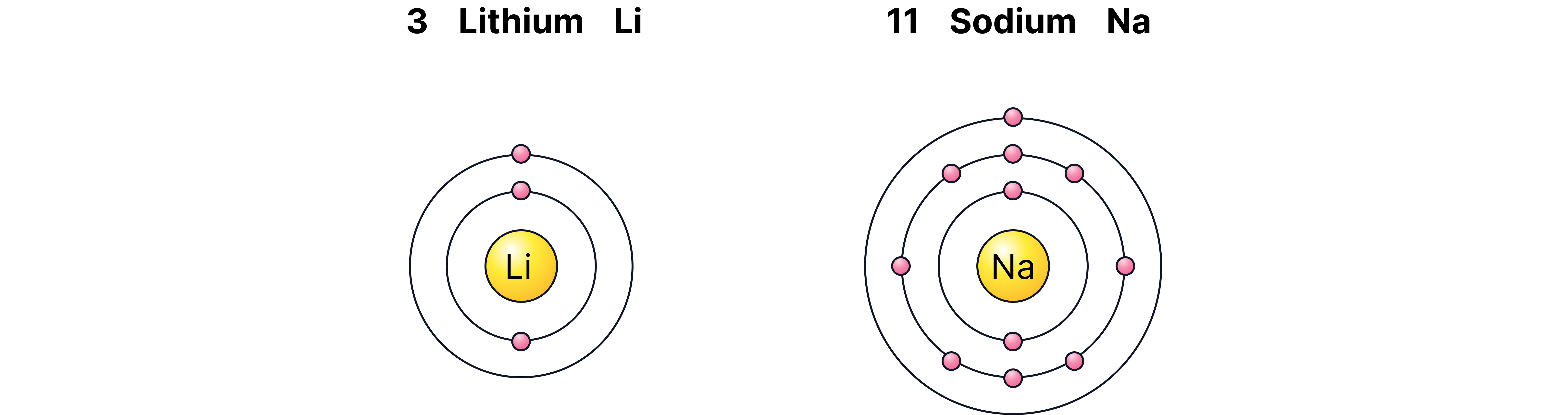
Compare Li (2,1) and Na (2,8,1).
Both have outer electrons in an s orbital, but Li’s is 2s, Na’s is 3s (further from nucleus).
Despite Na having more protons, the increased r and shielding weaken the attraction.
Consequence: Na has a lower ionisation energy than Li.
3. Shielding (Effective Nuclear Charge)
Inner shells repel outer electrons, reducing the attraction from the nucleus.
Coulomb’s Law still applies, but the effective nuclear charge is less than the actual nuclear charge (number of protons).
Worked Example 3:
Compare Li (1s² 2s¹) and K
In potassium, increase in proton number (nuclear charge) is offset by the increased distance of the outer electron to the nucleus
And the outer electron is more shielded by the core electrons
Resulting in a weaker force of attraction between the valence electron and positive nucleus
So less energy is required to remove it so a lower ionisation energy
4. Electron Repulsion in Orbitals
Two electrons sharing the same orbital repel each other.
Repulsion makes it easier to remove one electron.
Worked Example 4:
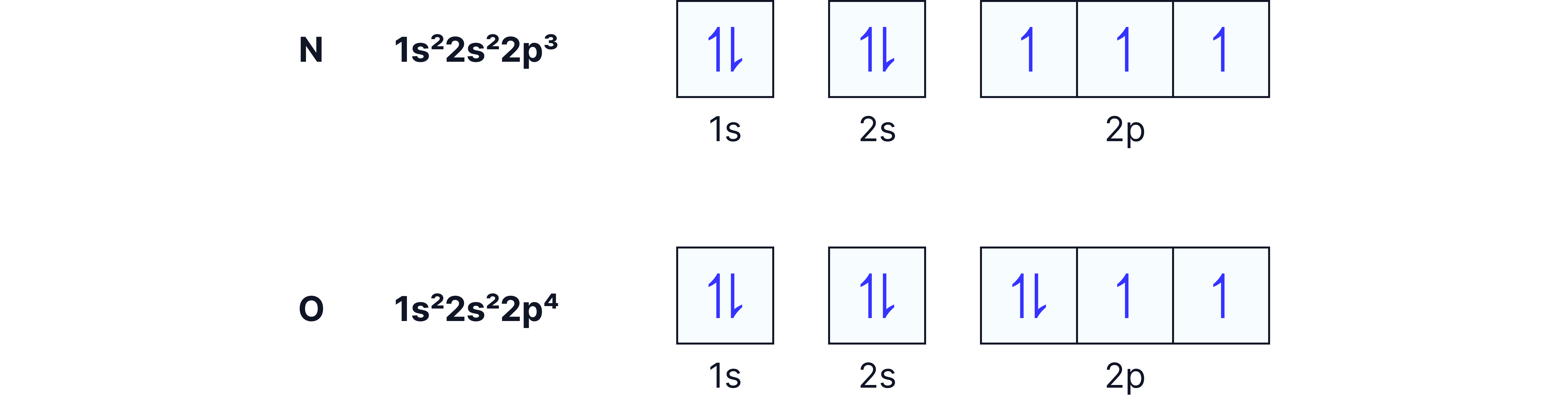
N (1s² 2s² 2p³): three p orbitals singly occupied.
O (1s² 2s² 2p⁴): one orbital now has a pair, causing repulsion.
Consequence: oxygen has a lower ionisation energy than nitrogen.
General Trends
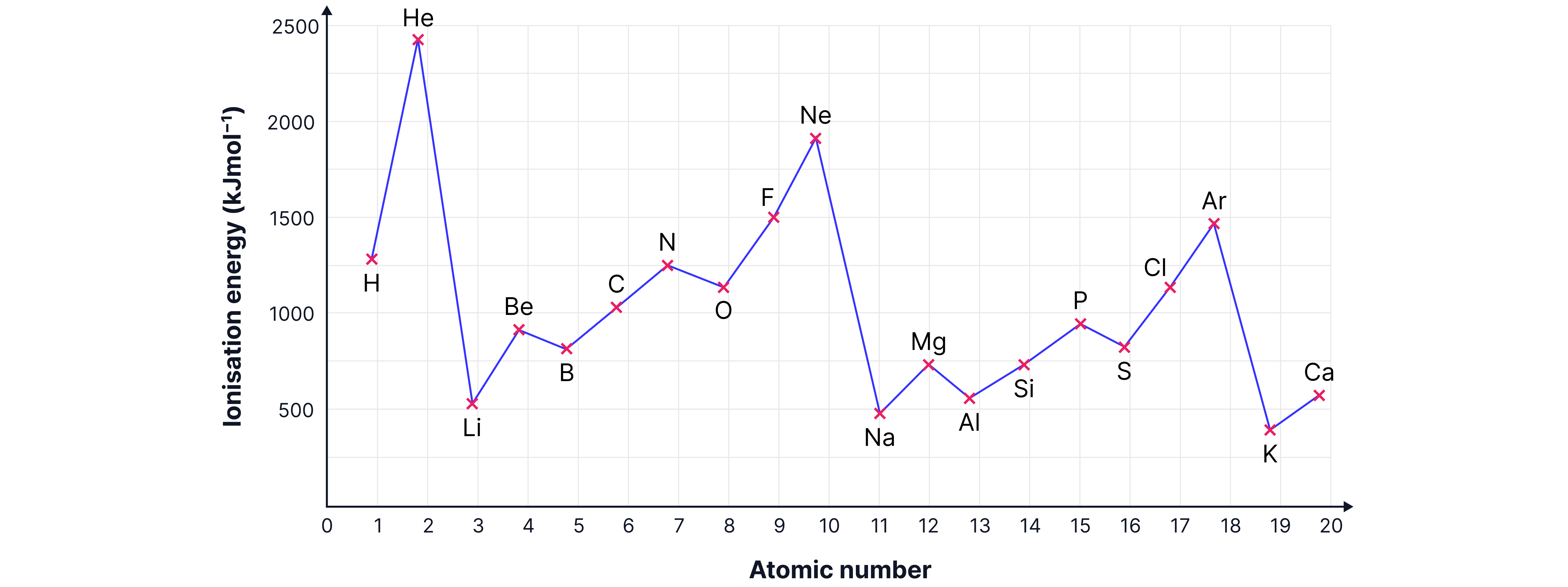
Across a Period
Ionisation energy generally increases.
More protons
Outer electrons in the same shell (r roughly constant, shielding unchanged).
Consequence: stronger nuclear attraction → more energy required to remove electrons.
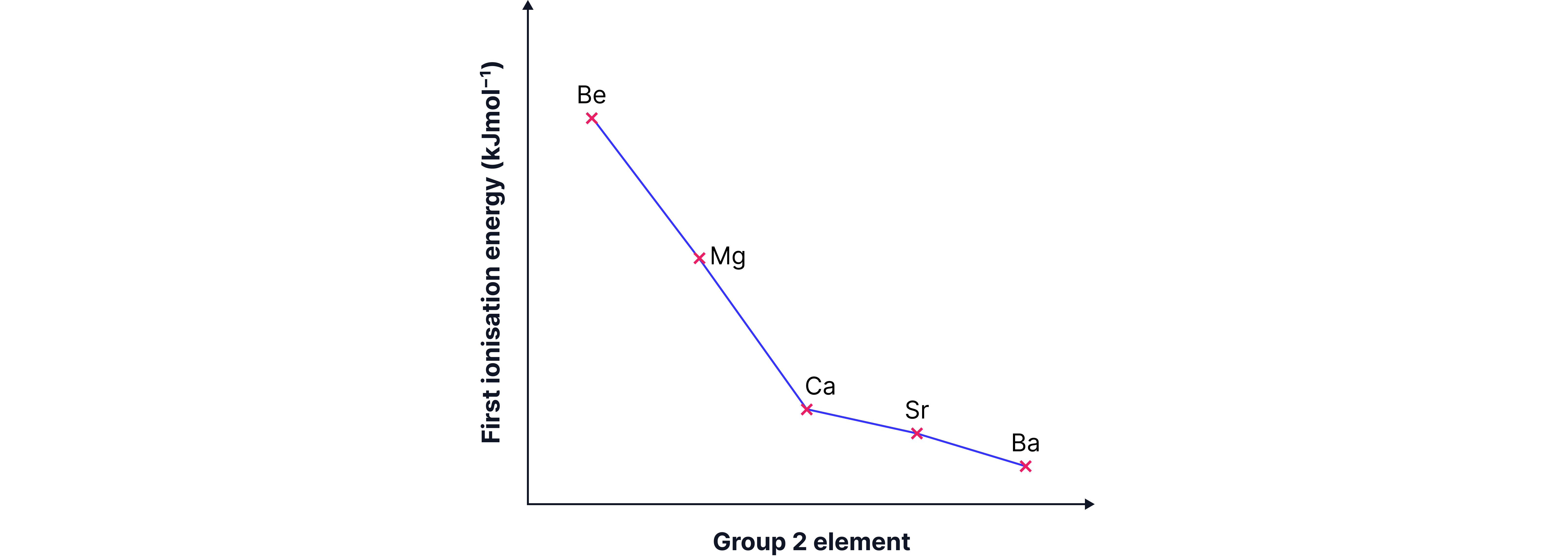
Down a Group
Ionisation energy decreases.
Electrons are further from nucleus (r increases).
More shielding from inner shells.
Although nuclear charge increases, distance and shielding dominate.
Key Anomalies
1. Be → B (Mg → Al)
B’s outer electron enters a new p orbital (higher in energy, further from nucleus).
2. N → O (P → S)
O’s p orbital now has a paired electron.
Extra electron–electron repulsion lowers ionisation energy.
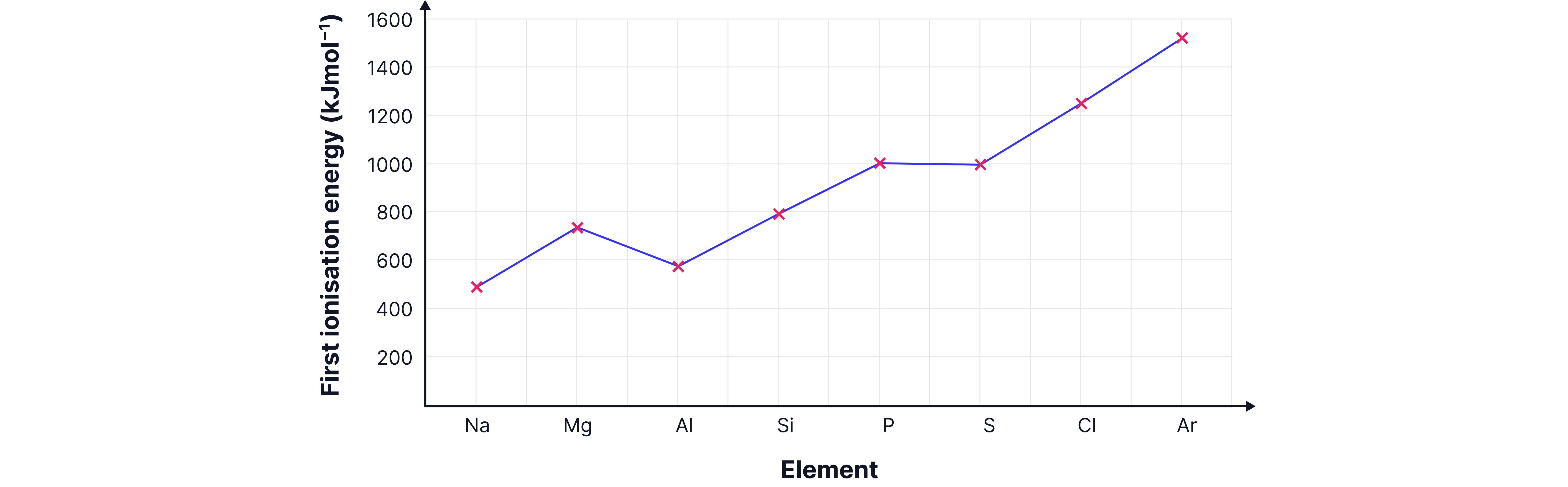
Worked Example
Question: Why does Na have a much lower first ionisation energy than Ne?
Step 1 – Compare shells
Ne: outer electrons in 2p (close to nucleus).
Na: outer electron in 3s (further away).
Step 2 – Consider shielding
Na’s outer electron shielded by 2 inner shells.
Ne’s only shielded by 1 inner shell.
Final Answer: Na’s electron is further away from the positive nucleus and more shielded by the core electrons, so easier to remove.
Explain why the second ionisation energy of calcium is greater than the first.
The first ionisation energy of calcium involves removing an electron from a neutral atom
(Ca → Ca⁺ + e⁻).
The second ionisation energy involves removing an electron from a Ca⁺ ion
(Ca⁺ → Ca²⁺ + e⁻).
The Ca⁺ ion has a higher effective nuclear charge and a smaller radius, compared to the neutral atom, making the second electron harder to remove.
This means that the second ionisation energy of calcium will be higher as it is harder to remove an electron from an already positive ion than it is to remove an electron from a neutral atom
Successive Ionisation Energies
What does “successive” mean?
Successive means one after another, in sequence.
Successive ionisation energies are the series of values you get if you keep removing electrons from the same atom/ion, step by step.
Example:
First IE (IE₁): X(g) → X⁺(g) + e⁻
Second IE (IE₂): X⁺(g) → X²⁺(g) + e⁻
Third IE (IE₃): X²⁺(g) → X³⁺(g) + e⁻
…and so on. Each one is harder, because the ion left behind is more positively charged and holds on more strongly to its remaining electrons.
Why do successive ionisation energies increase?
Nuclear charge stays the same – same number of protons. But each electron is harder to remove, because the ion left behind is more positively charged and holds on more strongly to the remaining electrons. The result is that each successive IE is bigger than the last.
Using Successive Ionisation Energies to Find Group Number
The key idea:
Small increases between successive values = removing electrons from the same shell.
One very large jump = you’ve moved into a new, inner shell (closer to nucleus, much stronger attraction).
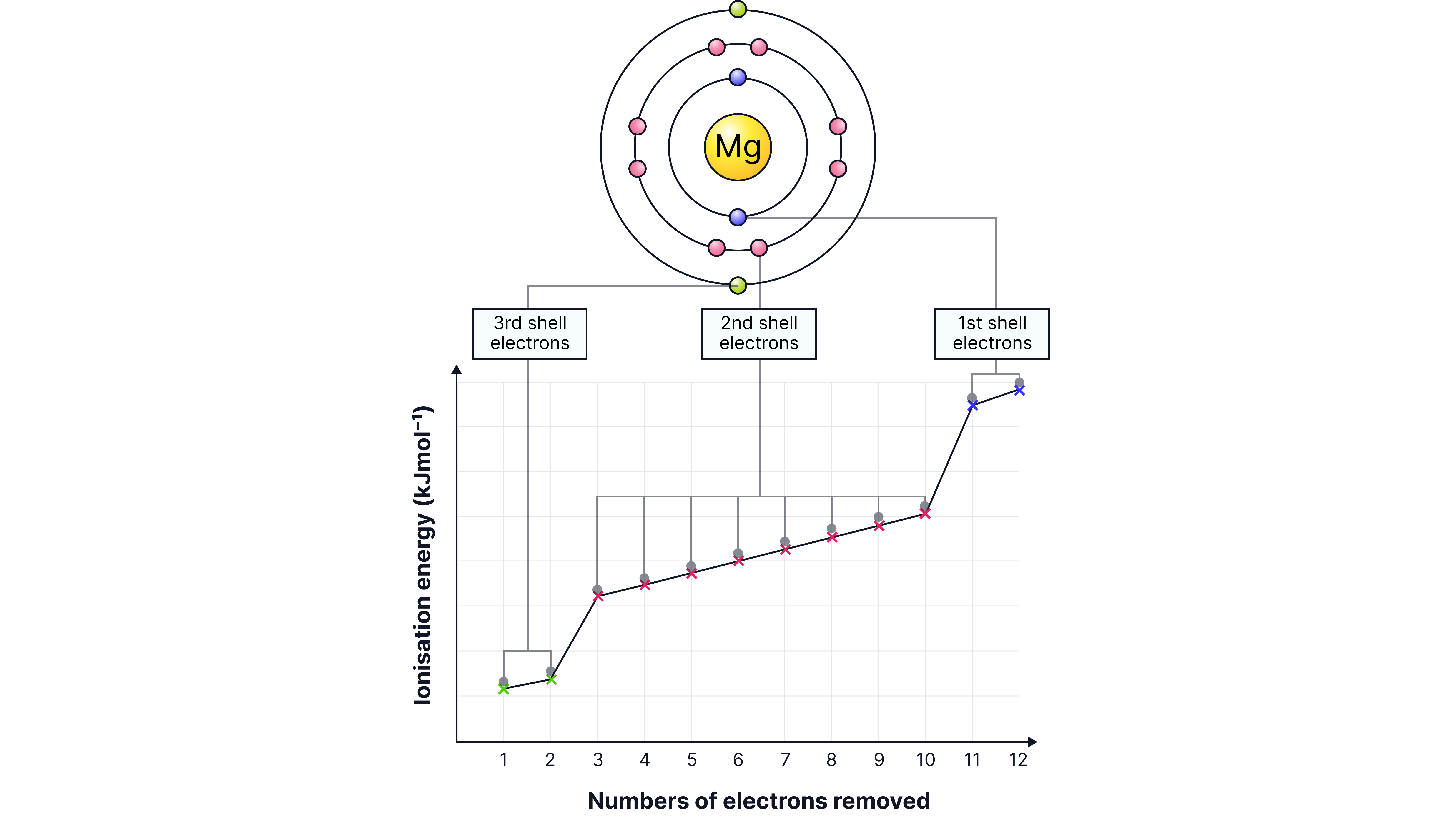
Worked Example 1 (Magnitude of Jump)
Ionisation energies (kJ mol⁻¹):
IE₁ = 600, IE₂ = 1200, IE₃ = 5000, IE₄ = 7000, IE₅ = 9000
Between IE₁ and IE₂ → energy roughly doubles (small increase, same shell).
Between IE₂ and IE₃ → energy suddenly jumps by more than 4×.
Interpretation → first two electrons were in outer shell, next electron comes from inner shell.
Conclusion: Element is in Group 2 (two outer electrons).
Worked Example 2 (Orders of Magnitude)
Ionisation energies (kJ mol⁻¹):
IE₁ = 800, IE₂ = 1600, IE₃ = 2500, IE₄ = 11000, IE₅ = 15000…
IE₁ → IE₂ → IE₃ → gradual increases (outer shell).
IE₃ → IE₄ → sudden jump of more than 4× (order of magnitude).
Big jump after the 3rd IE → 3 outer electrons.
Conclusion: Element is in Group 13 (3 outer electrons).
How to Spot the “Big Jump” Step by Step
1. Look down the list of IEs.
2. Compare each increase – is it gradual (same shell) or sudden, huge (new shell)?
3. The number of electrons before the jump = group number.
Question 1
Explain why the jump in IE values is much larger when moving from outer shell to inner shell.
Answer:
Inner shell electrons are closer to nucleus (r smaller), less shielded, and therefore feel a much stronger attraction. As the distance decreases → F increases dramatically.
Question 2
An element has IEs (kJ mol⁻¹): 1000, 2200, 3400, 4500, 5900, 7000, 8500, 10600, 13300, 33000.
Answer:
The first 8 increases are fairly small.
Huge jump after the 8th → means 8 outer electrons.
Conclusion: Group 18 element.
Worked Examples
Question 1
Why is the first ionisation energy of Sulfur lower than that of Phosphorus?
Answer:
Sulfur’s 3p⁴ configuration has an electron pair in one of its 3p orbitals.
Electron repulsion between these paired electrons lowers the energy needed to remove one.
Phosphorus 3p³ configuration has no paired electrons, so its ionisation energy is higher than sulfur’s.
Question 2
Explain why the second ionisation energy of sodium is much higher than the first.
Answer:
First IE: Na loses its outermost 3s electron.
Second IE: The next electron must be removed from the full 2p orbital (much closer to the nucleus, with stronger attraction).
This requires significantly more energy relative to the 1st IE.
Practice Questions
Question 1
MCQ: Which element has the highest first ionisation energy?
a) Sodium (Na)
b) Magnesium (Mg)
c) Argon (Ar)
d) Aluminium (Al)
Answer:
c) Argon Highest nuclear charge, smallest atomic radius, full outer shell, very stable so it requires a lot of energy to remove an electron.
Question 2
Short Answer: Explain why the first ionisation energy decreases down Group 2.
Answer:
Increasing atomic radius → Outer electron further from nucleus, decreasing electrostatic attraction to the nucleus.
Increased shielding → Increasing number of shells down the group, increasing the shielding by electrons in inner shells which reduces nuclear attraction.
Despite increased nuclear charge, these two factors outweigh it, making it easier to remove an electron.