Elimination of Halogenoalkanes
Lajoy Tucker
Teacher

Contents
Elimination with alcoholic hydroxide ions
Change in functional group:
Reagents:
Potassium or sodium hydroxide
Conditions:
Dissolved in ethanol, heat under reflux
Mechanism:
Base-induced elimination (E1 or E2 depending on the conditions)
Type of reagent:
Hydroxide ion () acting as a base when dissolved in ethanol.
Key concept:
Elimination is the removal of a small molecule (usually or ) to form a double bond – resulting in the formation of an alkene.
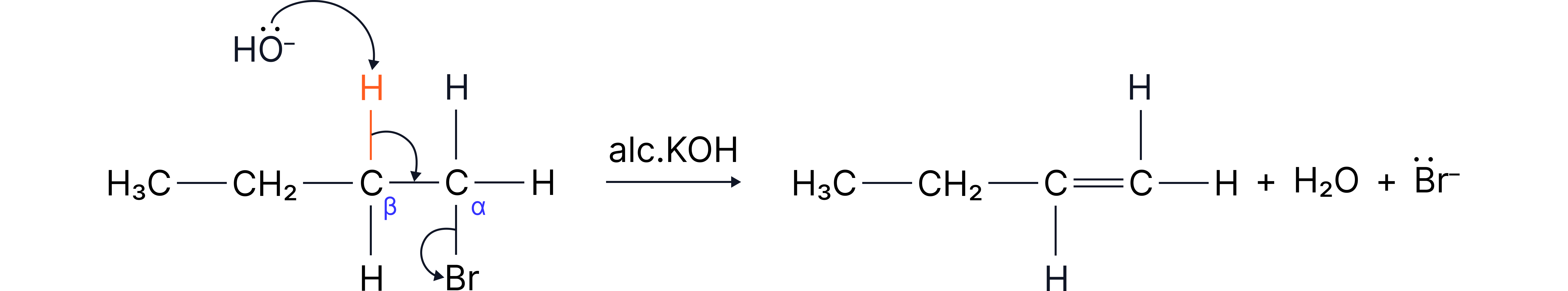
Example reaction
In this reaction:
The ion removes a hydrogen from a (a carbon next to the one bonded to the halogen).
A double bond forms between the and .
The halide ion () leaves.
Definitions:
– The carbon directly bonded to the halogen.
– The carbon(s) adjacent to the, where the is removed from.
Mechanism (summary):
Curly arrow from to form
Curly arrow from to halide ion
Base removes
Solvent makes a difference
Aqueous (water as solvent):
Favoured pathway: Substitution
Typical transformation:
Alcoholic (ethanol as solvent):
Favoured pathway: Elimination
Typical transformation:
Exam tip:
Mention the solvent every time – it directly affects the reaction mechanism.
Structural variation → multiple alkenes
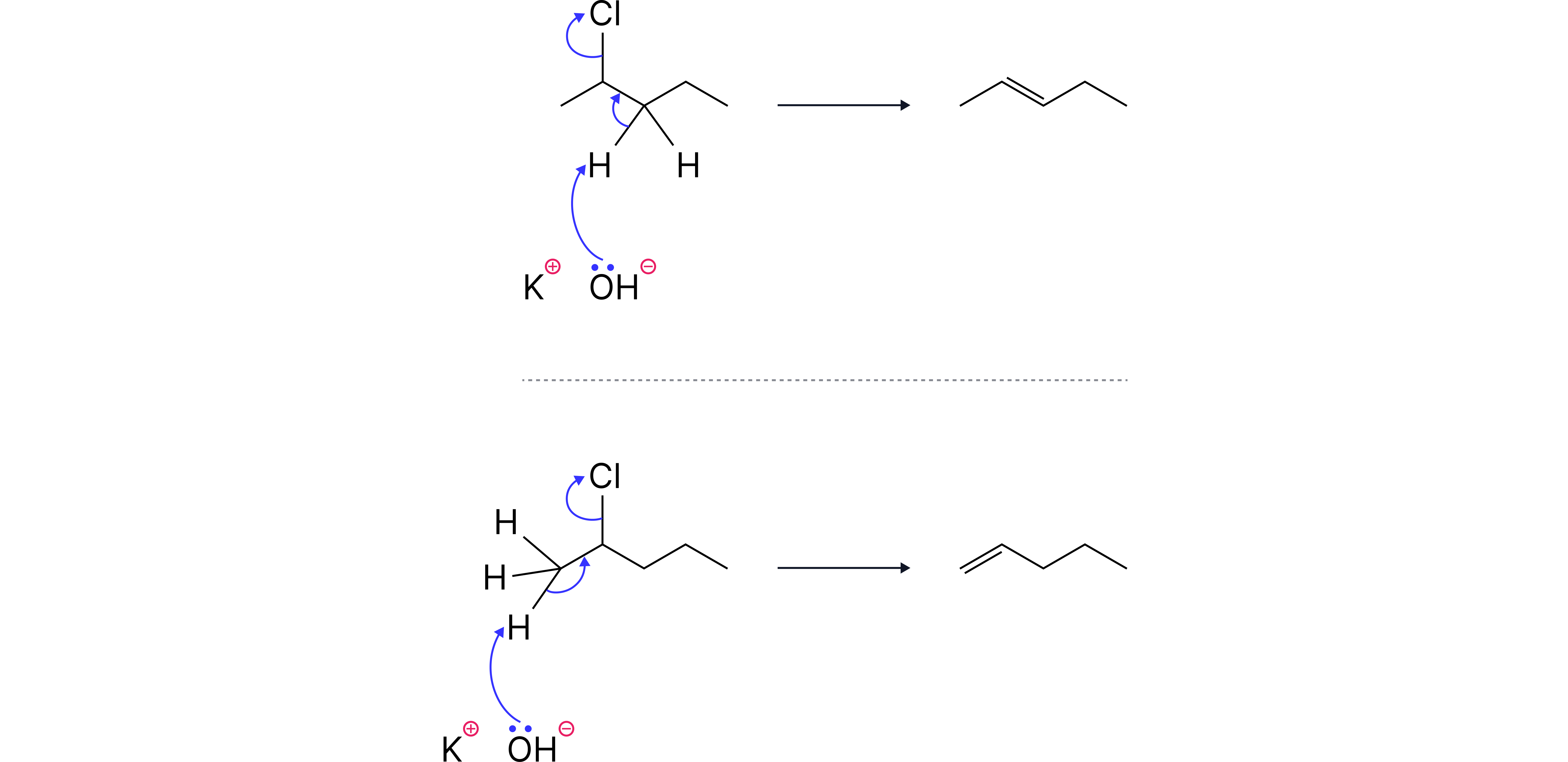
Example: 2-chloropentane
This secondary halogenoalkane can form two different alkenes via elimination:
pent-1-ene
pent-2-ene
This is due to the availability of two different hydrogens. The more substituted alkene (pent-2-ene) is often favoured.
In pent-2-ene, the double bond is between carbon-2 and carbon-3. Each of these carbons has two different groups attached, so E/Z (geometric) isomerism is possible:
is bonded to:
a methyl group ()
a hydrogen atom
is bonded to:
an ethyl group ()
a hydrogen atom
The isomers:
• Z-pent-2-ene: the two larger groups (ethyl and methyl) are on the same side of the double bond
• E-pent-2-ene: the two larger groups are on opposite sides
In practice, both isomers can form, but the E-isomer is usually more stable due to less steric hindrance – so it often predominates in the mixture.
Exam point: If the question asks for “all possible alkenes” from elimination, be sure to include both structural and E/Z isomers if applicable.
Effect of halogenoalkane class
Primary mostly substitution
Secondary mix of both (depends on conditions)
Tertiary mostly elimination
Steric hindrance around the makes substitution harder for tertiary halogenoalkanes.
Extra tips
Always look for multiple multiple possible alkenes.
Consider E/Z isomerism if relevant.
Always state heat and ethanolic solvent for elimination.
Know the definitions of and – they guide the mechanism.
In exam questions, if asked for a mechanism, draw it clearly with full curly arrows.