Nucleophilic Substitution
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Halogenoalkanes undergo substitution reactions when they react with nucleophiles.
Substitution – this is when a halogen atom in an organic molecule is replaced by a different atom or group.
Nucleophile – a species that donates a lone pair of electrons, such as or .
Understanding the mechanism
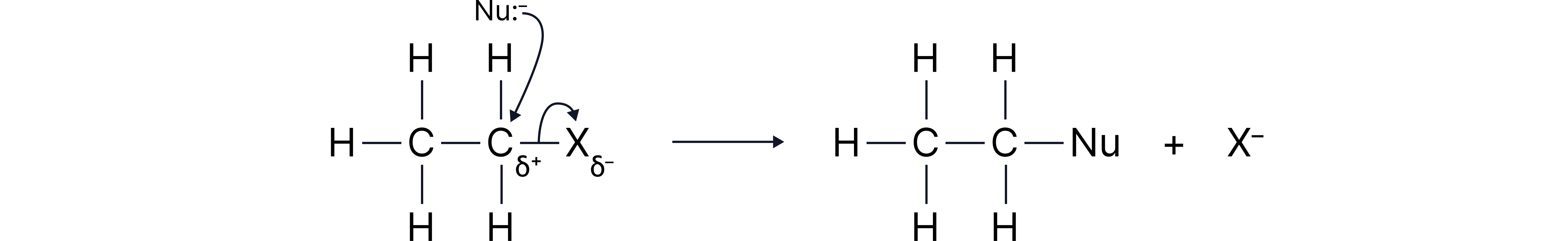
In organic chemistry, mechanisms are used to show the step-by-step process of how a reaction occurs.
Nucleophiles are negatively charged or neutral species with a lone pair of electrons. They attack carbon atoms that are slightly positive (δ⁺) due to the polar nature of the carbon–halogen bond. The halogen is more electronegative than carbon, pulling electrons towards itself and leaving the carbon slightly electron-deficient – making it open to attack by a nucleophile.
We use curly arrows to represent the movement of a pair of electrons. These always start from either a lone pair or the middle of a bond.
Reaction rate and bond strength
The speed of substitution depends heavily on how easy it is to break the C–X bond.
Weaker bonds are easier to break – so they react faster.
Stronger bonds are harder to break – so substitution is slower.
Bond enthalpies of C–X bonds :
Bond | Enthalpy/ |
238 | |
276 | |
338 | |
484 |
So, iodoalkanes (containing iodine) react the fastest, and fluoroalkanes are the least reactive due to the very strong C–F bond.
Comparing the Rate of Hydrolysis in Halogenoalkanes
Hydrolysis is the breaking of a chemical bond through reaction with water. In the case of halogenoalkanes, the C–X bond is broken and replaced with an –OH group:
General reaction:
(where or )
Why water?
Although water is a weak nucleophile, it can slowly attack the carbon bonded to the halogen in a nucleophilic substitution reaction, especially under warm conditions. The lone pair on the oxygen atom in water is attracted to the slightly positive carbon atom.
Tracking the reaction – using silver nitrate
To compare how quickly different halogenoalkanes undergo hydrolysis, we use aqueous silver nitrate as a test reagent.
When the halogenoalkane is hydrolysed, the halide ion is released.
These halide ions react with ions from silver nitrate to form a silver halide precipitate.
E.g.
(yellow precipitate)
The silver halide forms only once the halide ion has left, so the speed at which the precipitate appears tells us the rate of hydrolysis.
How to interpret the results
The faster the precipitate forms, the faster the substitution reaction is.
This tells us how reactive the halogenoalkane is.
Reactivity depends on the strength of the carbon–halogen bond (C–X):
Weaker bonds break more easily – faster reaction.
Stronger bonds are more difficult to break – slower reaction.
Trend in bond strengths and precipitate colour
Halogenoalkane | C–X bond strength | Silver halide formed | Precipitate colour |
Iodoethane | Weakest | Yellow | |
Bromoethane | Medium | Cream | |
Chloroethane | Strongest | White |
This matches the bond enthalpies so forms fastest and forms last. As a result, iodoalkanes hydrolyse fastest, while chloroalkanes hydrolyse slowest.
Nucleophilic Substitution with Aqueous Hydroxide Ions
Functional Group Change:
Halogenoalkane → Alcohol
Reagents and Conditions:
Reagent: Sodium hydroxide () or potassium hydroxide ()
Conditions: Warm, in aqueous solution
Mechanism: Nucleophilic substitution
Type of reagent: Nucleophile () – donates a lone pair of electrons
Example Reaction:
1-bromoethane + (aqueous) → ethanol +
The hydroxide ion attacks the carbon bonded to the bromine.
The bond breaks, and the bromide ion () leaves.
This forms ethanol, a primary alcohol.

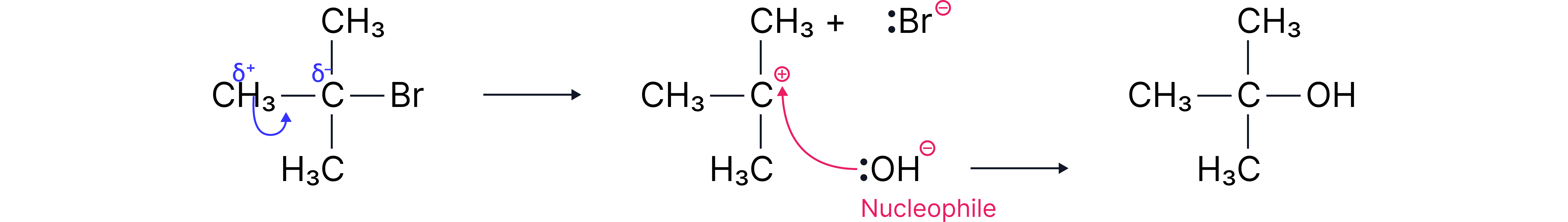
Alternative Mechanism: (Tertiary Halogenoalkanes)
Tertiary halogenoalkanes, such as 2-bromo-2-methylpropane, undergo nucleophilic substitution via a two-step mechanism known as (substitution, nucleophilic, unimolecular).
Example Reaction:
2-bromo-2-methylpropane + 2-methylpropan-2-ol +
Step 1: Formation of a carbocation
The C–Br bond breaks heterolytically.
A tertiary carbocation forms (this is the slow, rate-determining step).
Step 2: Nucleophilic attack
– The hydroxide ion donates its lone pair to the positively charged carbon, forming a new bond.
Why ?
Tertiary carbocations are stable due to the three methyl groups surrounding the positive carbon. These groups push electron density towards the carbocation via the inductive effect, helping to stabilise it.
Steric hindrance: The bulky groups make it difficult for OH⁻ to directly attack the carbon in a one-step () mechanism, so the reaction proceeds via the carbocation instead.
Nucleophilic Substitution with Cyanide Ions ()
Functional Group Change:
Halogenoalkane → Nitrile
Reagents and Conditions:
Reagent: Potassium cyanide ()
Solvent: Ethanol/water mixture
Conditions: Heat under reflux
Mechanism: Nucleophilic substitution
Type of reagent: Nucleophile ()
Example Reaction:
1-bromoethane + → propanenitrile +
The cyanide ion () attacks the carbon bonded to the bromine.
The bromide ion leaves, and a new bond forms.
The resulting nitrile has one more carbon than the original halogenoalkane.
Useful in synthesis – this method is ideal for increasing the length of a carbon chain.

Naming Nitriles:
The C in the –CN group counts as part of the main chain.
Start numbering from the nitrile carbon.
Example:
→ propanenitrile (3 carbons in total)
Not “propanitrile” – the e stays!
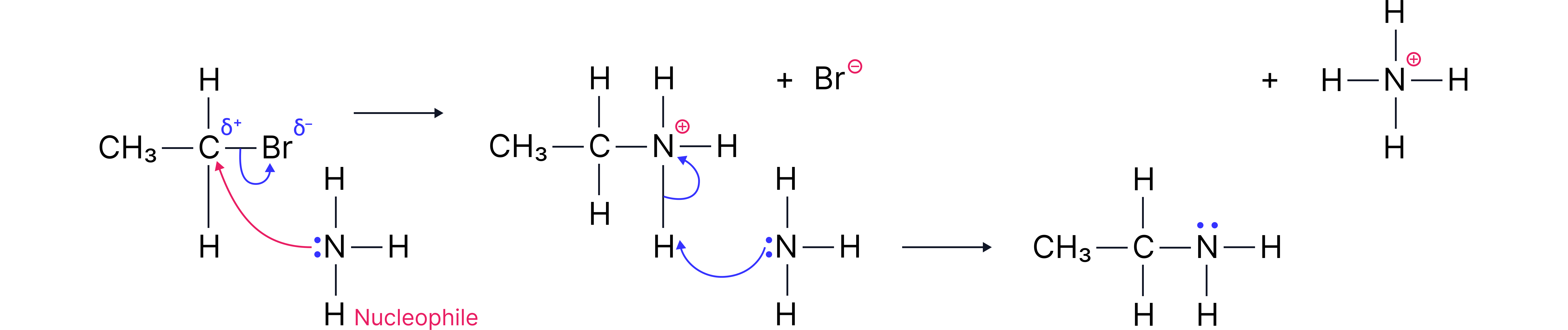
Nucleophilic Substitution with Ammonia ()
Functional Group Change:
Halogenoalkane → Primary Amine
Reagents and Conditions:
Reagent: Ammonia () in ethanol
Conditions: Heated under pressure in a sealed tube
Mechanism: Nucleophilic substitution
Type of reagent: Nucleophile ()
Example Reaction:
1-bromoethane ethylamine
Step 1: attacks the carbon and displaces .
Step 2: Another molecule removes a proton from the group, leaving a neutral primary amine and forming ammonium bromide.
Naming Amines:
Ethylamine refers to a 2-carbon chain with an group.
Or use IUPAC: ethanamine
Minimising Further Substitution:
Ethylamine, like ammonia, has a lone pair and can act as a nucleophile – potentially forming a secondary amine if it reacts again.
(diethylamine)
To limit this, use excess ammonia. This ensures ammonia outcompetes the amine in further substitution.
Worked Example
What is the organic product formed when 1-bromopropane reacts with under reflux in ethanol?
Answer
Butanenitrile
Which of these haloalkanes will hydrolyse fastest: 1-chlorobutane, 1-bromobutane, or 1-iodobutane? Explain why.
Answer
1-iodobutane. Because bond is weakest (lowest bond enthalpy), it breaks most easily.
Practice Questions
Question 1
Write the mechanism for the reaction between 1-bromopropane and hydroxide ion. Include curly arrows and lone pairs.
Answer
attacks carbon
bond breaks
Forms
Question 2
Explain why chloroalkanes are less reactive than iodoalkanes in nucleophilic substitution.
Answer
bond is stronger (higher bond enthalpy) than , so it requires more energy to break.
Question 3
Describe a test-tube experiment to compare the rate of hydrolysis of different haloalkanes.
Answer
Mix haloalkane with aqueous in ethanol
Measure time for precipitate to form
Faster formation = faster rate
Question 4
Give the organic product when 2-bromopropane reacts with aqueous ammonia.
Answer
Propan-2-amine