Ethanol Production
Lajoy Tucker
Teacher

Contents
Introduction
Alcohols have the functional group.
Ethanol is an important alcohol produced for fuel, industrial, and pharmaceutical uses.
It can be made by:
Hydration of alkenes (from crude oil)
Fermentation of glucose (from biomass)
Hydration of Alkenes
Reaction:
Step 1
Step 2
Overall:
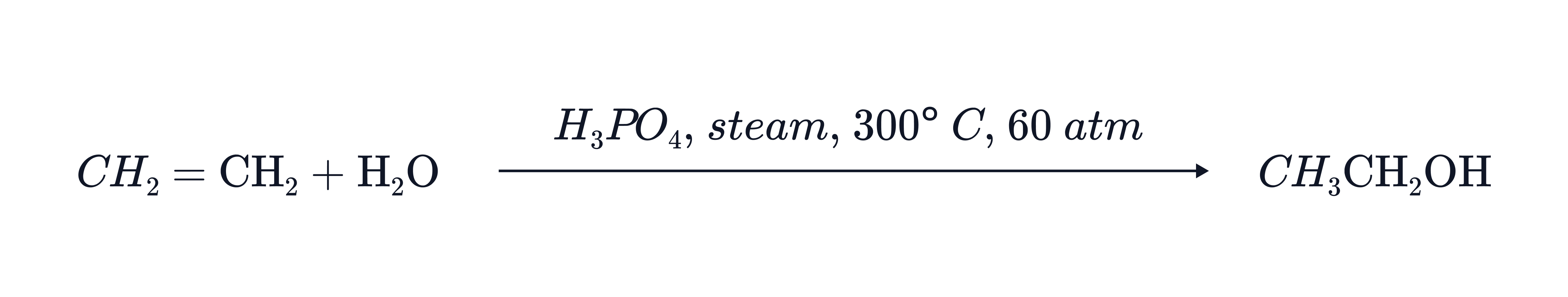
Conditions:
Reagent: Steam
Catalyst: Concentrated sulfuric acid or phosphoric acid
Temperature:
Pressure: 60–70 atm
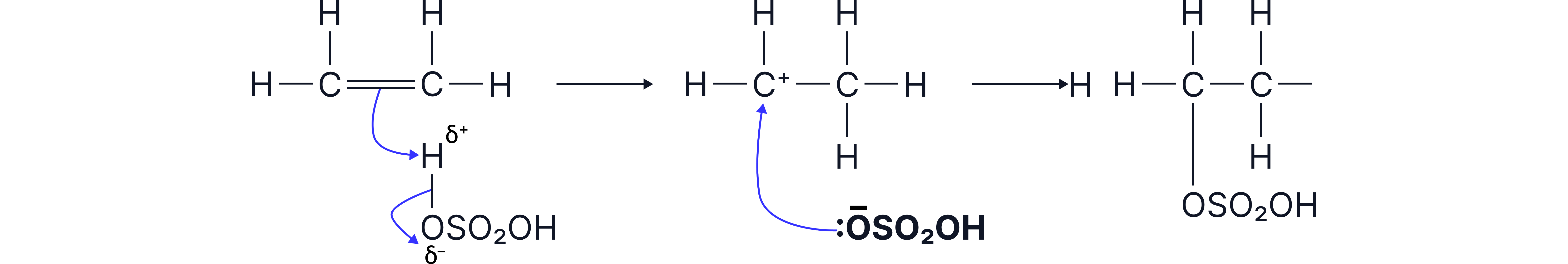
Mechanism:
Electrophilic addition via:
1. Protonation of alkene
2. Formation of a carbocation intermediate
3. Attack by the ion to form the alkyl hydrogen sulfate.
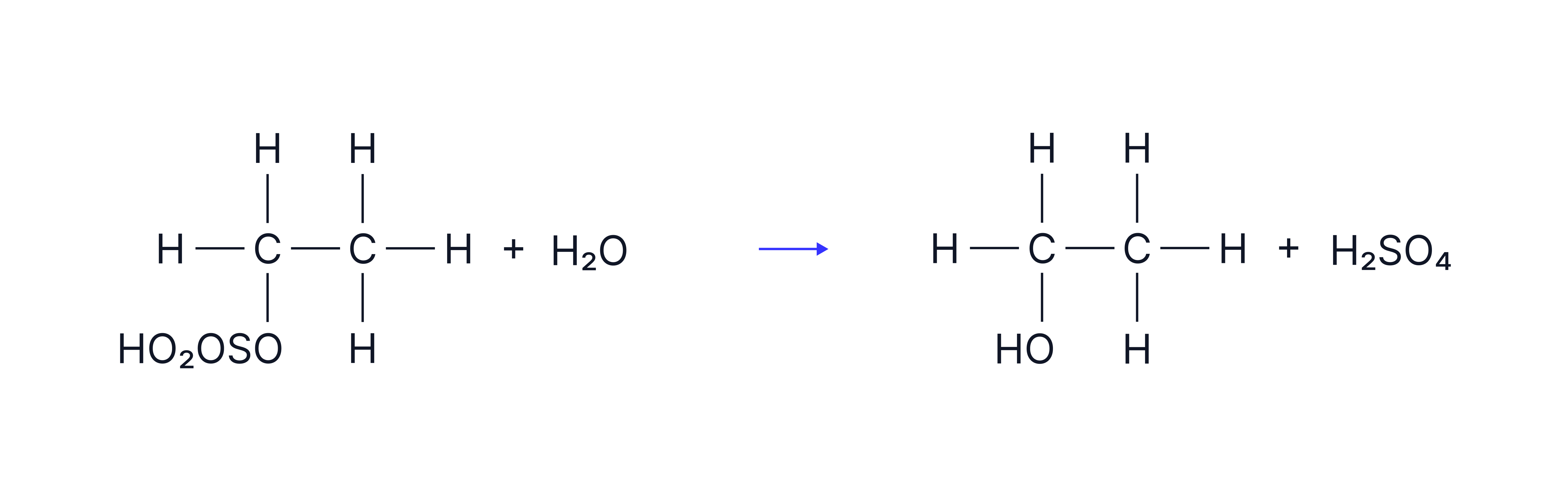
Followed by:
Hydrolysis with steam to produce the alcohol and regenerate the acid catalyst.
Ethanol by Fermentation of Glucose
Equation:
Conditions:
- Catalyst: Yeast (provides enzymes)
- Temperature:
(optimum temperature for enzyme activity)
- Anaerobic conditions (no oxygen)
- Neutral aqueous solution
Process:
- Ethanol is purified by fractional distillation.
Comparing Methods
| Hydration of Ethene | Fermentation of Glucose |
| + A continuous, rapid process where there is a flow of reactants passing over the catalyst resulting in a high percentage yield. | - Processed in batches, so production is slower and has a lower percentage yield. |
| - Carried out at high temperatures and pressures which are expensive to maintain. | + Cheaper as carried out at much lower temperatures and pressures. |
| + Greater purity of ethanol produced. | - Low purity (yeast is killed at approx. 15% ethanol) which must then be purified by fractional distillation. |
| + High atom economy | - Low atom economy |
| - Obtained from crude oil which is a finite raw material. | + Obtained from plant material (sugar cane) which is renewable. |
Biofuel & Carbon Neutrality
A biofuel is a fuel made from recently living biological material, e.g. sugar crops, which can be regrown. Bioethanol is ethanol produced by glucose fermentation and is an example of a biofuel.
Why bioethanol from fermentation is claimed to Be Carbon Neutral:
absorbed during photosynthesis is equal to released during fermentation and combustion.
There is no NET release of carbon dioxide.
Equations:
Photosynthesis:
Fermentation:
Combustion of Ethanol:
Why It Is Not Truly Carbon Neutral:
Fossil fuels used in:
Farming equipment
Transport
Distillation process
Deforestation and land-use change may increase
Environmental and Ethical Issues with Biofuel Use
Environmental Issues:
Deforestation: clearing land to grow biofuel crops
Loss of biodiversity from monocultures
Ethical Issues:
Food vs Fuel: land for biofuels may reduce food crop production
Rural Displacement for large-scale farming
Energy use in bioethanol may come from fossil fuels
Worked Examples
Question 1
Write the balanced equation for fermentation of glucose.
Question 2
State three conditions for fermentation of glucose.
Temperature:
Anaerobic conditions
Yeast catalyst
Neutral pH
Question 3
Give one reason why fermentation-based ethanol is not completely carbon neutral.
Energy from fossil fuels is used in farming, transport, and distillation, releasing additional
Question 4
Define a biofuel and give one example.
A biofuel is a fuel made from (recently) living biological material such as plants. Example: ethanol produced by fermentation of glucose.
Question 5
Explain one environmental and one ethical concern with large-scale bioethanol production.
Environmental: Monocultures reduce biodiversity.
Ethical: Land used for biofuel crops could reduce food supply.
Tips
Hydration of alkenes follows an electrophilic addition mechanism via the alkyl hydrogen sulfate.
When asked to ‘discuss’, ensure you include both sides of the argument