Elimination Reactions of Alcohols
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Alcohols are converted into alkenes by the removal of a water molecule.
This is a dehydration reaction through an elimination mechanism.
This is an acid-catalysed process.
The alkenes formed can be used as monomers for addition polymers, offering a renewable route to polymers that avoids crude oil-derived alkenes.
Acid-Catalysed Elimination of Alcohols
General Reaction:
Example:
Reagents/Conditions:
Reagent: Concentrated sulfuric or phosphoric acid
Conditions: Heat under reflux
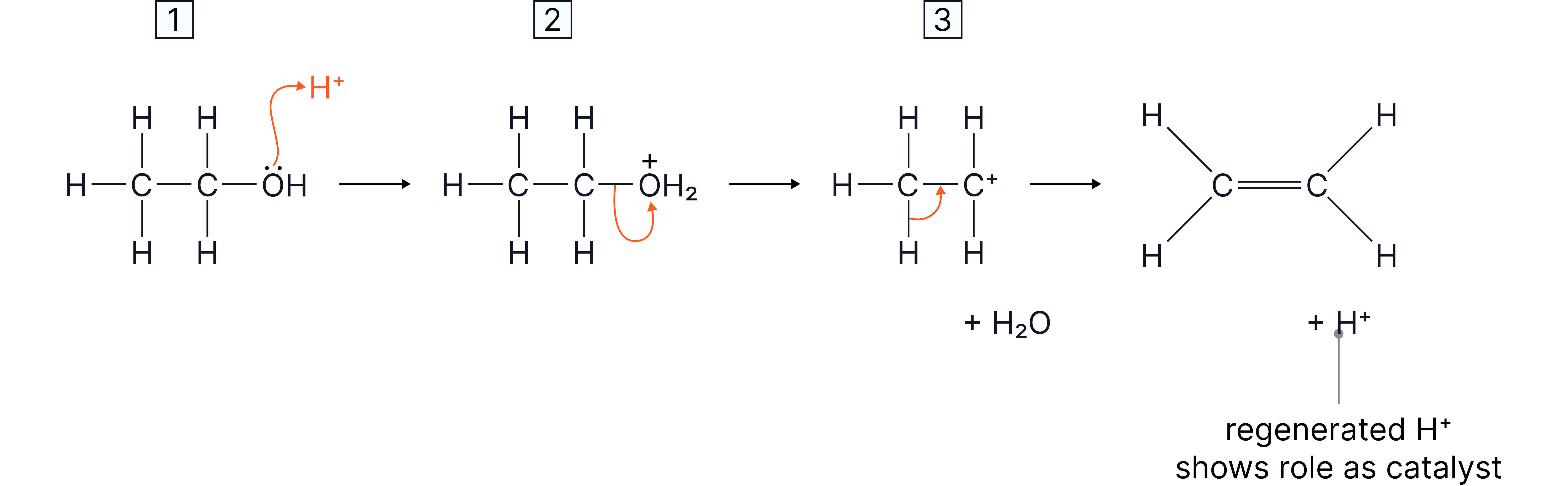
Mechanism of Elimination
1. Protonation of the group (making it a better leaving group as )
2. Loss of to form a carbocation intermediate
3. Loss of a proton from an adjacent carbon to form the C=C double bond
Diagram
Formation of Different Isomers
If there is more than bond adjacent to the , dehydration can occur at more than one position, producing a mixture of alkenes.
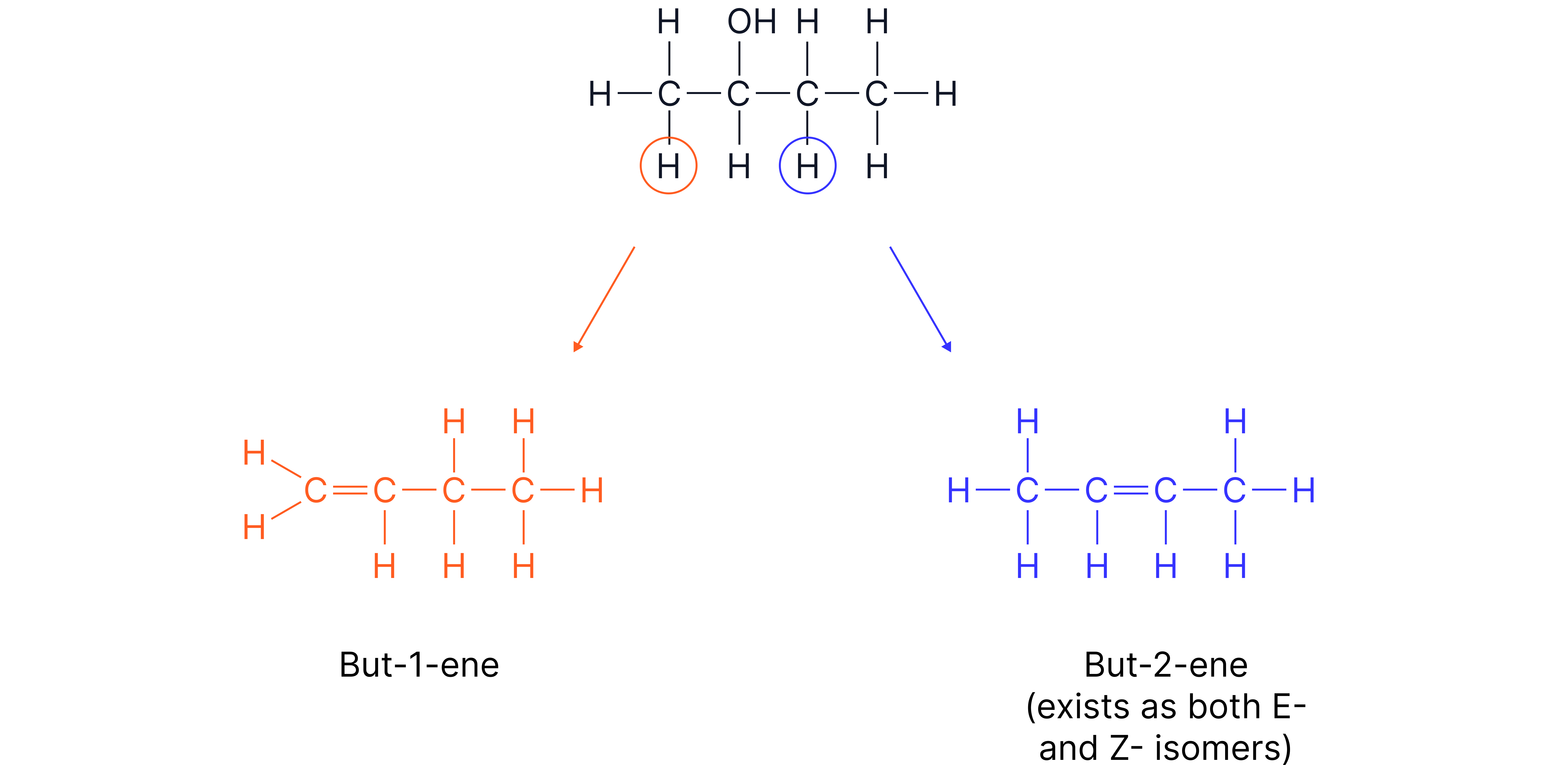
Dehydration of butan-2-ol will give a mixture of but-1-ene and but-2-ene which are structural isomers (specifically position isomers).
But-2-ene can also exist as E/Z stereoisomers.
Exam Tip!
When producing alkenes, always look out for position isomers based on removing different AND isomerism in the products
Worked Example
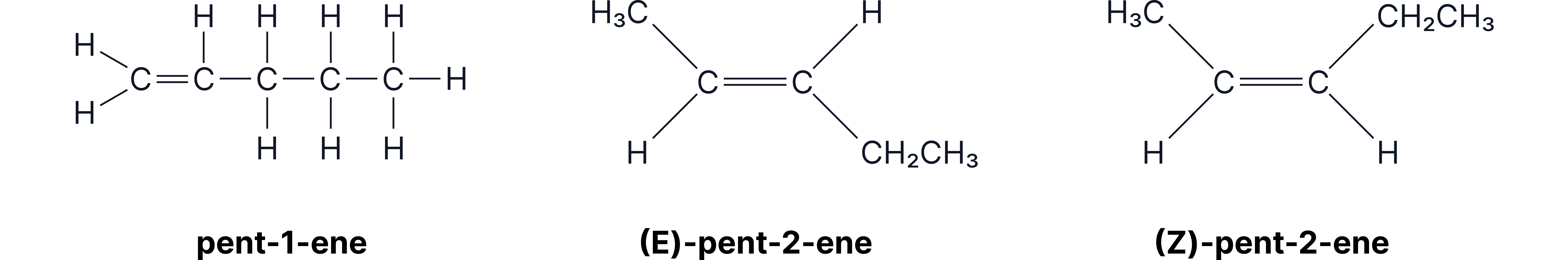
Why can the elimination of pentan-2-ol produce three isomeric alkenes?
Answer
Hydrogen can be removed from either adjacent carbon to the , giving structural isomers, pent-1-ene and pent-2-ene.
Pent-2-ene exists as stereoisomers due to restricted rotation around the double bond and two different groups attached to each C of the double bond.
Practice Questions
Question 1
Name the mechanism for the reaction that occurs when ethanol is heated with concentrated phosphoric acid.
Answer
Elimination
Question 2
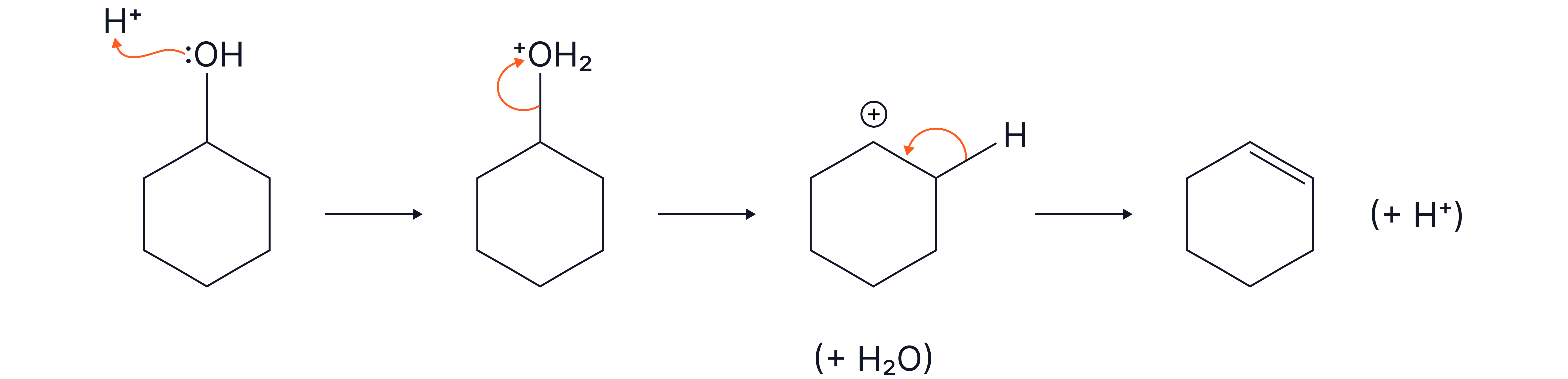
Outline the mechanism for the acid-catalysed elimination of cyclohexanol. Show all relevant curly arrows and lone pairs

Answer
Tips
Heat and acid are both essential for dehydration.
If the group is on a carbon with more than one adjacent , expect isomer formation.
Elimination forms unsaturated products (alkenes) that can undergo electrophilic addition later or addition polymerisation.