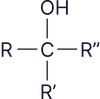Oxidation of Alcohols
Lajoy Tucker
Teacher

Definition
Oxidation can be defined as the:
- Gain of oxygen
- Loss of hydrogen
- Loss of electrons
- Increase in oxidation state
Some alcohols can be oxidised by reaction with a suitable oxidising agent. The oxidation products depend on the classification of the alcohol and the reaction conditions.
Classification of Alcohols
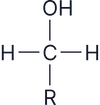
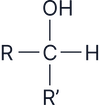
Alcohols are classified based on the number of alkyl (R) groups attached to the carbon bonded to the –OH group:
| Alcohol Type | Structure | Oxidation product(s) |
|
Primary (1°)
|
Oxidised to aldehydes, then carboxylic acids
|
|
|
Secondary (2°)
|
|
Oxidised to ketones only
|
|
Tertiary (3°)
|
Not readily oxidised
|
Suitable Oxidising Agent
Potassium dichromate(VI) with sulfuric acid (“acidified potassium dichromate”) is used as a suitable oxidising agent.
- In chemical equations the oxidising agent
/
is more simply represented as [O].
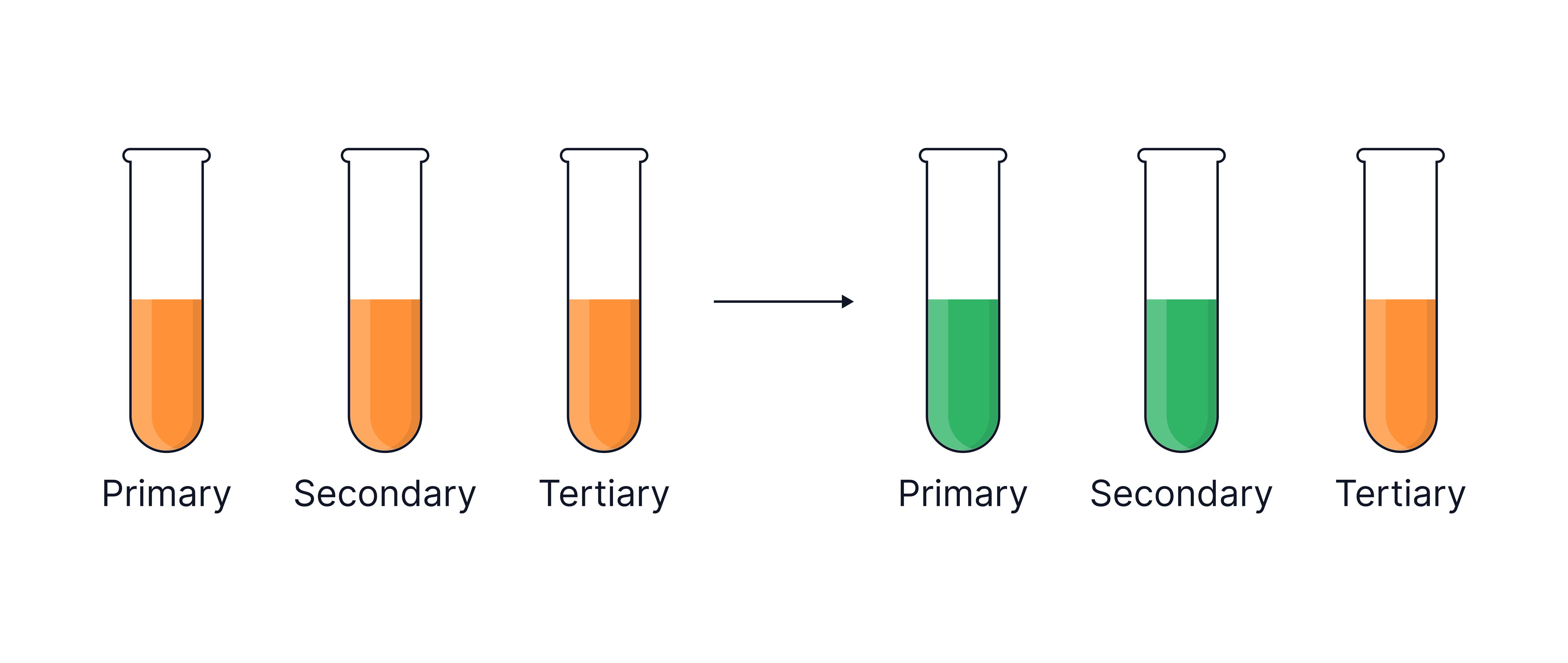
- When oxidation takes place using acidified potassium dichromate, a colour change of orange to green is observed as the dichromate ion is itself reduced to
.
Oxidation Reactions of Alcohols
A. Primary Alcohols
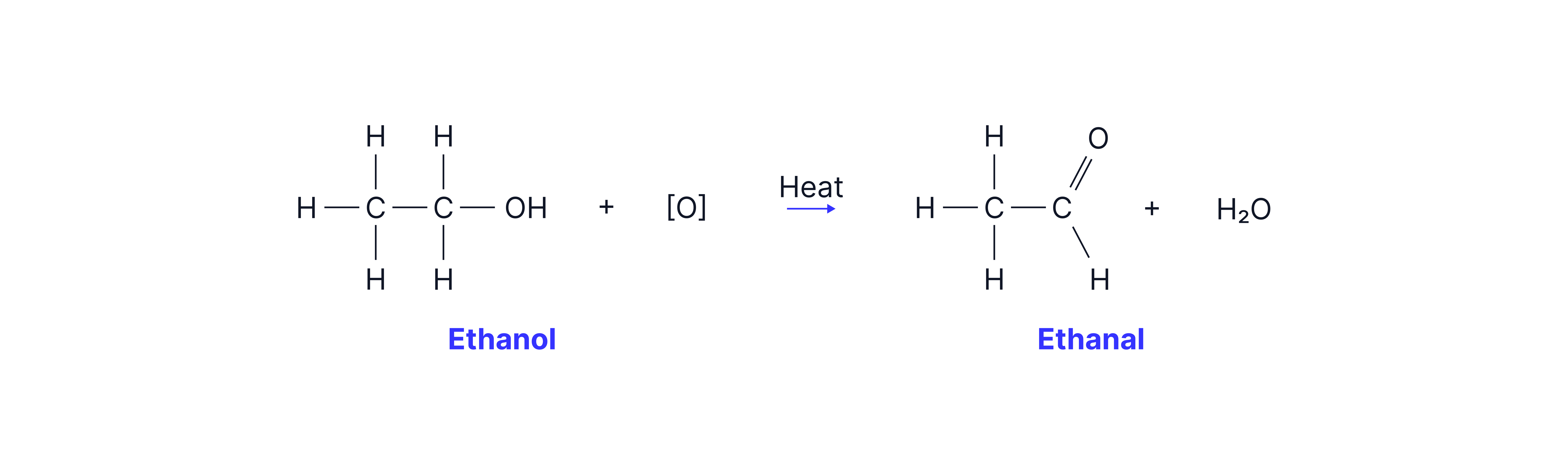
Primary alcohols can be partially oxidised to aldehydes and fully oxidised to carboxylic acids.
Partial Oxidation → Aldehyde
Conditions:
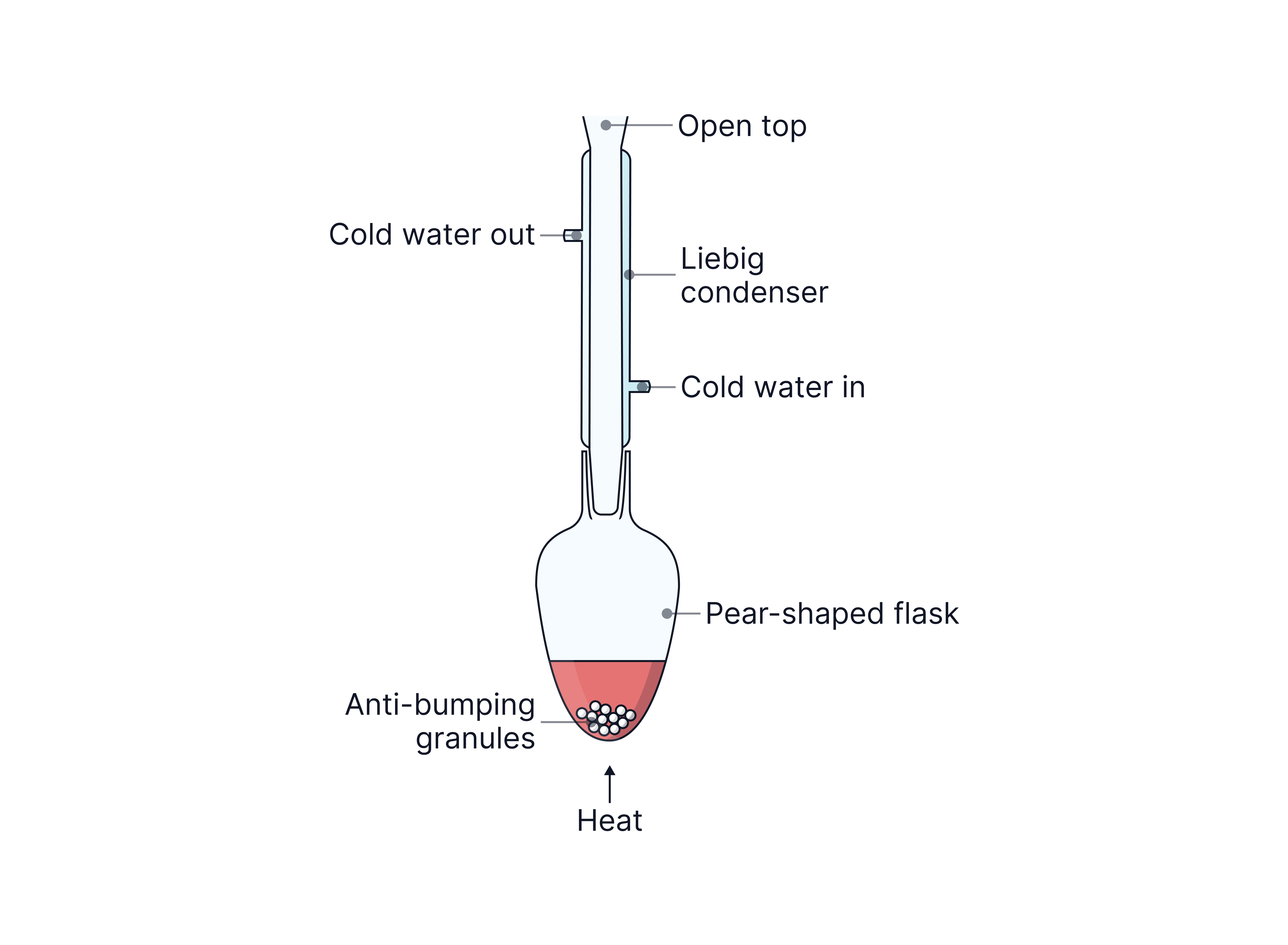
Distillation
This reaction must be carried out in distillation apparatus to ensure the aldehyde is removed as soon as it is formed so it is not oxidised further to the carboxylic acid.
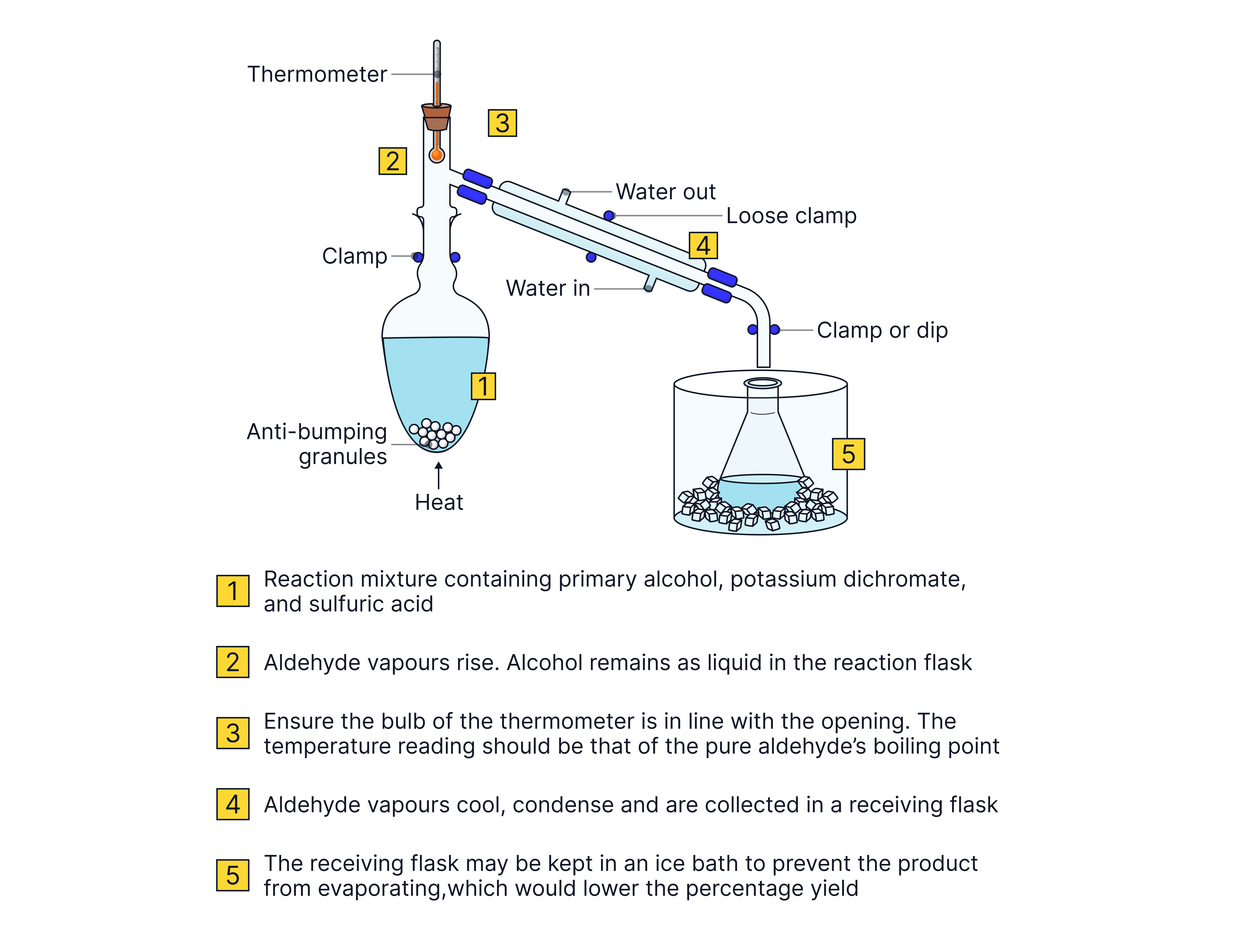
This is possible because aldehydes have lower boiling points than the corresponding alcohol. Alcohols can form hydrogen bonds between molecules which are stronger than the permanent dipole-dipole interactions between aldehyde molecules thus requiring more energy to overcome.
Complete Oxidation → Carboxylic Acid
The aldehyde formed can be oxidised further to a carboxylic acid
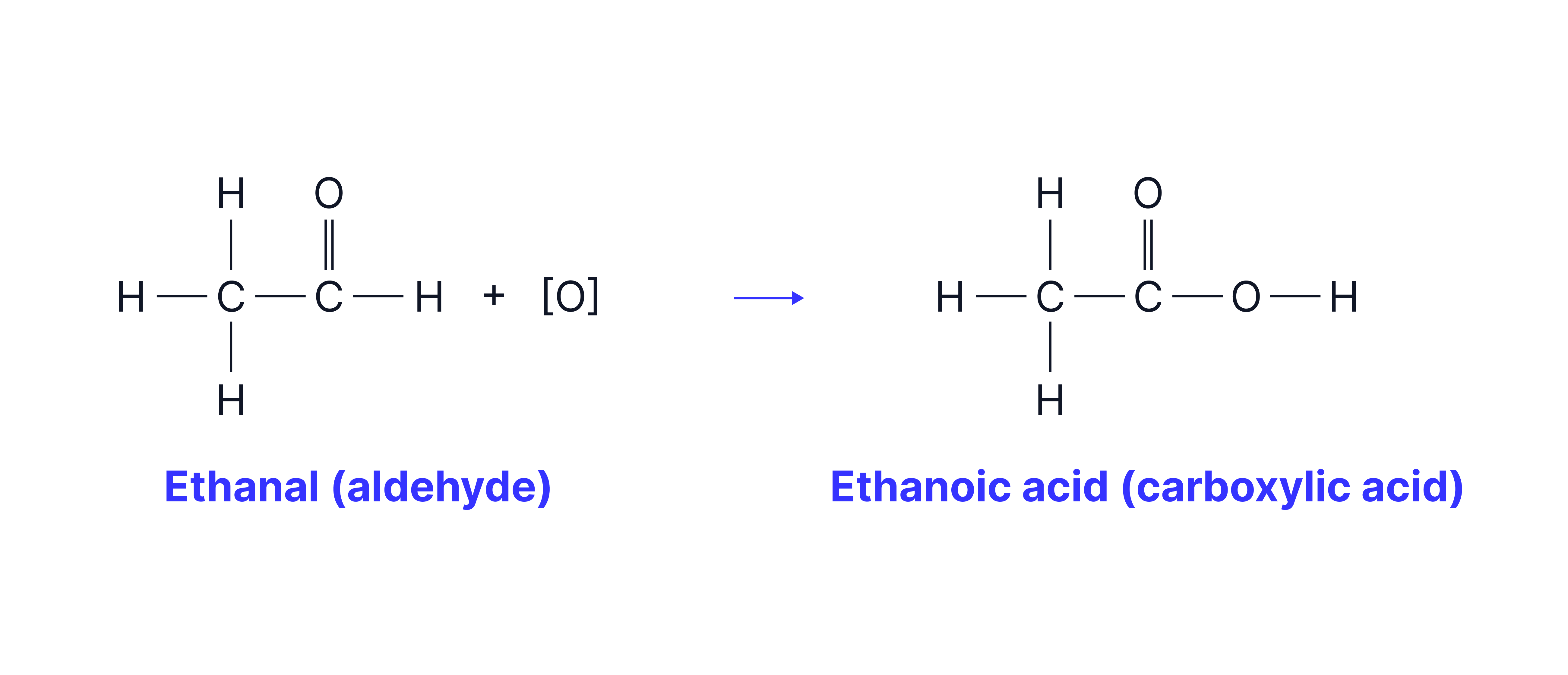
Note that no water is formed in the second oxidation step.
The carboxylic acid can also be formed from the primary alcohol.
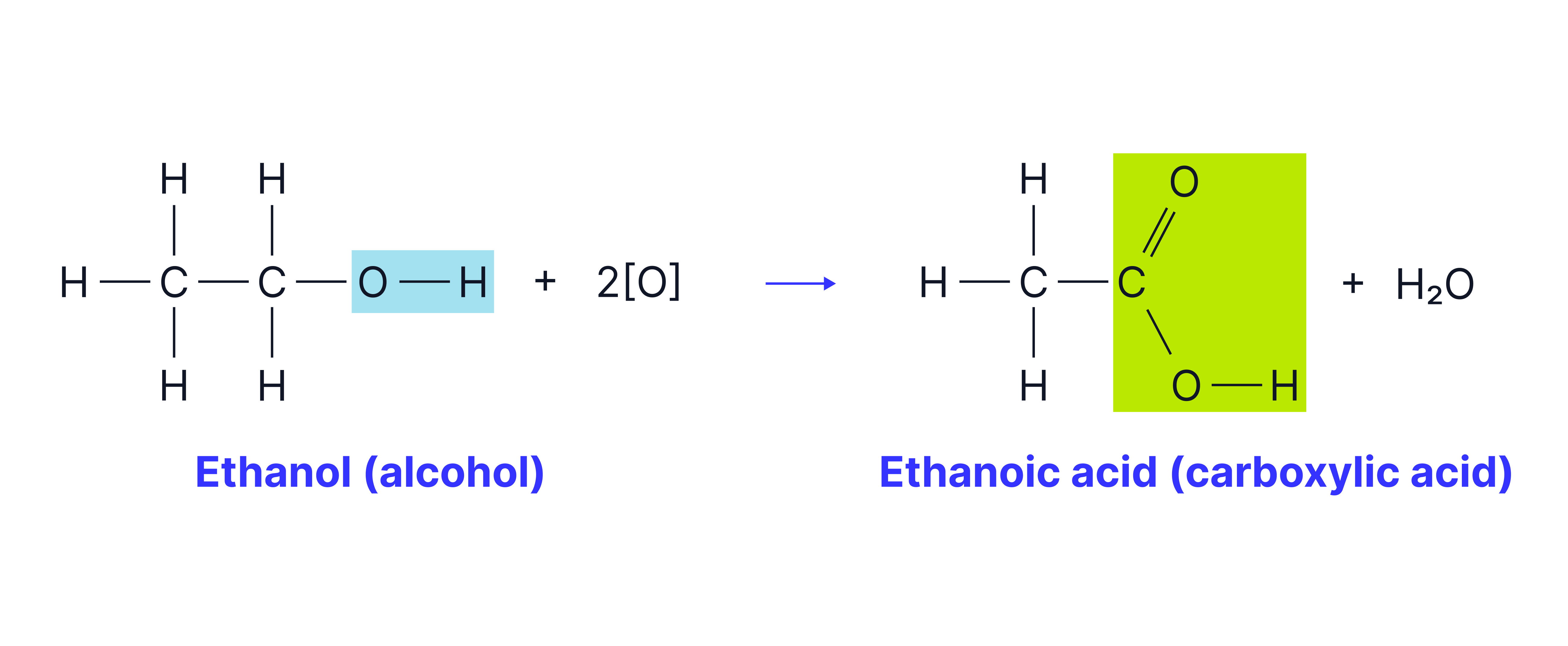
Conditions:
Excess acidified potassium dichromate
Reflux
By heating under reflux, any aldehyde formed falls back into the reaction mixture allowing further oxidation to the carboxylic acid
B. Secondary Alcohols
Oxidised to Ketones:
e.g
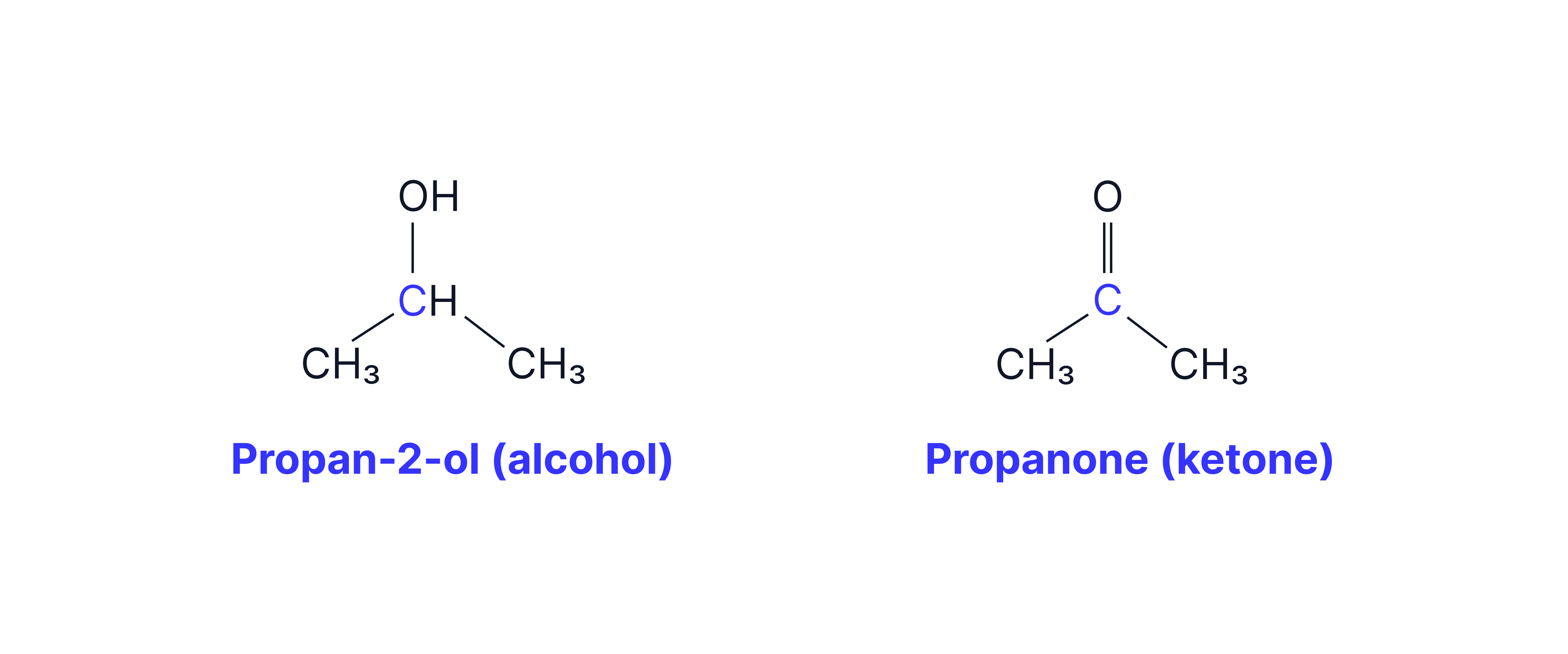
Conditions:
As the ketone is the only possible oxidation product, the heating conditions are less important.
C. Tertiary Alcohols
Do not oxidise this way (no H atom on the carbon bearing the –OH group)
No colour change with acidified .
Example:
Summary Table
Alcohol Class | Oxidised To | Test Outcome |
|---|---|---|
Primary | Aldehyde → Carboxylic acid | Orange → Green |
Secondary | Ketone | Orange → Green |
Tertiary | No reaction | No colour change |
Worked Examples
Example 1
Write the equation for the complete oxidation of propan-1-ol. Use [O] to represent the oxidising agent.
Example 2
State and explain the reaction conditions required to produce a carboxylic acid from a primary alcohol.
Heat under reflux with excess acidified potassium dichromate(VI)
Allows complete oxidation to carboxylic acid
Question
Explain why the aldehyde can be separated from the reaction mixture by distillation (3 marks).
Aldehydes vaporise at lower temperatures than the corresponding alcohol as the permanent dipole-dipole interactions between their molecules are weaker than the hydrogen bonds between alcohol molecules.
Tips
Use distillation to isolate aldehydes, reflux to form carboxylic acids when oxidising 1o alcohols.
Include [O] in equations when the full formula of the oxidising agent isn't used.
Know the colour change for acidified oxidising 1o and 2o alcohols (orange to green).
Tertiary alcohols give no reaction and therefore no colour change with oxidising agents.