Fractional Distillation of Crude Oil
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Crude oil is a mixture of hydrocarbons (mostly alkanes) formed from the remains of marine organisms over millions of years.
Alkanes are saturated hydrocarbons (contain only and single bonds).
Petroleum is a term used for crude oil along with its refined products such as gasoline, diesel, and kerosene
These products can be separated by fractional distillation
Fractional distillation
Fractional distillation separates crude oil based on the boiling points of components.
Key Principles:
Short-chain hydrocarbons: lower boiling points, rise to the top.
Long-chain hydrocarbons: higher boiling points, collected at the bottom.
Process:
1. Crude oil is heated and vaporised.
2. Vapour enters a fractionating column which is hotter at bottom and cooler at top.
3. Hydrocarbon vapours condense at different heights based on boiling points.
4. Groups of hydrocarbons (fractions) have similar chain lengths and similar boiling points.
Diagram
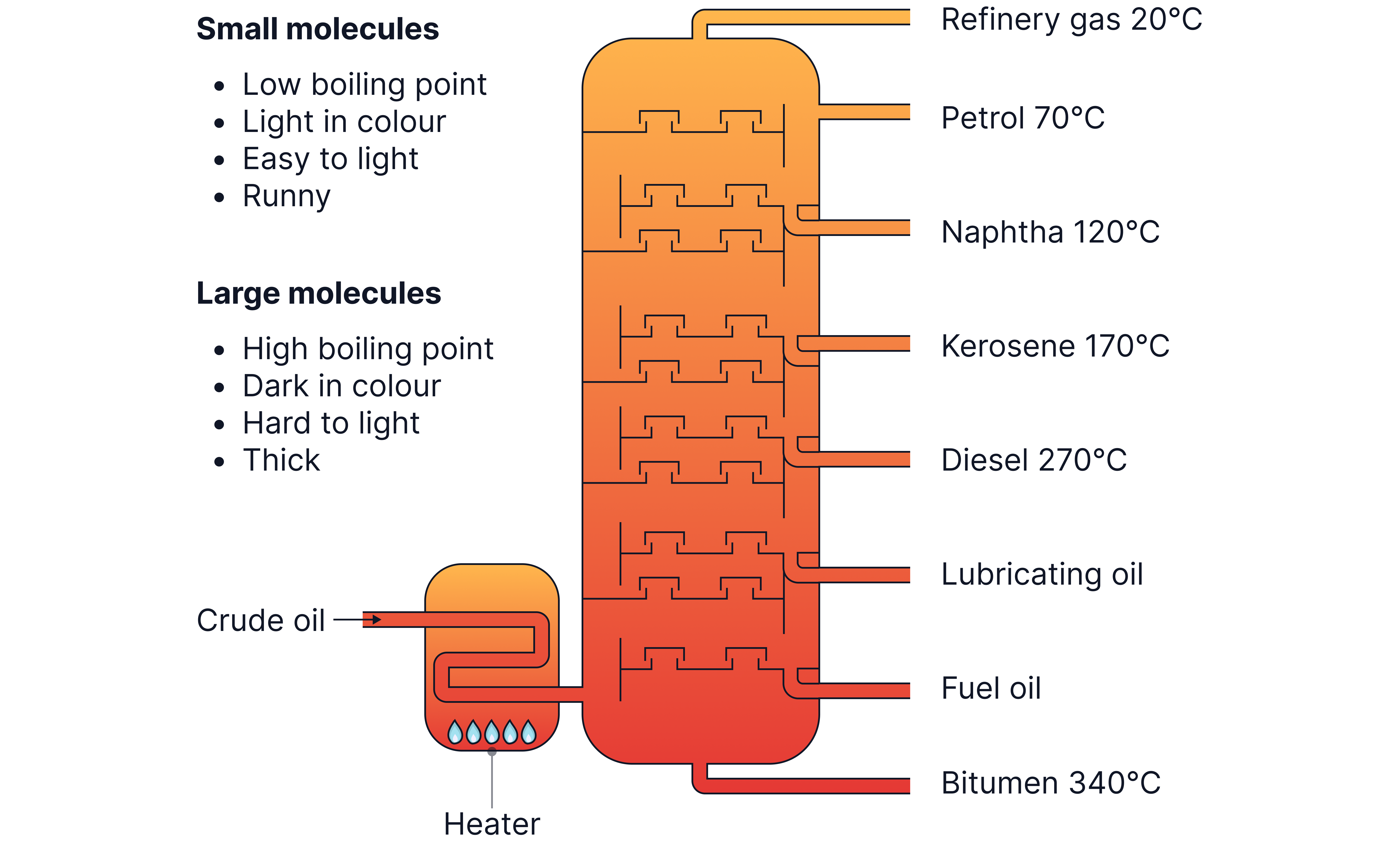
Common Fractions and Uses (not explicitly assessed)
Fraction | Approx. Boiling Range | Use |
|---|---|---|
Refinery gases | Bottled gas, heating | |
Gasoline (petrol) | Car fuel | |
Naphtha | Chemical feedstock | |
Kerosene | Jet fuel | |
Diesel | Vehicle fuel | |
Fuel oil | Ship fuel, power stations | |
Bitumen (residue) | Roads, roofing |
Worked Example
Why do long-chain alkanes have higher boiling points than short-chain alkanes?
Answer
Longer chain alkanes have larger surface areas/more electrons/a greater and stronger van der Waal’s forces between molecules, requiring more energy to overcome.
Tips
This is a very small specification point that is often assessed in relation to bonding and intermolecular forces.
Remember that fractional distillation is based on physical properties, not chemical changes.