Chlorination of Alkanes
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Chlorination of alkanes is an example of a free radical substitution reaction.
This reaction typically occurs between an alkane and chlorine () in UV light.
Radical – species with an unpaired electron
Chlorine radicals are formed through homolytic fission of the Cl–Cl bond using UV light.
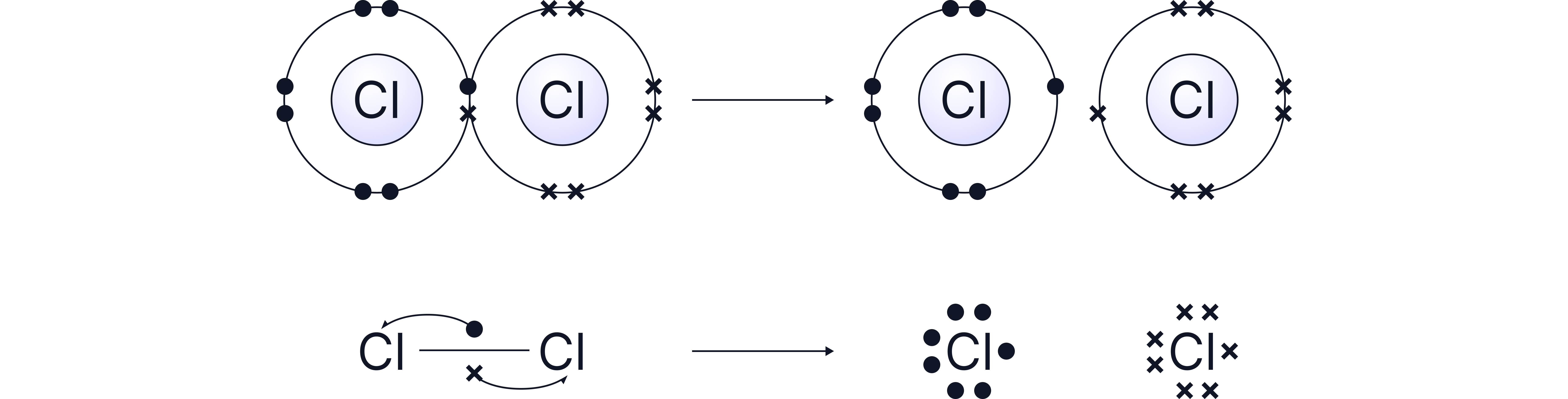
Homolytic fission means the covalent bond breaks evenly giving one electron to each of the bonded atoms
These radicals are highly reactive and drive the substitution of hydrogen atoms in alkanes with chlorine atoms.
General equation:
Where is an alkane and is the chloroalkane
Free Radical Substitution Mechanism
The mechanism consists of three distinct steps:
Initiation
Propagation
Termination
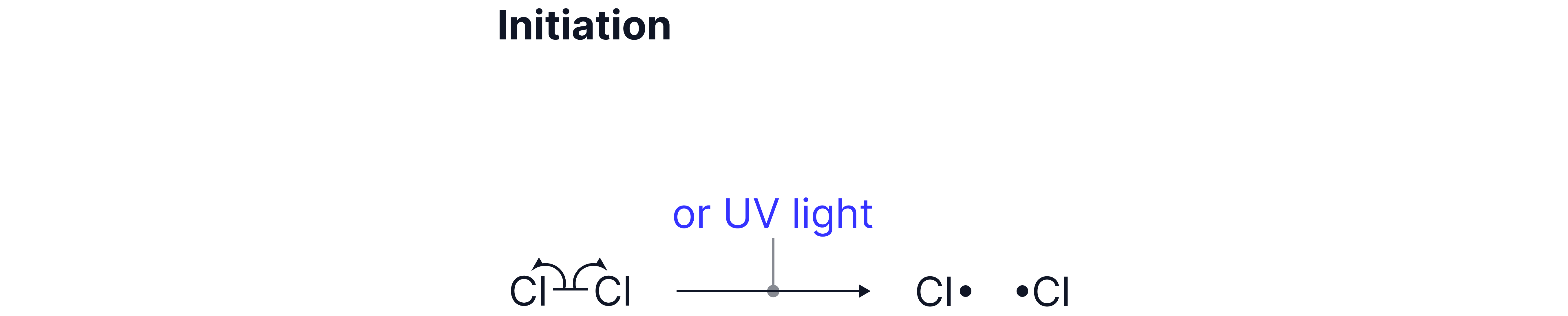
A. Initiation Step
UV light breaks the Cl–Cl bond by homolytic fission, producing two chlorine radicals.

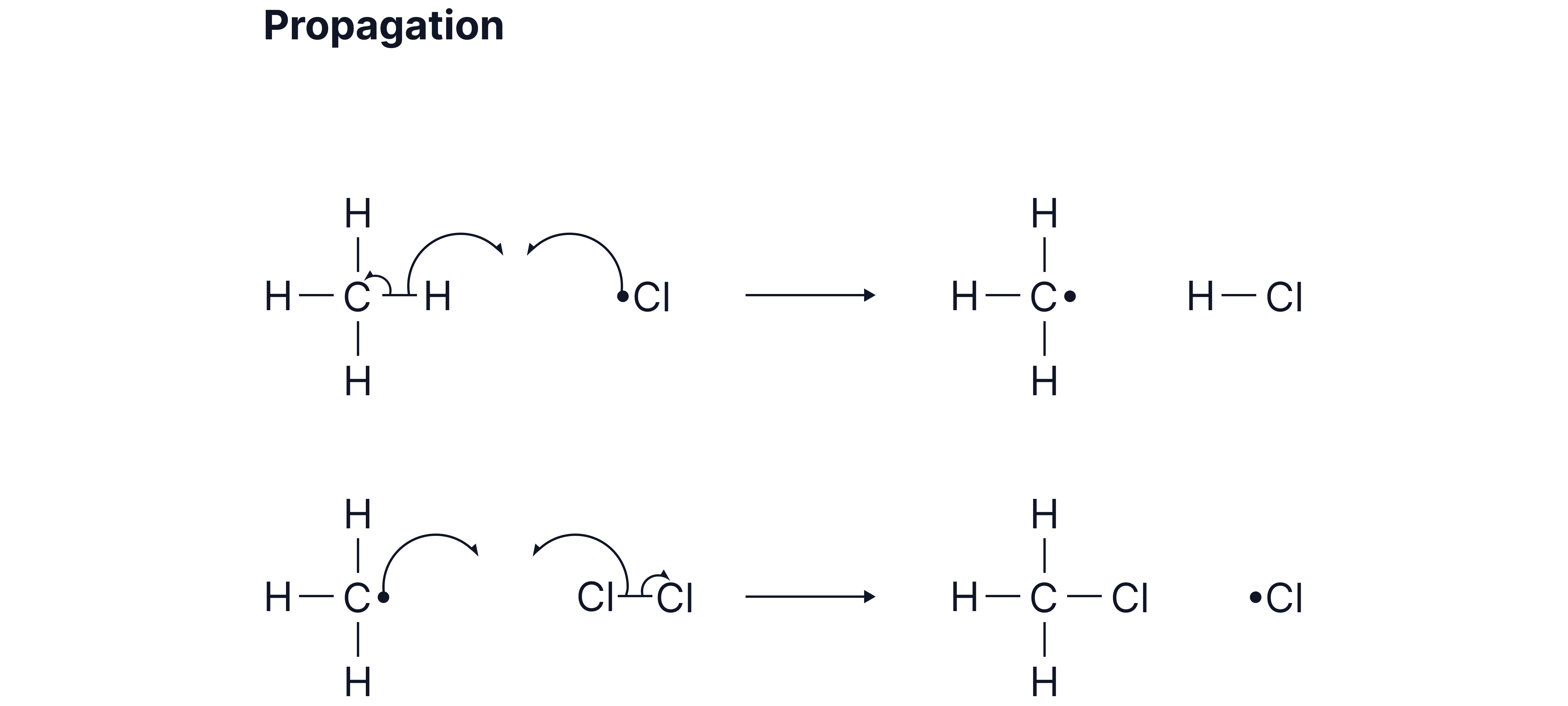
B. Propagation Steps
1. A chlorine radical reacts with the alkane to form an alkyl radical and hydrogen chloride e.g.

2. The alkyl radical then reacts with another molecule to produce chloromethane and regenerate a chlorine radical e.g.

These propagation steps repeat in a chain reaction.
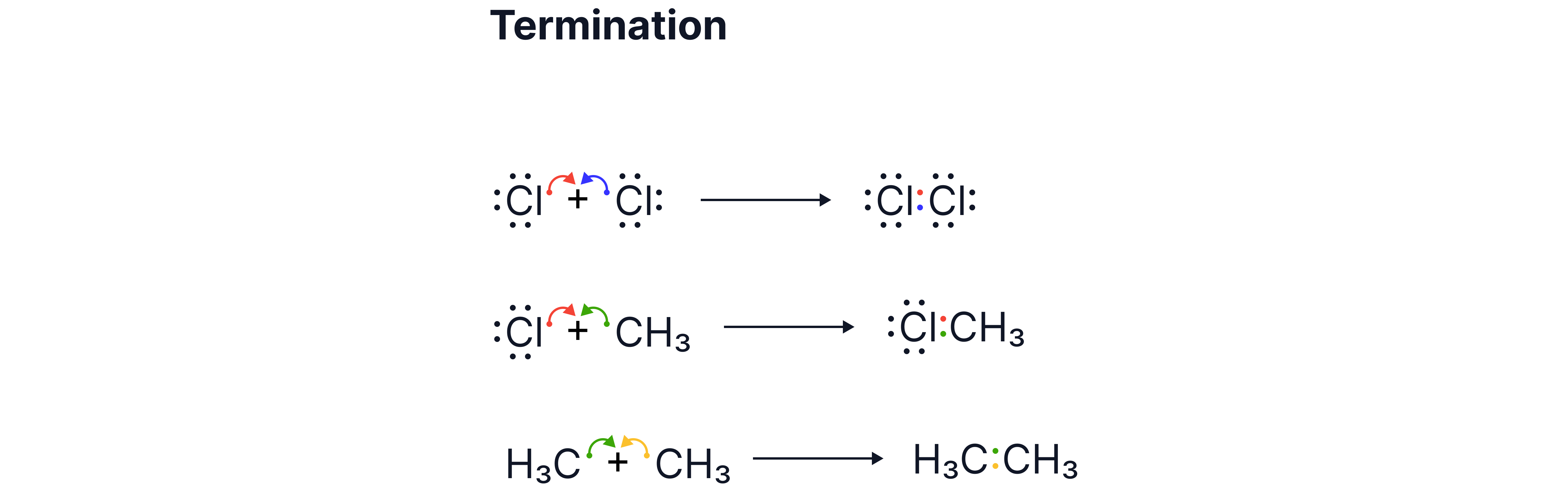
C. Termination Steps
Two radicals combine to form a stable molecule as the electrons become paired, ending the chain reaction.
Two chlorine radicals
Possible combinations:

An alkyl radical with a chlorine radical

Two alkyl radicals

D. Reaction Summary: Methane + Chlorine
Overall reaction:

The mechanism below shows the movement of electrons. Fish-hook arrows represent the movement of a single electron. This is not assessed by AQA but can be useful to understand how bonds are breaking and forming.
Mixture of Products
This reaction mechanism can form a mixture of products
1. Different H atoms substituted
The chlorine radical could attack any H atom leading to position isomers
e.g.
From propane, 1-chloropropane or 2-chloropropane could be formed through substitution of H atoms of C-1 or C-2.
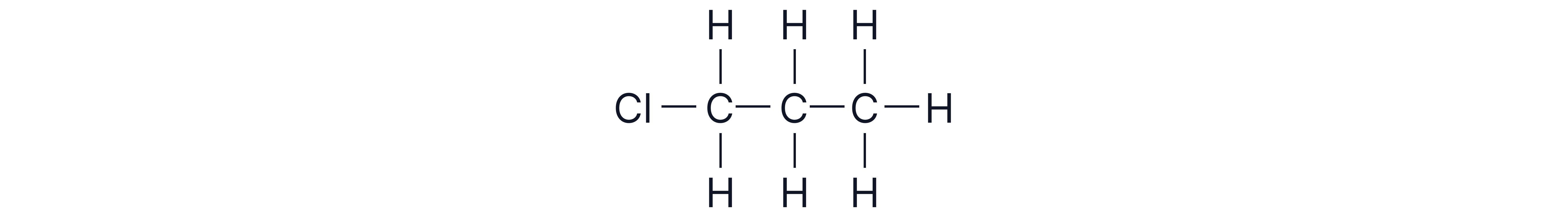
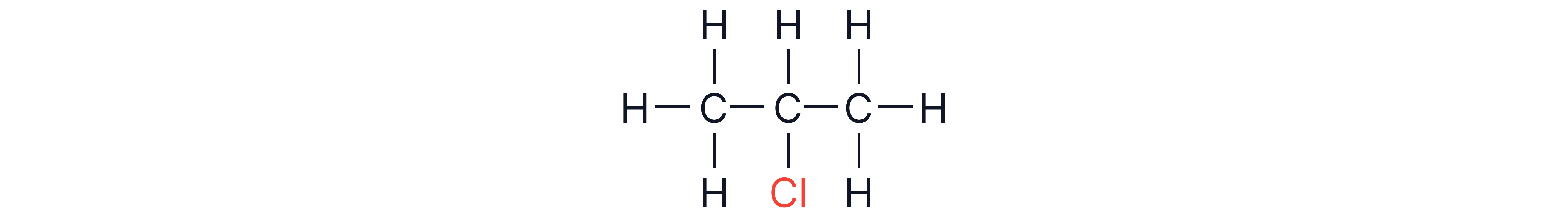
2. Further substitution
The chlorine radical is regenerated in the second propagation step and could go on to substitute another H atom.
This further substitution can form di-, tri- and tetra- substituted products e.g. dichloromethane (), trichloromethane (), and tetrachloromethane ().
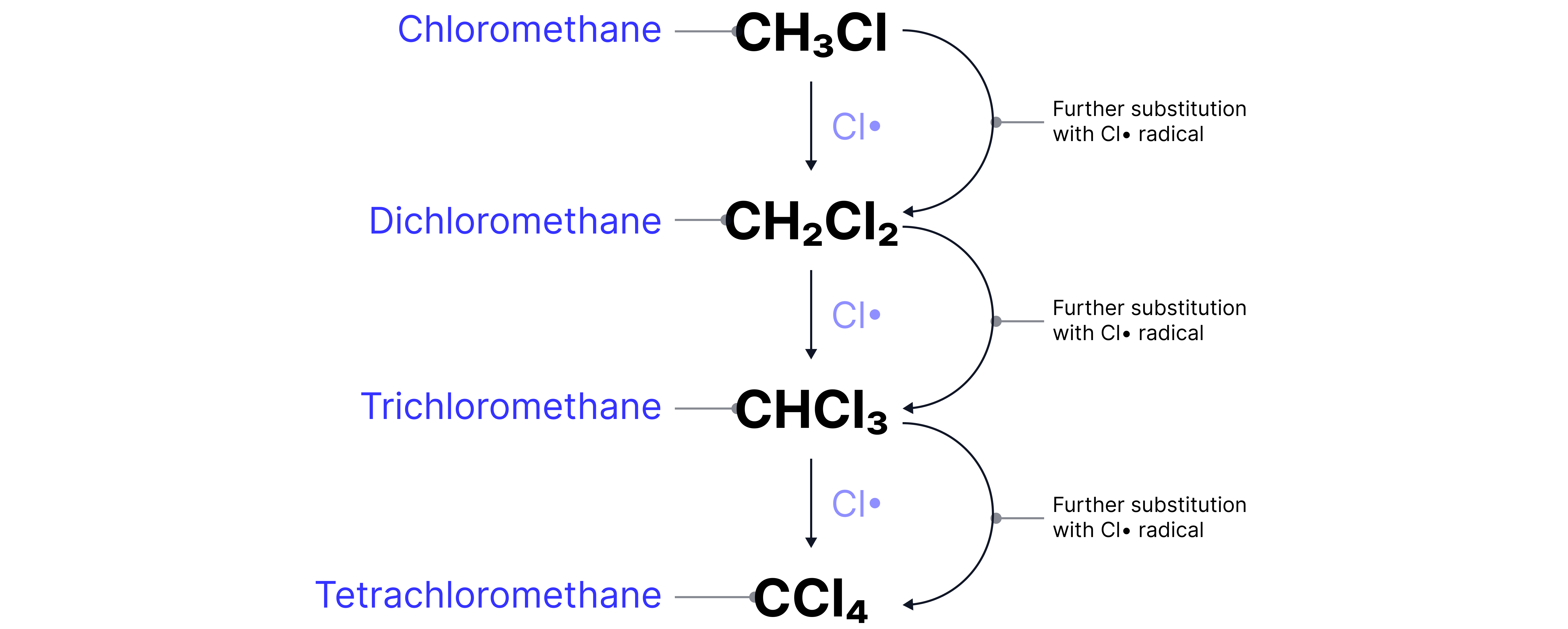
Practice Questions
Example 1
Write equations for the propagation steps for the reaction between butane and chlorine to produce 2-chlorobutane.
Answer:
1 – alkane with chlorine radical
2- alkyl radical with chlorine molecule
Tip – use structural formula in your equations to clearly show which hydrogen atom is being substituted.
Example 2
Di- and tri-substituted chlorobutanes are also isolated during this reaction. Explain why this is the case.
Answer:
The chlorine radical is regenerated in the second propagation step.
This results in a chain reaction where it can substitute another hydrogen atom.
Key Tips and Reminders
There are three possible termination products depending on the combination of radicals
Use dot notation (·) at the radical atom
Use structural formulae in equations for unambiguous products.