Combustion of Alkanes
Lajoy Tucker
Teacher

Contents
Introductions and Definitions
Combustion: The reaction of a substance with oxygen, releasing heat and light.
Complete Combustion: Occurs when alkanes react fully with excess oxygen to produce carbon dioxide and water .
Incomplete Combustion: Occurs when there is a limited supply of oxygen, producing carbon monoxide or carbon (, soot) and water.
Basic Principles
Alkanes are used as fuels because they release large amounts of energy on combustion.
The general equation for complete combustion of an alkane is:
however it is possible to balance these equations quite easily step by step:
Balance Carbons in
Balance Hydrogens in
Balance Oxygens in
In incomplete combustion, products can include CO (toxic gas) or C (soot), alongside water.
These reactions are exothermic.
Explanations
A. Complete Combustion
Example with butane :
Used in heaters and fuels where oxygen supply is plentiful.
B. Incomplete Combustion
Occurs in restricted oxygen supply (e.g. faulty boilers, vehicle engines).
Example (carbon monoxide produced):
Example (carbon/soot produced):
Carbon monoxide is toxic, binds to haemoglobin and reduces oxygen transport.
Soot causes breathing problems and environmental damage.
Pollutants from Combustion
The internal combustion of engines produces several harmful gases because of incomplete combustion and high-temperature reactions inside of them.
Main Pollutants
Pollutant | Source | Environmental/Health Effect |
CO (carbon monoxide) | Incomplete combustion of hydrocarbons | Toxic; binds to haemoglobin, reducing oxygen transport in blood |
C (carbon/soot) | Incomplete combustion | Causes respiratory problems and contributes to global dimming |
NO and NO₂ (collectively NOₓ) | Reaction of N₂ and O₂ at high temperatures in engines | Forms acid rain and photochemical smog; causes respiratory issues |
Unburned hydrocarbons | Escape from exhaust due to incomplete combustion | Contribute to photochemical smog and formation of ozone |
SO₂ (sulfur dioxide) | Combustion of sulfur impurities in fuels | Forms acid rain when dissolved in atmospheric moisture |
Catalytic Conventers
Structure and Materials:
Made from a ceramic honeycomb to maximize surface area.
Coated with a thin layer of transition metal catalysts: platinum (), palladium (), and rhodium ().
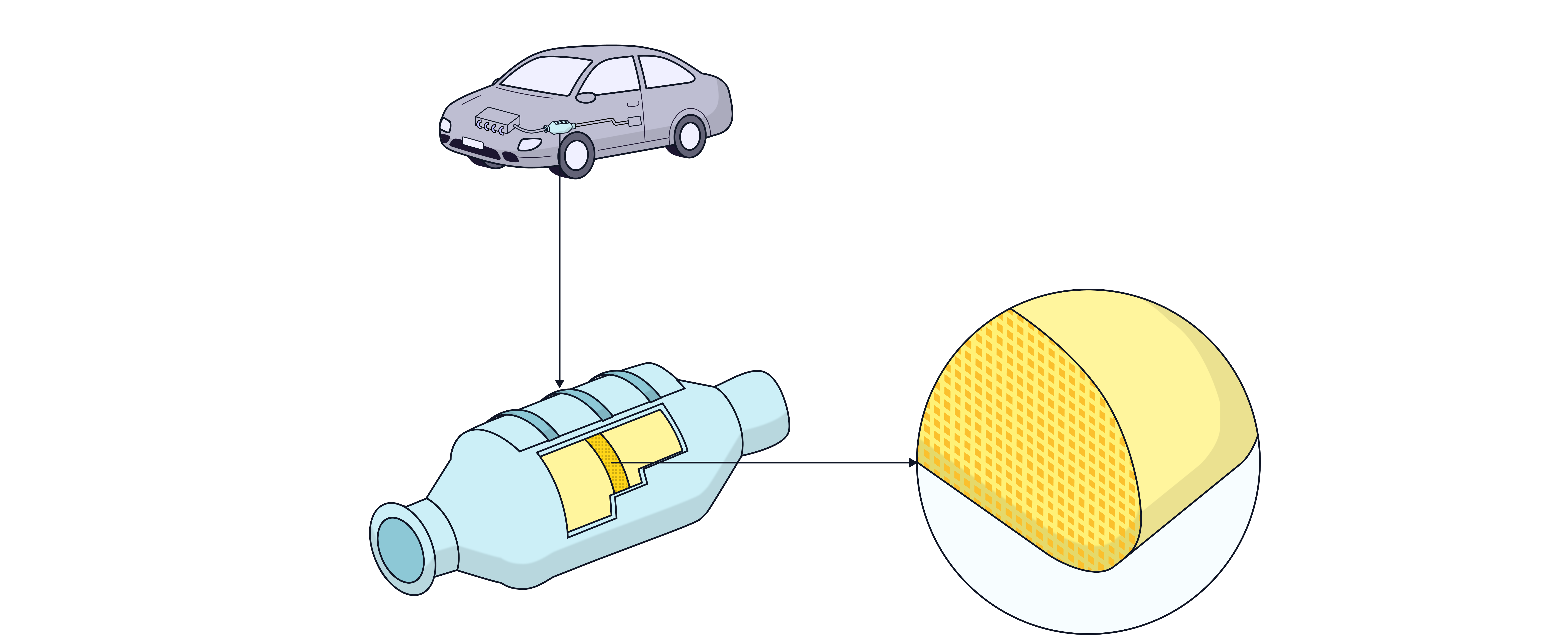
How They Work:
They catalyse redox reactions that convert harmful gases into harmless ones.
Key Reactions
Carbon monoxide + nitrogen monoxide:
Unburned hydrocarbons + nitrogen oxides:
e.g.
General:
1. and unburnt hydrocarbons are oxidised
2. are reduced.
Sulfur Dioxide and Flue Gas Desulfurisation
Sulfur dioxide is produced from burning fuels containing sulfur impurities.
The sulfur dioxide dissolves in rainwater and clouds producing acid rain, which damages buildings, ecosystems, and aquatic life.
Sulfur dioxide can be removed in power stations before they can impact the environment. This process is called Flue Gas Desulfurisation.
This process removes from exhaust gases in power stations.
Reactants Used
Calcium oxide ()
Calcium Carbonate
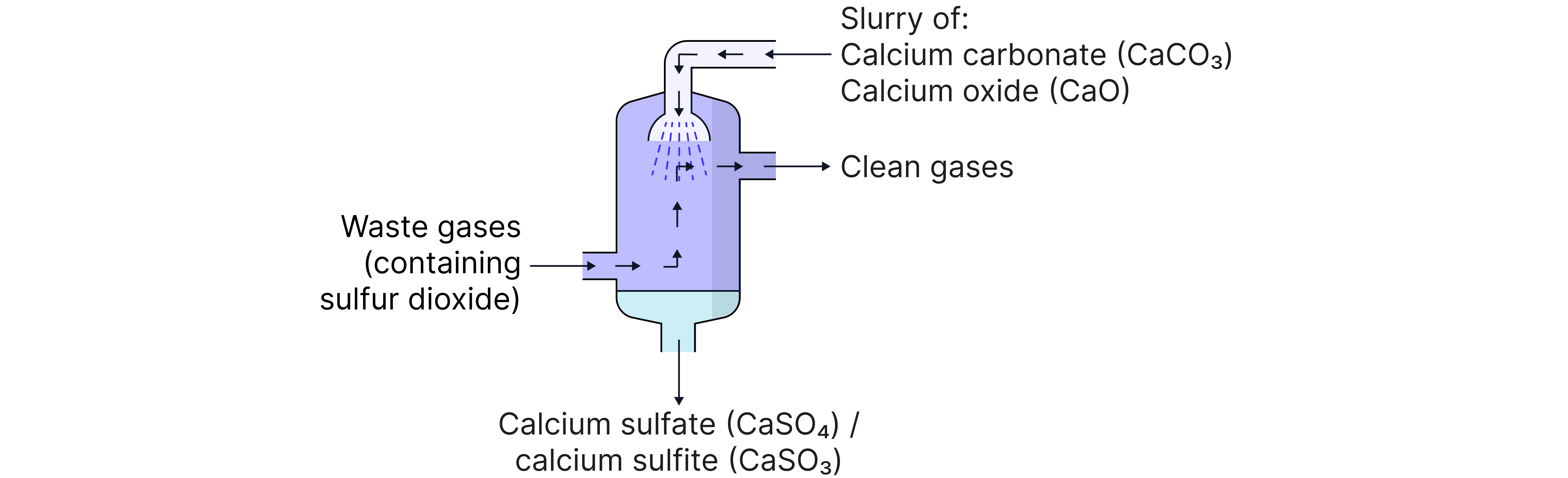
Example equations
1.
2.
These reactions produce solid calcium sulfite which can be safely disposed of or further processed.
Worked Examples
Example 1
Write a balanced equation for the complete combustion of hexane.
Answer:
Example 2
Write a balanced equation for the incomplete combustion of heptane () forming and :
Answer:
Practice Questions
Question 1
Which product is NOT formed during incomplete combustion of alkanes?
A. Water
B. Carbon Monoxide
C. Soot
D. Carbon Dioxide
Answer:
D. Carbon Dioxide
Question 2
A. Write a balanced equation for the complete combustion of decane .
B. Write a balanced equation for the incomplete combustion of decane to form carbon monoxide and water only.
C. State one reason why incomplete combustion occurs in petrol engines.
Answer:
A. or
B.
C. Incomplete combustion occurs due to insufficient oxygen in the combustion chamber, especially under high load or poor engine conditions.
Question 3
A. Name two harmful gases (other than carbon dioxide) produced by internal combustion engines.
B. Write an equation to show how a catalytic converter removes one of these gases.
C. Explain how the structure of a catalytic converter makes it efficient.
Answer:
A.
Carbon monoxide
Nitrogen monoxide
B.
C. Catalytic converters contain a ceramic honeycomb coated with platinum, palladium or rhodium. provides a large surface area for reactions, increasing the rate of conversion of harmful gases to less harmful products.
Question 4
A. Explain how sulfur dioxide is formed during the combustion of fossil fuels.
B. Give one environmental consequence of releasing sulfur dioxide into the atmosphere.
C. Write an equation to show how calcium carbonate removes sulfur dioxide from flue gases.
Answer:
A. Sulfur dioxide forms when sulfur impurities in fuels react with oxygen during combustion reactions.
B. Sulfur dioxide causes acid rain, which damages vegetation, aquatic ecosystems, and buildings.
C.
Tips
Balance combustion equations in the order: , and .
Use fractions for if needed.
Sulfur dioxide is not removed by catalytic converters — it's removed in power stations using .
Be clear which pollutants are removed where (cars vs flue gases).