Modification of Alkanes by Cracking
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Cracking: The process of breaking down large saturated hydrocarbon molecules (alkanes) into smaller hydrocarbons, including alkanes and alkenes, by breaking C–C bonds.
Purpose of Cracking: To meet the demand for shorter-chain hydrocarbons like petrol and to produce alkenes used in industrial synthesis (e.g. polymers).
Basic Principles
Crude oil contains a higher proportion of long-chain alkanes than is needed.
Shorter chain alkanes are in high demands as fuels.
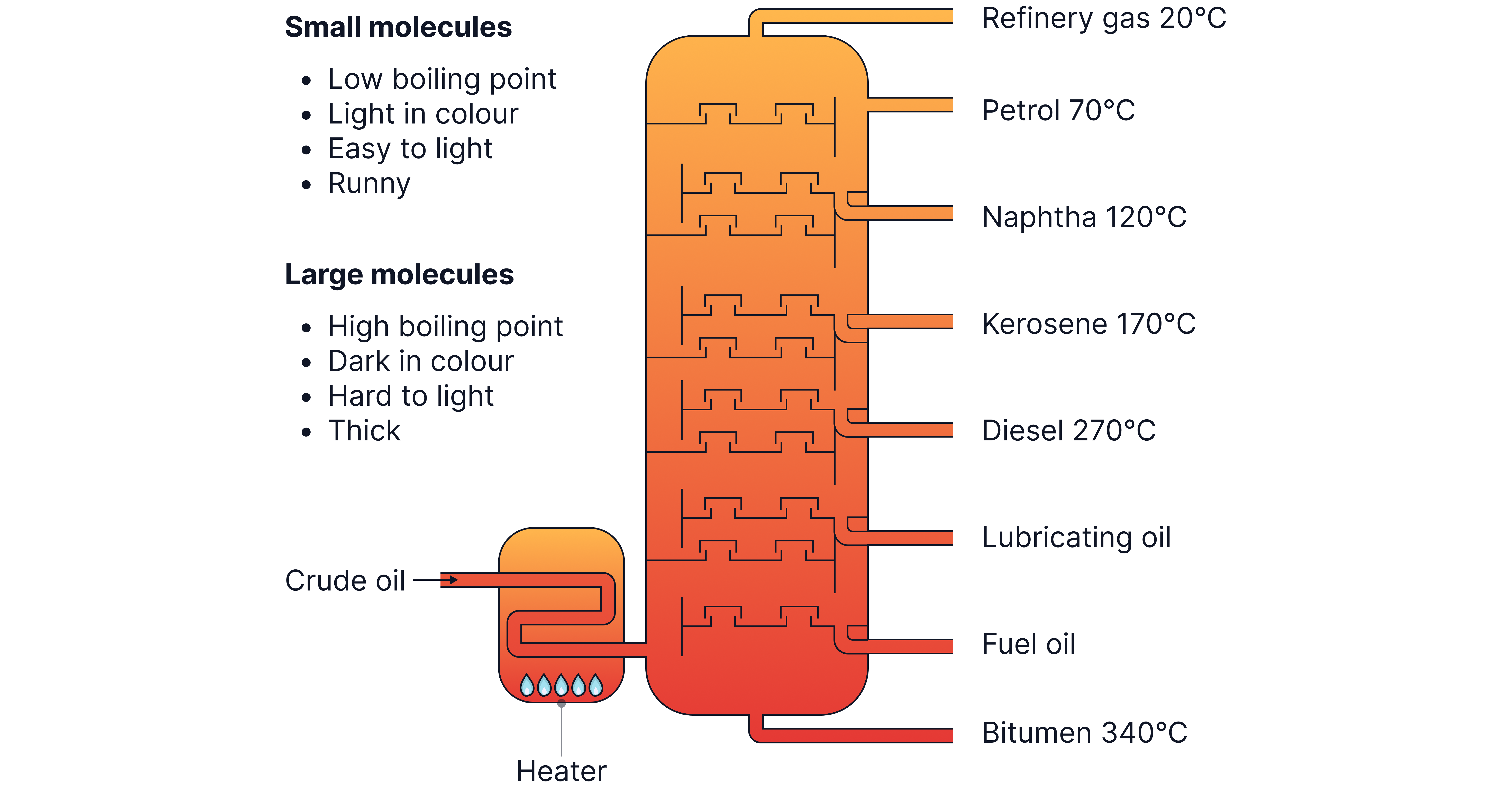
Cracking helps in converting unreactive, less useful large alkanes from fractional distillation into smaller, more useful hydrocarbons.
Two main types of cracking:
Thermal Cracking
Catalytic Cracking
Explanations
A. Thermal Cracking
Conditions: High temperature (typically ~900 °C) high pressure (up to 70 atm).
Products:
Produces high proportion of alkenes, and some shorter-chain alkanes.
Example:
B. Catalytic Cracking
Conditions: Lower temperature (~450°C) and normal pressure.
Catalyst: Zeolite (aluminosilicate), honeycomb structure for large surface area.
Products:
Produces branched alkenes, cycloalkanes and aromatic hydrocarbons for motor fuels.
More economical and efficient in producing desired fuels.
Why Cracking Is Important
Balances supply and demand of shorter-chain hydrocarbons.
Produces alkenes for:
Polymer manufacture (e.g. ethene → polyethene).
Bulk chemicals (e.g. alcohols, detergents).
Worked Examples
Example 1 – Write an equation for the thermal cracking of decane () to form ethene and one other hydrocarbon.
Answer:
Example 2 – State one condition and one product for catalytic cracking.
Answer:
Condition: 450 °C, zeolite catalyst.
Product: Branched alkanes or aromatic hydrocarbons.
Practice Questions
Question 1
Which product is more likely formed in catalytic cracking?
A. Ethene
B. Propene
C. Benzene
D. Dodecane
Answer:
C. Benzene (aromatic hydrocarbon)
Question 2
Write an equation for the catalytic cracking of dodecane to form hexane and an alkene.
Answer:
Question 3
State and explain one economic reason why cracking is important in the petroleum industry.
Answer:
Cracking converts large hydrocarbons from fractional distillation of crude oil into shorter-chain alkanes, which are in higher demand for petrol production, increasing profitability.
Key Tips and Reminders
Common Mistake: Confusing catalytic cracking (which produces aromatic hydrocarbons) with thermal cracking (which mainly produces alkenes).
Remember the general formulae for alkanes and alkenes to easily identify them in cracking equation.
Writing equations for cracking just depends on conservation of mass. Ensure the carbons and hydrogens are the same on both sides of the arrow.