Fundamental Particles
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
The atomic model has evolved over time as scientific discoveries have improved our understanding of atomic structure. Modern atomic theory describes atoms as consisting of a dense, positively charged nucleus surrounded by negatively charged electrons in specific energy levels.
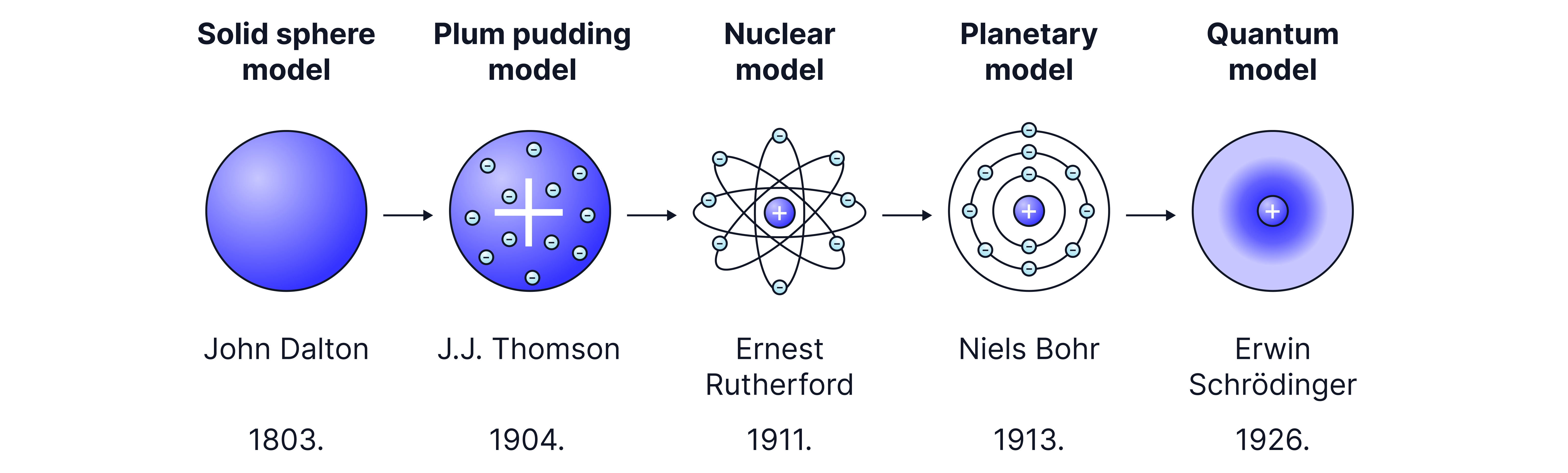
Evolution of the model for atomic structure
Solid Sphere Model, John Dalton in 1803 : Atoms theorised as indivisible spheres.
Plum Pudding Model, JJ Thomson in 1904 : Initially thought that atoms consisted of a sphere of positive charge with small negative charges (electrons) distributed evenly within it.
Rutherford’s Nuclear Model, 1911: Gold foil experiment resulted in the theory that atoms have a small, dense, positively charged nucleus at the centre with electrons moving around it. Most of the atom is empty space.
Bohr’s Quantised Shell Model, 1913: Refinement to Rutherford’s model proposing electrons orbit the nucleus in fixed energy levels –shells –and can move between these energy levels by absorbing or emitting energy.
Quantum model, 1926: Schrodinger and Heisenberg further developed atomic theory by introducing quantum mechanics. Instead of fixed orbits, electrons are wave-particles that exist in probabilistic regions called orbitals.
Fundamental Particles
Protons determine the atomic number and the identity of the element.
Neutrons contribute to the mass of the atom but do not affect chemical properties.
Electrons are responsible for chemical bonding and reactivity.
Particle | Relative Charge | Relative Mass | Location |
Proton | +1 | 1 | Nucleus |
Neutrom | 0 | 1 | Nucleus |
Electron | -1 | 1/1840 | Orbitals around the nucleus |
Atomic Number and Mass Number
Atomic Number (Z) = Number of protons in the nucleus.
Mass Number (A) = Total number of protons and neutrons in the nucleus.
Number of Neutrons = Mass Number – Atomic Number
Example Calculation
For Lithium
Atomic number = 3 (so 3 protons)
Mass number = 7
Number of neutrons = 7 - 3 = 4
Number of electrons = 3 (in a neutral atom)
Isotopes
- Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
-
Since they have the same number of electrons, isotopes exhibit identical chemical properties but may have different physical properties such as mass and density.
Example: Hydrogen Isotopes
| Isotope | Protons | Neutrons | Electrons |
| Hydrogen |
1 | 0 | 1 |
| Deuterium |
1 | 1 | 1 |
| Tritium |
1 | 2 | 1 |
Practice Question
Give two differences between the modern model of an atom and Rutherford’s model depicted below.
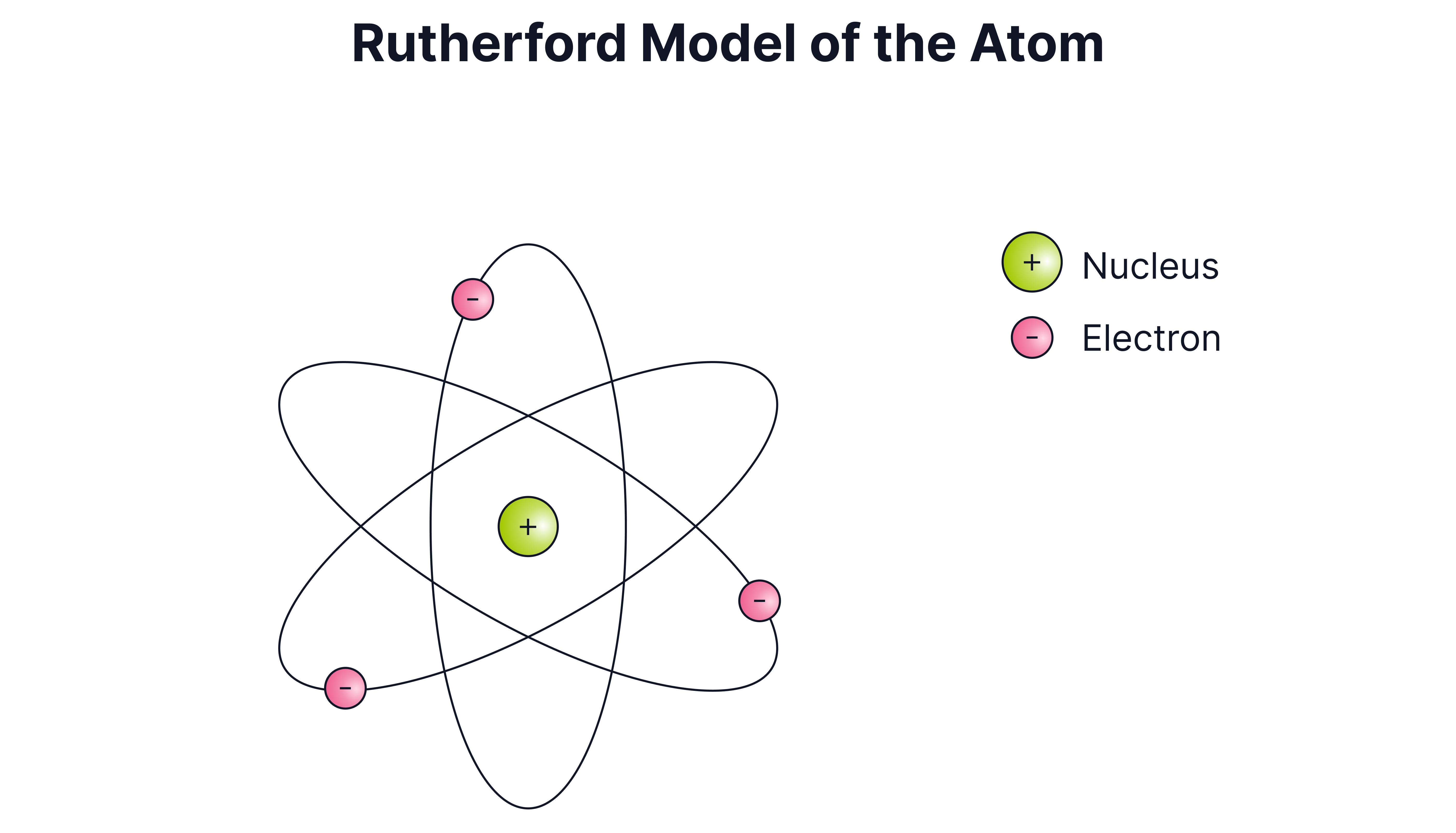
Answer
In the modern model
The nucleus is made up of two subatomic particles (protons and neutrons)
In the current model
The electrons orbit the nucleus at fixed energy levels/specific distances)