Time of Flight Mass Spectrometry
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Mass spectrometry is a technique used to determine the atomic or molecular mass of substances by converting them into ions and analysing their mass-to-charge ratio .
As the charge of the ions is typically just 1+, the mass to charge ratio is typically equal to the mass of the ion
Time of Flight (TOF) Mass Spectrometry is a specific type of mass spectrometry that determines the mass of ions based on the time taken to travel through a flight tube.
The relative atomic mass displayed on the periodic table represents a weighted average of all the naturally occurring isotopes of an element. This value is calculated based on the mass and relative abundance of each isotope. This information comes from the results of time of flight (TOF) mass Spectrometry.
A mass spectrum displays the different isotopes of an element and their relative abundances (percentage or ratio). The peaks on the graph correspond to the mass-to-charge ratio of each isotope, with taller peaks representing more abundant isotopes.
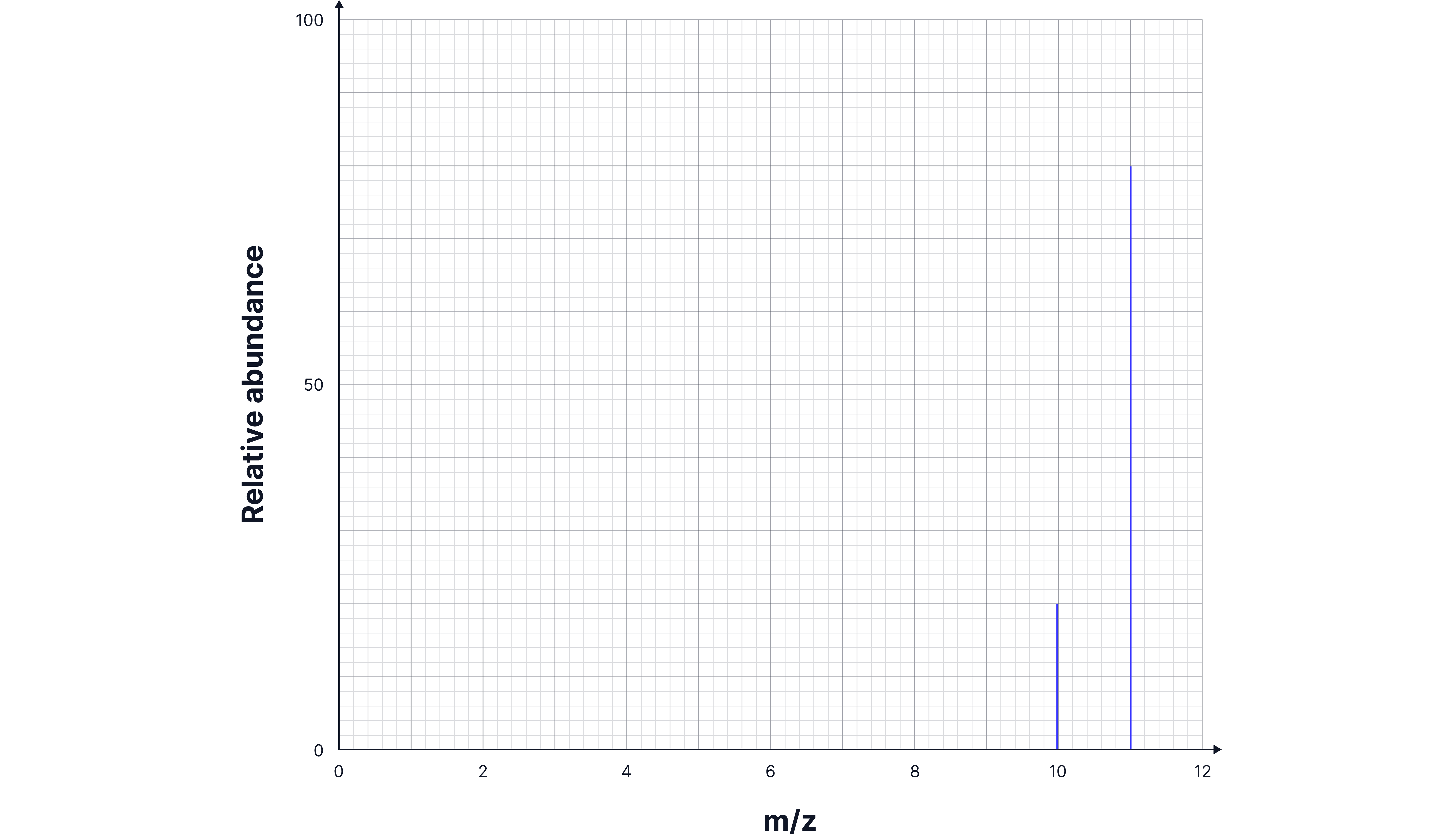
For example, the mass spectrum for Boron shows the following isotope abundances:
More detail on relative atomic mass calculations can be found in the ‘Isotopes and Relative Atomic Mass’ note
Process of Time of Flight (TOF) Mass Spectrometry
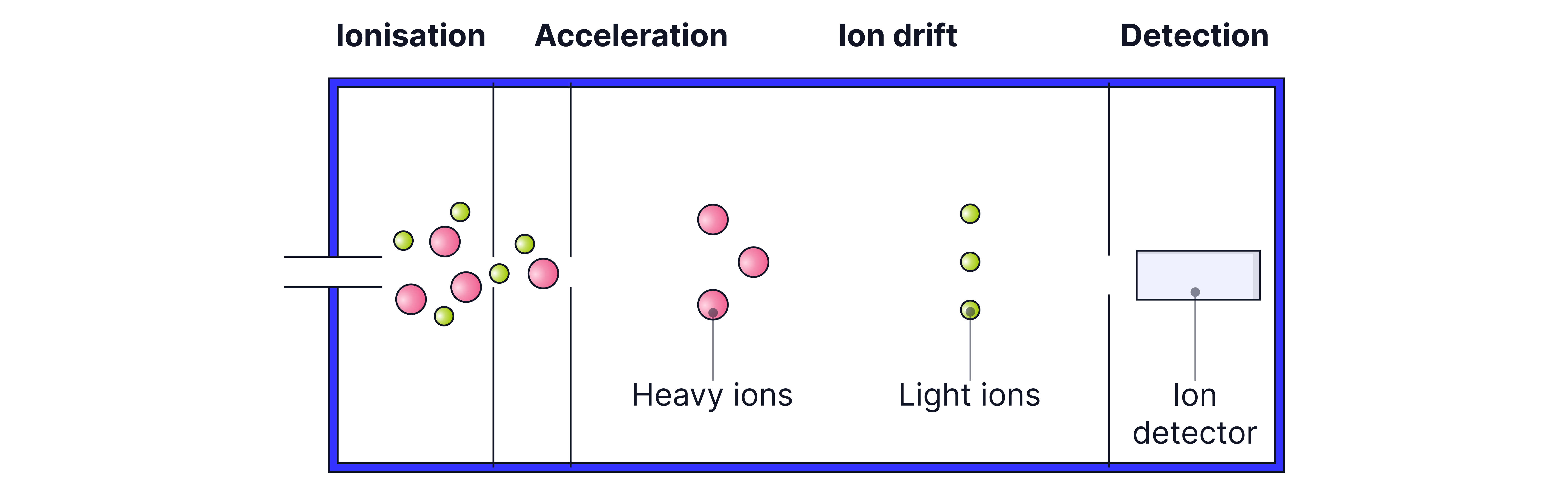
Vacuum Requirement:
A vacuum ensures that the ions travel freely through the machine without colliding with air molecules, preventing loss or deflection of particles.
The process involves four main stages:
Stage 1: Ionisation
Two methods are commonly used:
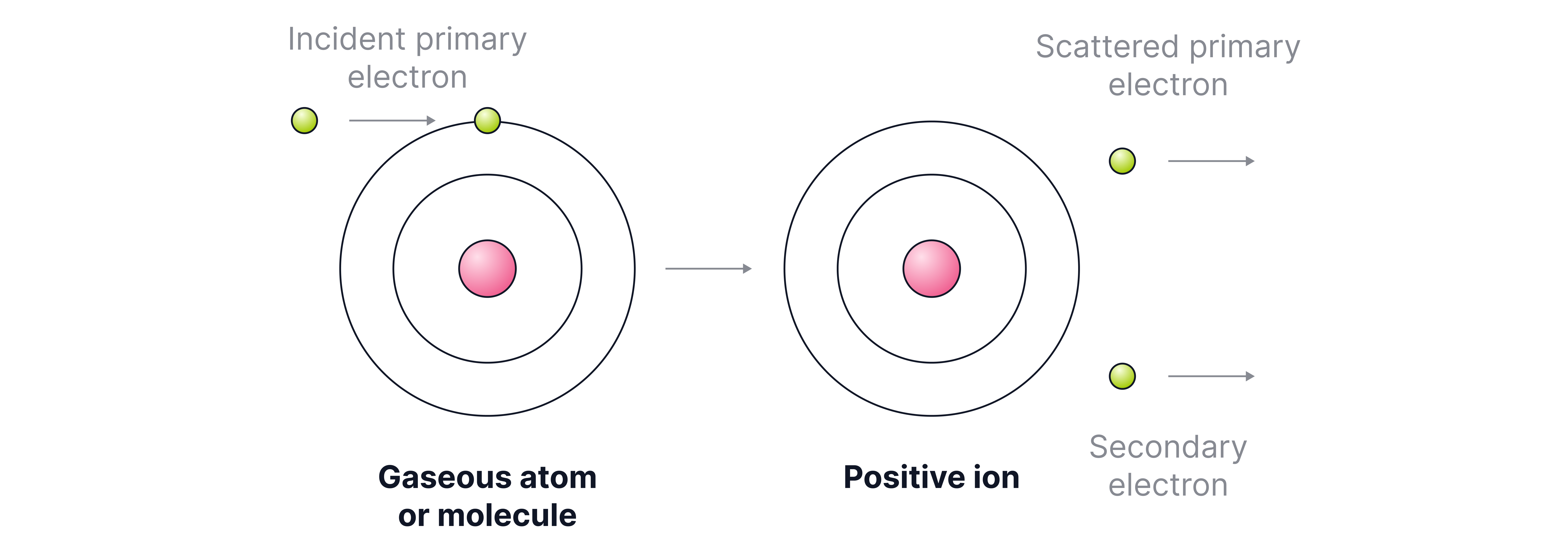
(i) Electron Impact (used for elements and small molecules):
1. The sample is vaporised and bombarded with high-energy electrons.
2. This removes an electron, forming positive ions.
Equation:
Net equation:
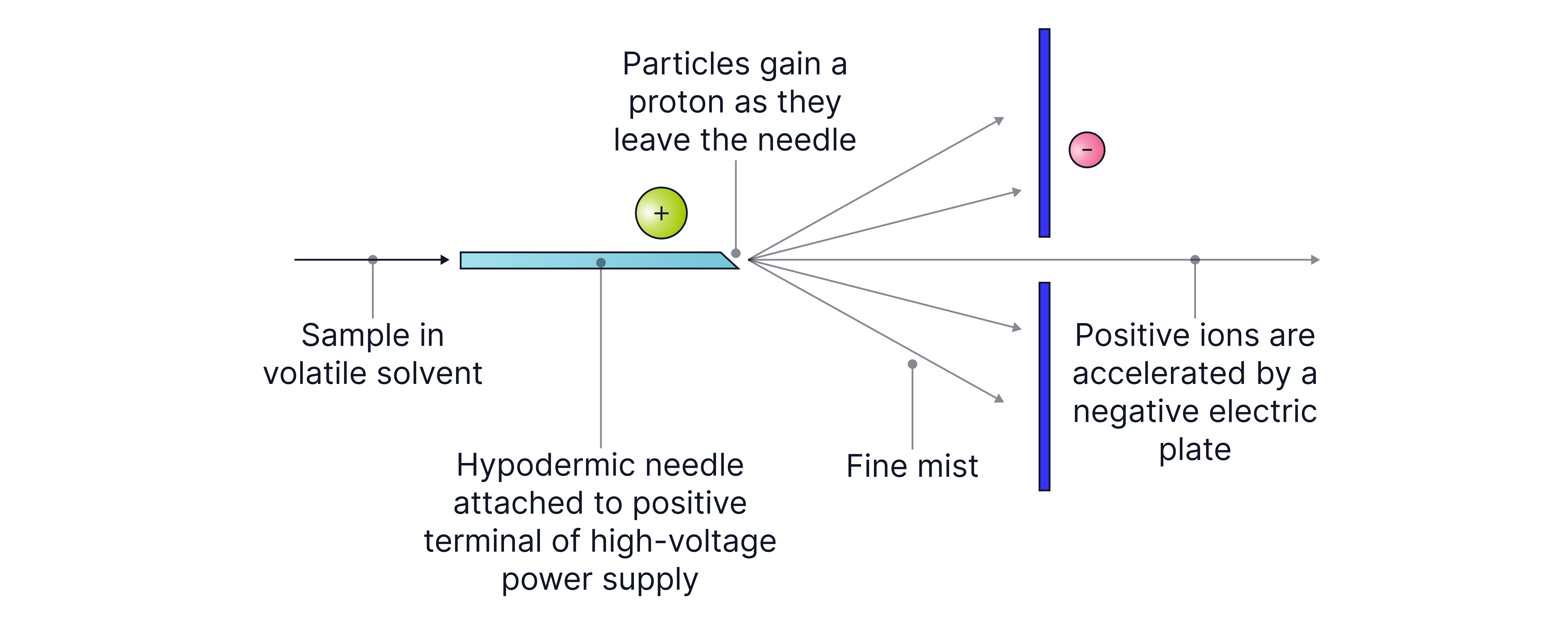
(ii) Electrospray Ionisation (used for larger molecules (e.g., proteins, pharmaceuticals).
1. The sample is dissolved in a polar solvent and passed through a fine needle at high voltage.
2. It gains a proton from the solvent to form ions.
Equation:
Stage 2: Acceleration
Positive ions are accelerated by an electric field, ensuring they have constant kinetic energy.
kinetic energy
mass of particle
velocity of particle
If we rearrange this for v we get:
Stage 3: Ion Drift (Flight Tube)
Ions move through a drift region where no forces act on them.
Lighter ions move faster, while heavier ions move more slowly.
Formula for Time of Flight (TOF):
Where:
time
distance
velocity
Combining (2) and (3) we get:
Stage 4: Detection
Since KE is constant for all ions, ions with lower mass take less time to reach the detector.
When the ions hit the detector, they gain an electron, producing a current.
The size of the current is proportional to the abundance of the ion.
Worked Example
A substance Z is dissolved in a volatile polar solvent and is ionised using electrospray ionisation.
The ions produced are accelerated, achieving a kinetic energy of .
The kinetic energy of an ion is given by the equation where:
mass /
speed /
The ions travel through a long flight tube, with a time of flight recorded as .
Calculate the mass, in grams, of one mole of Z.
Avogadro’s constant is.
Answer
1. Rearrange for
2. Calculate V
3. Calculate m in kg
4. Convert to
Practice Questions
Question 1
An ion travels through a flight tube with an energy of .
The ion takes to reach the detector.
Calculate the length of the flight tube in metres.
Answer
Question 2
A titanium sample was ionised through electron impact in a TOF mass spectrometer. The table below provides data from the mass spectrum for the titanium isotopes present in the sample.
| 46 | 47 | 48 | 49 |
Percentage Abudance | 9.3 | 6.5 | 73.2 | 11.0 |
Write a balanced equation, including state symbols, to demonstrate how a titanium atom is ionised using electron impact . Additionally, provide the formula for the ion that would be detected first.
Answer
Iorn detected first (highest ion)
If asked to identify a peak, always provide the species with its charge and mass number.