General Properties of Transition Metals
Dr. Davinder Bhachu
Teacher

Contents
The Characteristics Properties of Transition Metals
Complex formation
Formation of coloured ions
Variable oxidation states
Catalytic activity
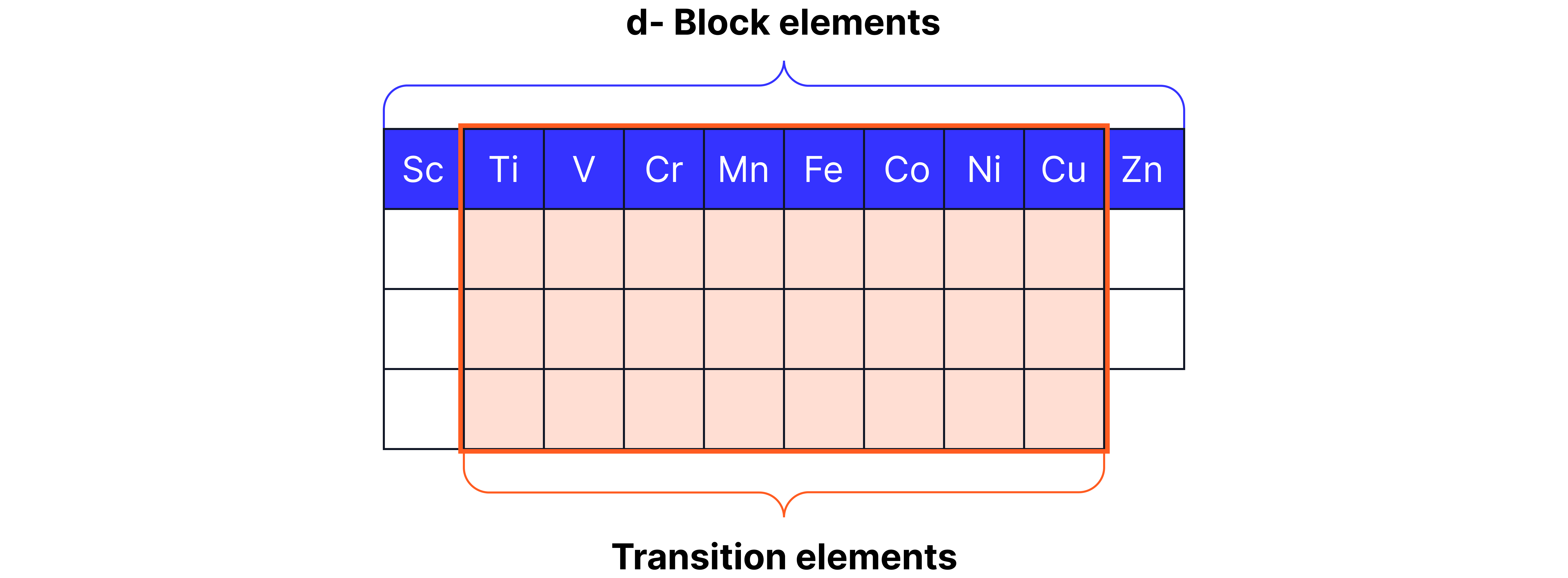
d-block vs Transition Metals Elements Question
d-block vs Transition Metals
d-block Elements
Elements where the -orbital is being filled.
Located in the central block of the periodic table, typically from Scandium to Zinc .
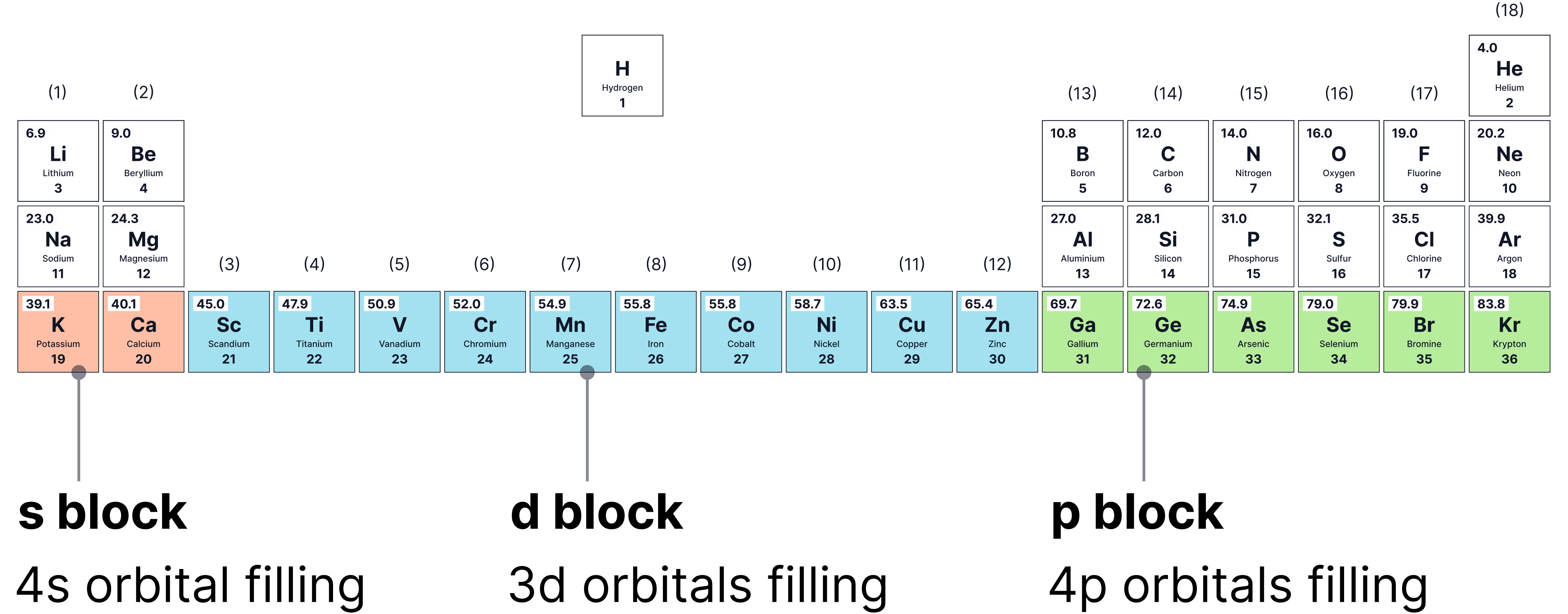
Electron Configurations of First-Row d-Block Elements ( to )
Element | Atomic Number | Electron Configuration |
Scandium (Sc) | 21 |
|
Titanium (Ti) | 22 |
|
Vanadium (V) | 23 |
|
Chromium (Cr) | 24 | |
Manganese (Mn) | 25 |
|
Iron (Fe) | 26 |
|
Cobalt (Co) | 27 |
|
Nickel (Ni) | 28 |
|
Copper (Cu) | 29 |
|
Zinc (Zn) | 30 |
|
Why Chromium and Copper are Exceptions:
Chromium :
Prefers a half-filled d sub-shell which gives extra stability due to symmetrical electron distribution and reduced electron repulsion.Copper :
Prefers a fully filled d sub-shell , which is more stable than the expected configuration.
Transition Metals
Definition: A transition metal is a d-block element that forms at least one ion with an incomplete sub-shell.
Element | d-block? | Transition Metal? | Explanation |
Scandium | ✓ | ✖ | has a configuration (empty sub-shell) |
Titanium | ✓ | ✓ | has a configuration |
Zinc | ✓ | ✖ | has a configuration (full sub-shell) |
Exam Tip:
When writing electron configurations for transition metals, always consider:
Full configuration if required (not just shorthand)
Ionic forms (e.g., , not – remove electrons first)
Characteristic Properties of Transition Metals
Complex Formation
Transition metals form complex ions by accepting lone pairs from ligands.
A ligand is an ion or molecule that donates a lone pair of electrons to a central metal ion to form a coordinate bond.

Example: In the complex ion , a ion is surrounded by six water ligands, forming an octahedral shape.
Formation of Coloured Ions
Transition metal ions with partially filled -orbitals can absorb visible light, promoting electrons between split -orbital energy levels in a complex.
The observed colour is the complement of the wavelength absorbed.
Factors affecting colour:
Identity of metal
Oxidation state
Type of ligands
Coordination number
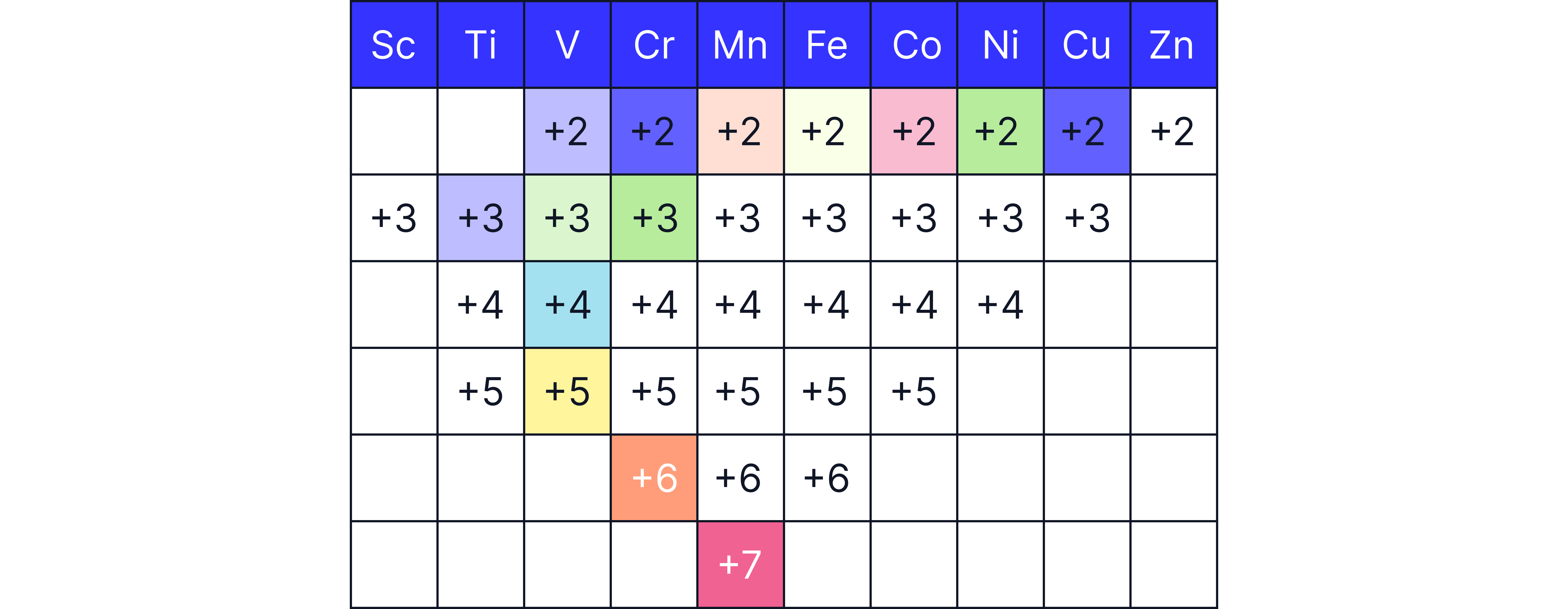
Variable Oxidation States
Transition metals exhibit multiple stable oxidation states, because the energy gap between the and orbitals is small. This allows electrons to be lost from both the and sub-shells.
Examples:
Iron: and
Manganese:
Chromium:
Note: The oxidation state (loss of two electrons) is common across many first-row transition metals.
Because of these variable oxidation states, Roman numerals are used in the names of compounds to indicate the oxidation state of the metal cation.
Examples:
Iron sulfate → contains
Iron chloride → contains
Tip: Always include the Roman numeral in the name when a metal can form more than one stable ion.
Catalytic Activity
Transition metals and their compounds act as catalyst due to:
Ability to change oxidation states, enabling electron transfer
Availability of partially filled -orbitals to adsorb reactants on their surface
Examples:
Iron in the Haber process:
Vanadium oxide in the Contact process:
Manganese oxide catalyses hydrogen peroxide decomposition: →
Transition metals and their compounds act as catalyst due to:
Question 1
Explain why zinc is not classified as a transition metal.
Answer:
Zinc only forms , which has a full sub-shell. Transition metals must form at least one ion with a partially filled d sub-shell, which does not.
Question 2
State three properties of cobalt.
Answer:
Any 3 of the following:
Complex formation
Formation of coloured ions
Variable oxidation states
Catalytic activity
Summary Table
Property | Explanation |
Complex formation | Metal ions accept lone pairs from ligands via coordinate bonds |
Coloured ions | Due to electron transitions when -orbitals split in complexes |
Variable oxidation states | Similar energy levels of and orbitals allow multiple stable states |
Catalytic activity | Transition metals can vary their oxidation states or adsorb reactants on their surface |
Key Tips and Reminders
Make sure you can explain which d-block elements are not transition metals .
electrons are lost BEFORE .
When asked for the characteristics properties of a specific transition metal, simply state the general properties of all transition metals.