Complex Ions
Lajoy Tucker
Teacher

Contents
Introduction
A complex ion consist of a central metal ion bonded to surrounding ligands via coordinate bonds.
A ligand is a molecule or ion that donates a lone pair of electrons to the metal ion.
The coordination number os the number of coordinate bonds formed to the metal ion.
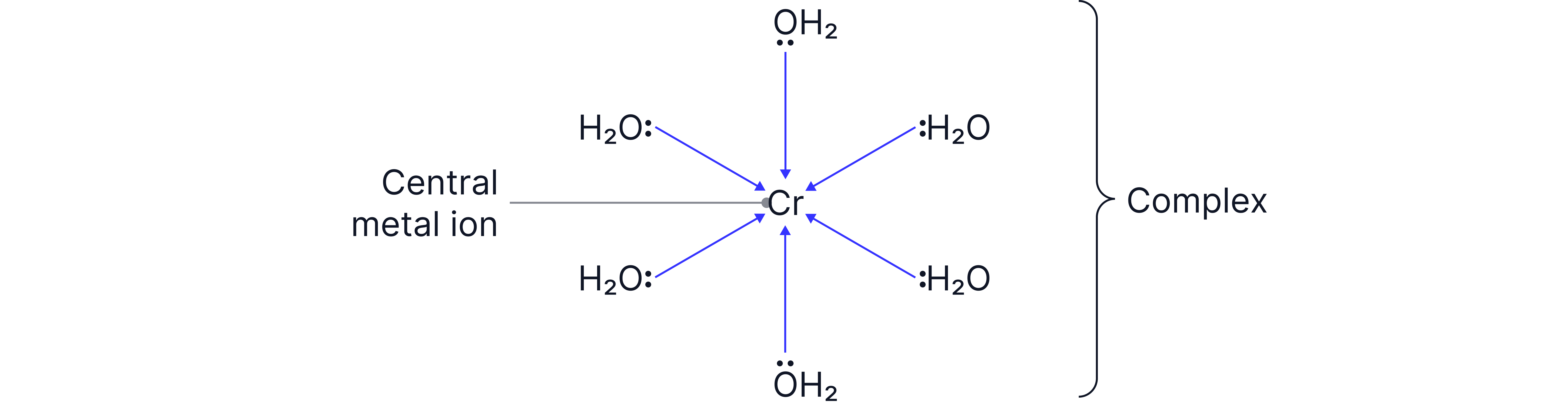
Shapes of Complex Ions
Octahedral Complexes (Coordination Number = 6)
Most common with small, uncharged ligands like and .
Six ligands arranged around the central ion.
Example:
- iron (III) hexaaqua complex
- distorted octahedral
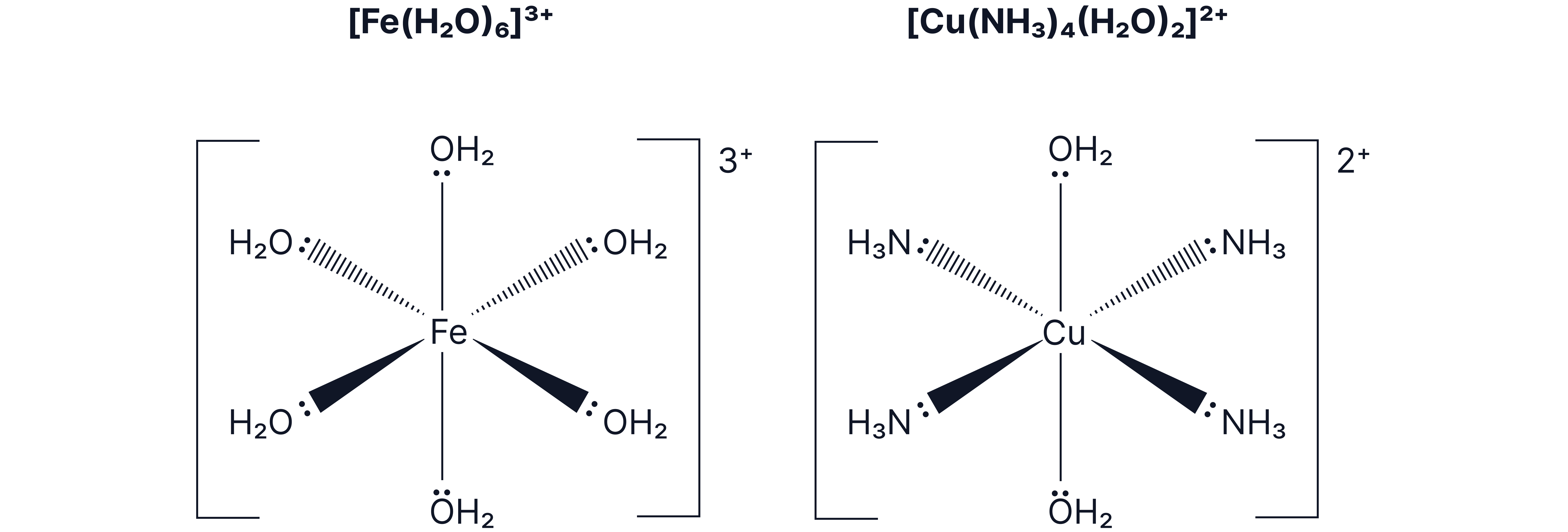
Bond angle:
Tetrahedral Complexes (Coordination Number = 4)
Often form with larger ligands, such as CI, as fewer ligands can fit around the central metal atom or ions.
Example:
![Diagram of the tetrahedral complex ion [CuCl₄]²⁻ showing a central Cu²⁺ ion bonded to four chloride ligands with wedge and dashed bonds indicating the 3D arrangement.](https://cdn.myedspace.co.uk/images/04e58f59-a6b3-48bc-9761-37237dfed0a4.png)
Bond angle:
Square Planar Complexes (Coordination Number = 4)
Typically seen with transition metals like platinum .
Example:
Cisplatin, - used in cancer treatment.

Bond angle:
Linear Complexes (Coordination Number = 2)
Rare, typically formed by silver and copper .
Example:
- used in Tollens' reagent

Shape is linear, bond angle =
Charges of Complex Ions
The charge of a complex ion can be deduced by considering the oxidation state of the central metal ion along with the charges of ligands attached.
The oxidation state of the central metal atom or ion can be deduced if the charge of the complex is known.
The charge is included outside of square brackets.
Example:
![Worked examples showing how to calculate the oxidation states of transition metals in complex ions, including Cu in [CuCl₄]²⁻ and Cr in a chromium complex ion.](https://cdn.myedspace.co.uk/images/6eb8fe30-5baa-49fb-afef-87193d683bfa.png)
Ligand Types and Denticity
Denticity refers to how may coordinate bonds a ligand can make to an indicidula metal atom or ion.
|
Ligand |
Denticity |
Example |
|
H₂O, NH₃, Cl⁻ |
Monodentate |
|
|
|
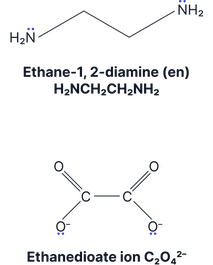
Bidentate (Donate 2 lone pairs) |
[Co(H2NCH2CH2NH2)3]2+
|
|
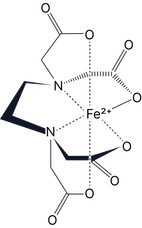
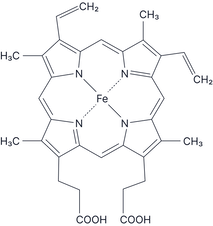
EDTA⁴⁻ (hexadentate) Haem (tetradentate) |
Multidentate* (Donates more than two lone pairs) |
|
You will not be expected to draw complexes with multidentate ligands.
Isomerism in Complex Ions
Transition metal complexes can show stereoisomerism:
Cis-Trans Isomerism (Geometrical)
Occurs in:
Octahedral complexes with 4 of one type and 2 of another type of monodentate ligand (e.g. ), or a pair of bidentate ligands.
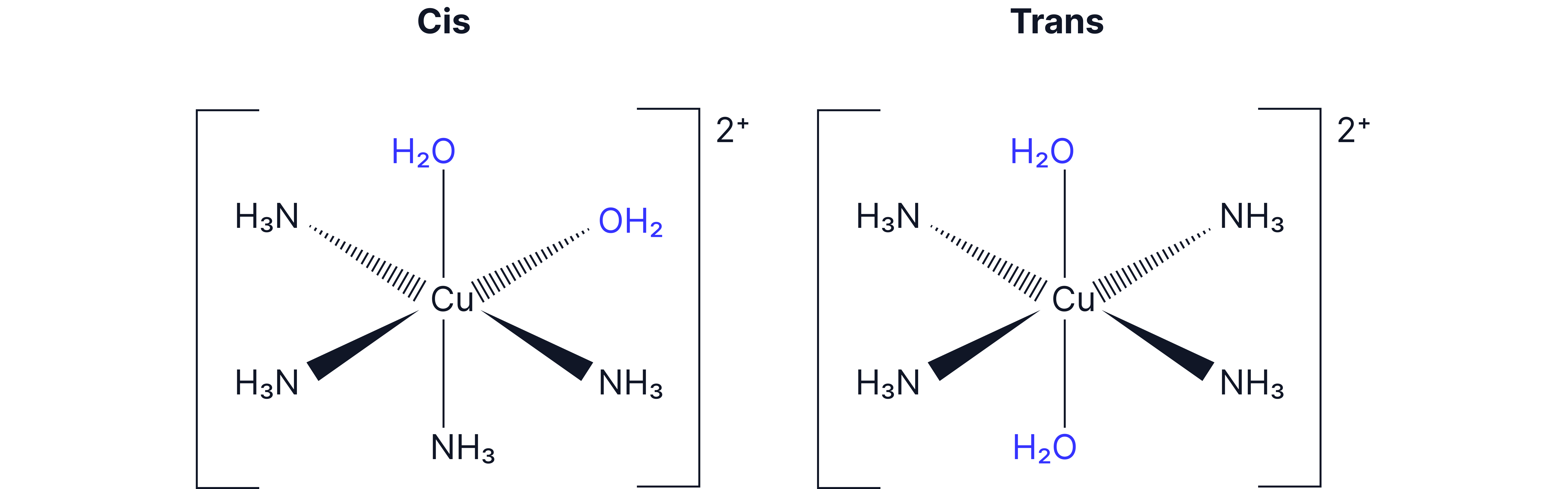
Square planar complexes with 2 pairs of ligands attached to the central metal ion, like cisplatin:
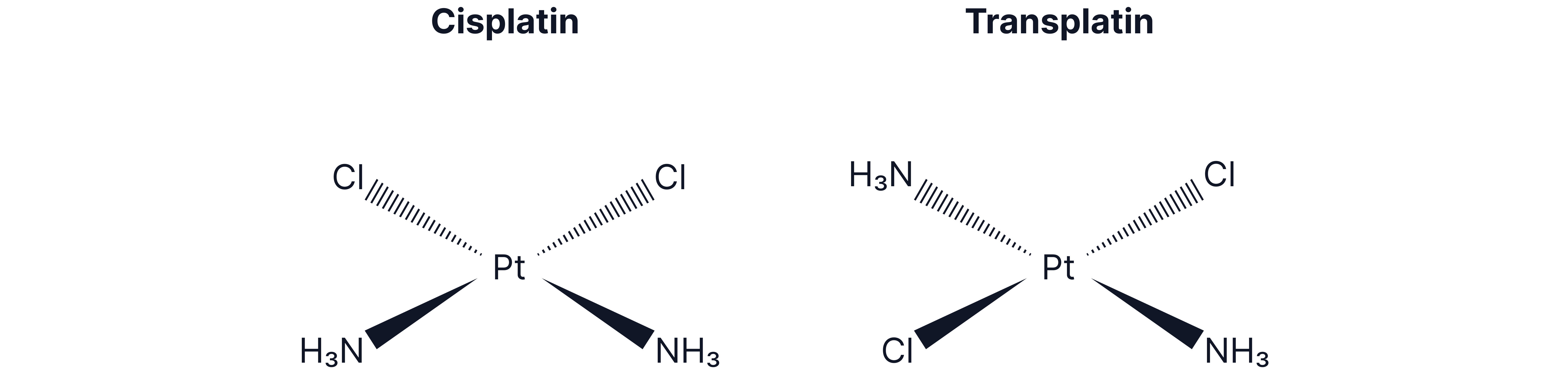
Cisplatin- :
Cis form is active as an anti-cancer drug.
Transplatin is inactive.
Optical Isomerism (Enantiomers)
These form non-superimposable mirror images (like left and right hands)
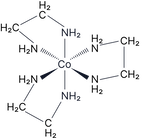
Occurs in octahedral complexes with two or more bidentate ligands, such as:
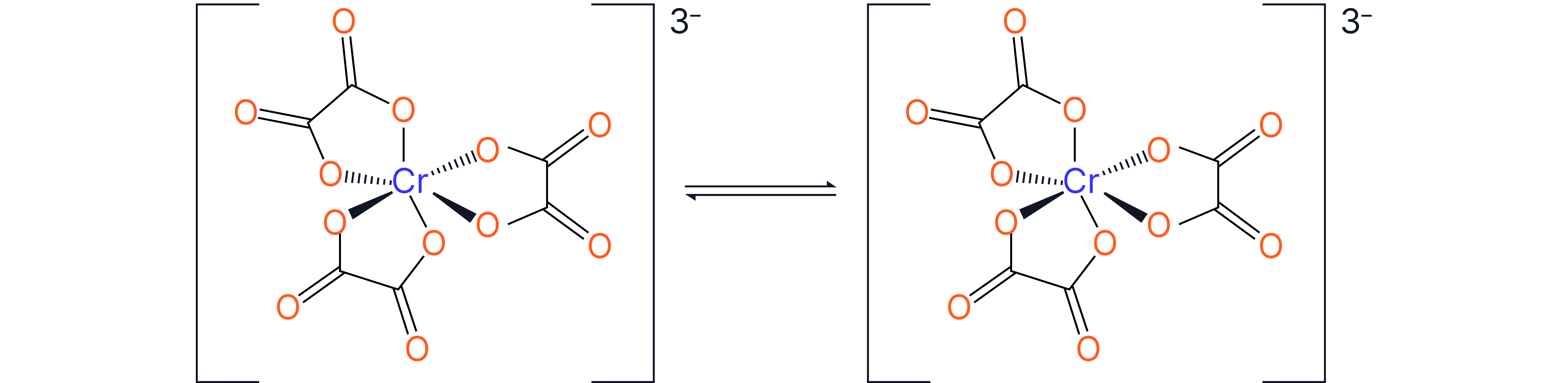
Complexes with two bidentate and two monodentate ligands show cis/trans and optical isomerism. For example:
![Displayed formula of the octahedral complex ion [Fe(C₂O₄)₂(H₂O)₂]⁻ showing an Fe ion bonded to two bidentate oxalate ligands and two water ligands.](https://cdn.myedspace.co.uk/images/525116d8-83cc-41e7-9f8a-f423035ebbf2.png)
Named Complexes to Know
Tollens' test:
Aldehyde reduces to metallic silver:
This gives a silver mirror on the test tube.
Question 1:
What shape would you expect for the complex ?
Answer:
Octahedral, because six small monodentate ligands coordinate with the ion.
Question 2:
Deduce the oxidation state of in
Answer:
Each ligand has a charge of (the bidantate ethanedioate ligand) contributing .
The overall charge of the complex is
Summary Table
Exam Tips
For shapes: know the coordination number and typical ligands.
Clearly label cis/trans isomers with diagrams is asked.
Complexes with the bidentate ligands ethane-1,2-diamine and ethanedioate are commonly assessed. Practice drawing these.
![1 Displayed formula of the octahedral complex ion [Cr(H₂O)₆]²⁺ showing a central chromium ion bonded to six water ligands.](https://cdn.myedspace.co.uk/images/12786f37-4551-4ca7-bea4-2bbca2dc1ba1.png)
![1 Displayed formula of the complex ion [Cr(C₂O₄)₃]³⁻ showing a chromium ion coordinated to three bidentate oxalate ligands in an octahedral arrangement.](https://cdn.myedspace.co.uk/images/22e22545-8c29-4477-aab7-648d5d7fba2a.png)