Formation of Coloured Ions
Lajoy Tucker
Teacher

Contents
Origin of Colour:
Transition metal ions with partially filled d-orbitals can form coloured compounds due to electronic transitions.
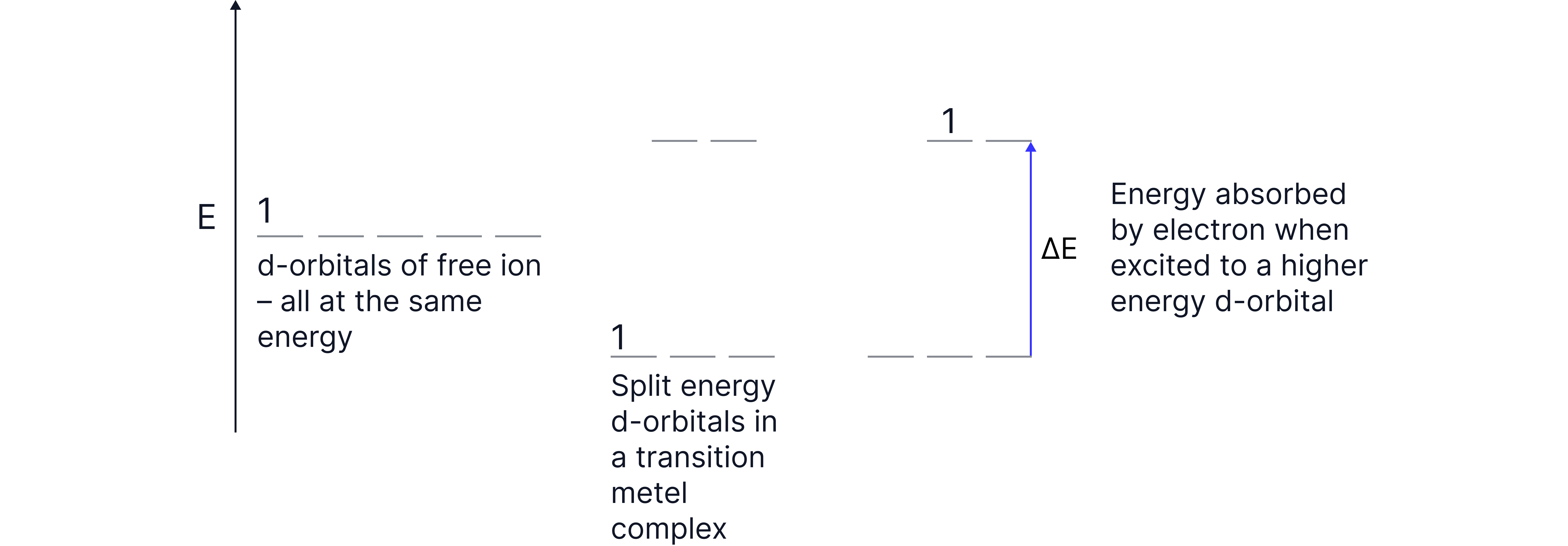
In a free ion, the five d-orbitals are degenerate (equal in energy).
When ligands approach and form a complex, the d-orbitals split into two energy levels. The orbitals are now non-degenerate.
An electron can be promoted to a higher energy d-orbital by absorbing a photon of visible light.
The energy absorbed corresponds to the specific energy difference (ΔE) between the orbitals.
This difference in energy corresponds to a certain wavelength of light
The wavelengths not absorbed are transmitted or reflected, which is the colour we see. For example, if a complex absorbs red light, it may appear green, the complementary colour.
Key Principle
The colour of a transition metal complex is due to the absorption of visible light that promotes a d electron from the ground state to an excited state. Colours not absorbed are transmitted or reflected and therefore seen.
The Energy Difference (ΔE)
The energy difference between the split d-orbitals can be calculated using:
Where:
= energy gap between d-orbitals (in joules, )
= Planck’s constant =
= frequency of absorbed light (in )
= speed of light =
= wavelength of absorbed light (in metres, )
Example Calculation
A complex absorbs light with a wavelength of 520 nm. What is the energy absorbed?
Answer
Step 1: Convert nm to m:
Step 2: Use the equation
Formation of Coloured Ions Explained
When Are Complexes Colourless?
The ion has no d electrons (e.g. is ), so no d–d transitions are possible
![Diagram showing that scandium forms Sc³⁺ ions with an empty 3d subshell ([Ar]3d⁰), so no d–d electron transitions occur and its compounds are colourless.](https://cdn.myedspace.co.uk/images/6ed68de7-e327-4d76-b04e-2335a402911a.png)
The ion has a full d sub-shell (e.g. is ), so electrons cannot be promoted between orbitals
![Diagram showing that zinc forms Zn²⁺ ions with a full 3d subshell ([Ar]3d¹⁰), so no d–d electron transitions occur and its compounds are colourless.](https://cdn.myedspace.co.uk/images/196e0d8f-b8fd-455f-9570-bd050c5ed2f7.png)
In both cases, no visible light is absorbed, so the complex appears as colourless solutions or white solids.
Factors Affecting the Colour of a Complex
The observed colour depends on the size of the energy gap between the split -orbitals. This is influenced by:
Factor | Effect on |
Metal ion | Different metals have different nuclear charges and electron arrangements, altering the extent of -orbital splitting |
Oxidation state of the central metal ion | A higher oxidation state increases the attraction between the metal ion and ligands, leading to a larger . |
Ligand type | Ligands cause different extents of -orbital splitting. For example, causes a larger than |
Coordination number and geometry | The shape of the complex (octahedral, tetrahedral, square planar) affects how -orbitals split and therefore the value of . Changing coordination number generally involves changing the ligand too. |
Example: vs
Both are complexes.
is a strong field ligand, causing a larger repulsion and larger .
absorbs higher-energy (shorter wavelength) light than .
As a result, the observed colours differ, despite being in the same oxidation state for both complexes.
Visible Light Spectroscopy & Colorimetry
Colorimetry can be used to determine the concentration of coloured transition metal ions in solution by measuring how much visible light is absorbed at specific wavelengths.
1. Transition metal complexes absorb certain wavelengths of visible light due to d–d electron transitions.
2. The more concentrated the solution, the more light is absorbed.
3. A colorimeter or spectrophotometer measures the absorbance of light at specific wavelengths.
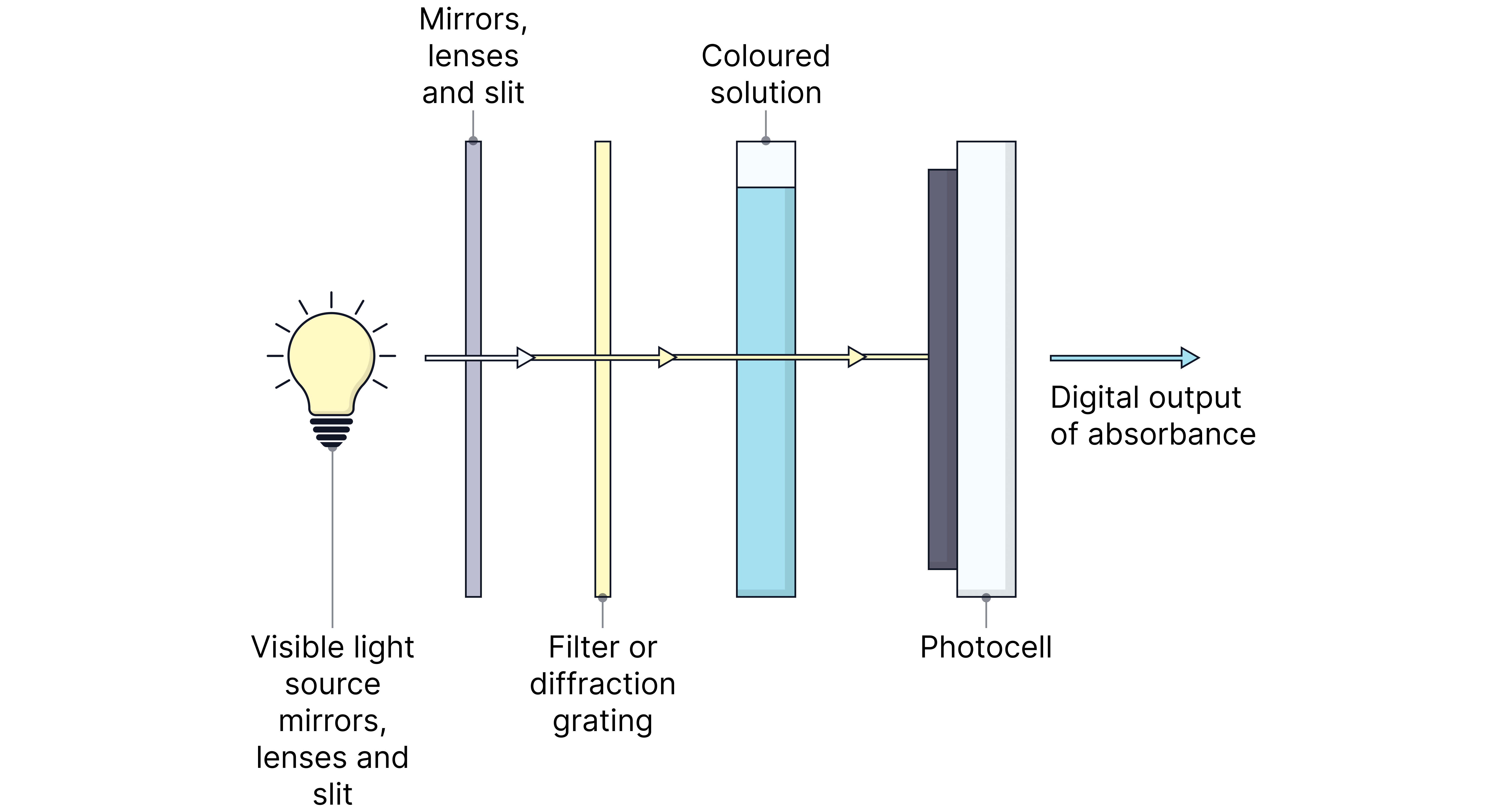
4. The absorbance is directly proportional to the concentration of the coloured ion (Beer–Lambert Law).
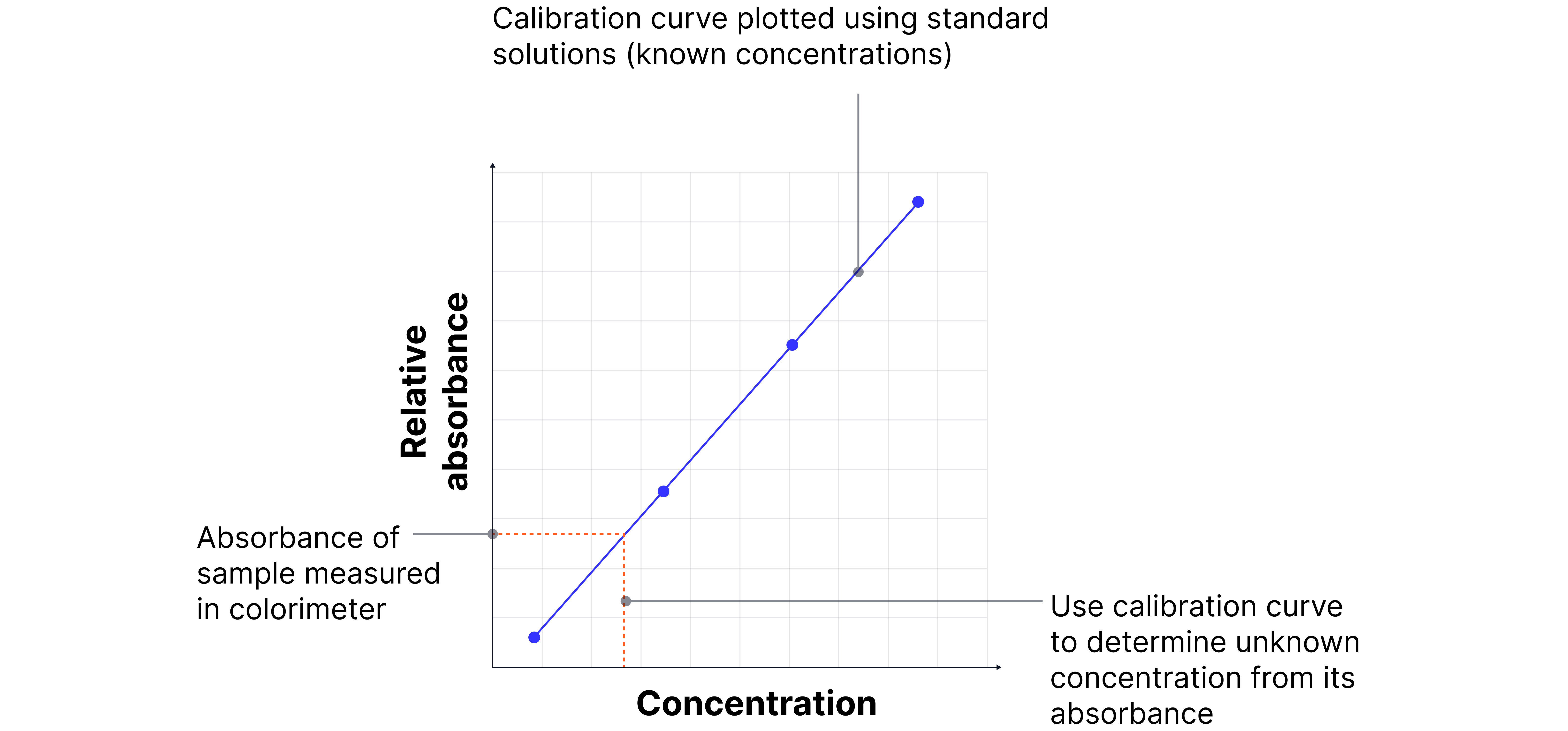
Determining unknown concentration of sample solution
1. Choose a filter (or wavelength) that corresponds to the maximum absorbance of the coloured ion (complementary colour to the solution).
E.g., a blue complex absorbs orange/red light → use a red/orange filter.
2. Zero the colorimeter using distilled water (solvent without the coloured ion).
3. Prepare standard solutions of known concentration.

4. Measure their absorbances using the colorimeter.
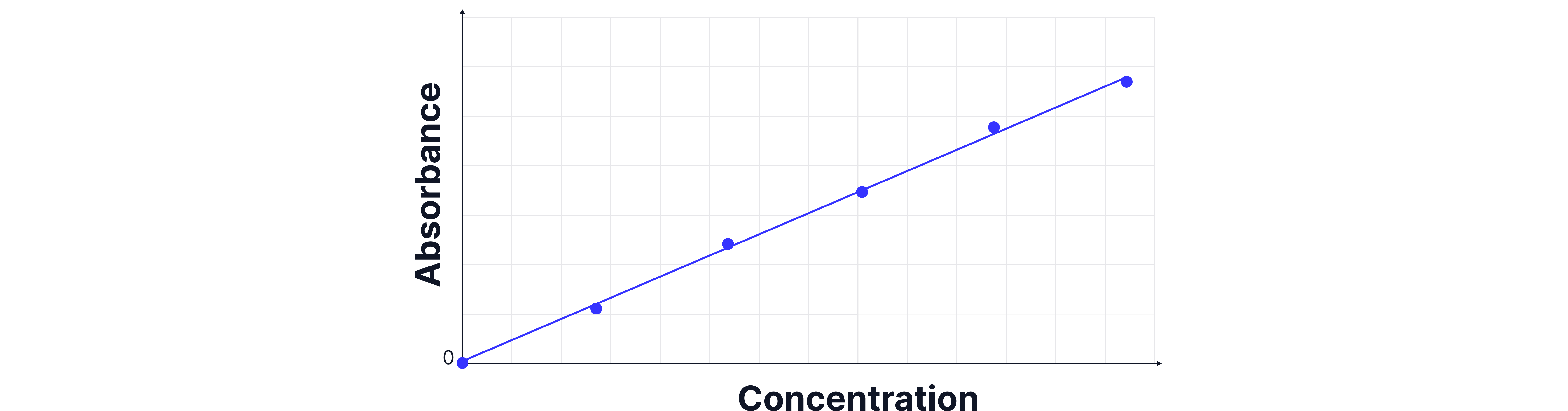
5. Plot absorbance vs concentration to create a calibration curve.
6. Measure the absorbance of the unknown sample and use the curve to determine its concentration.
Practice Questions
Question 1
Calculate the energy absorbed when a complex absorbs light of frequency
Answer
Use
Question 2
Identify the factor that leads to the different colours of and
Answer
Different ligands
Summary
Concept | Explanation |
Colour in complexes | Caused by transitions when visible light is absorbed |
formula | |
Factors affecting colour | Metal*, oxidation state, ligand type, coordination number |
No colour | (e.g. ) or (e.g. ) – no transitions |
Key Tips
Use correct units: for , for λ, for
Always refer to transitions when explaining colour in complexes
Colour changes in chemical reactions of complex ions are due to different ligands, oxidation states, or coordination number.
Be able to interpret or construct a calibration graph, and explain the experimental details for creating one