Ligand Exchange and Thermodynamics
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Introduction to Ligand Exchange and Thermodynamics
Ligand exchange is a reaction where one ligand in a complex ion is replaced by another.
This can result in:
A change in colour
A change in coordination number or geometry
Partial (incomplete) substitution
Formation of a more stable complex
Explainer Video
Ligand Exchange Reactions
Complete Ligand Exchange Without a Change in Coordination Number
The number of ligands (coordination number) remains the same after substitution.
Example:
Substitution of the water ligands in with on the addition of excess ammonia.
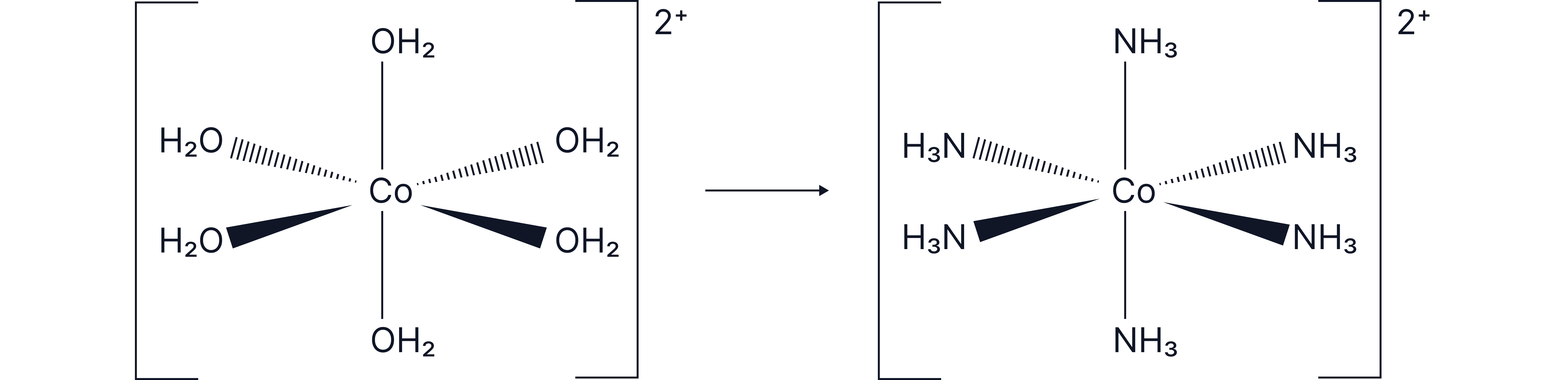
Coordination number = 6 throughout
Octahedral geometry is retained
Incomplete Ligand Exchange Without a Change in Coordination Number
The number of ligands (coordination number) remains the same after substitution.
Example:
Substitution in ⁺ occurs in two steps. The ligands are of similar sizes and so replaced gradually. Dropwise addition of NaOH gives a light blue precipitate:
The light blue precipitate then dissolves upon addition of excess ammonia to give:
The overall equation is therefore:
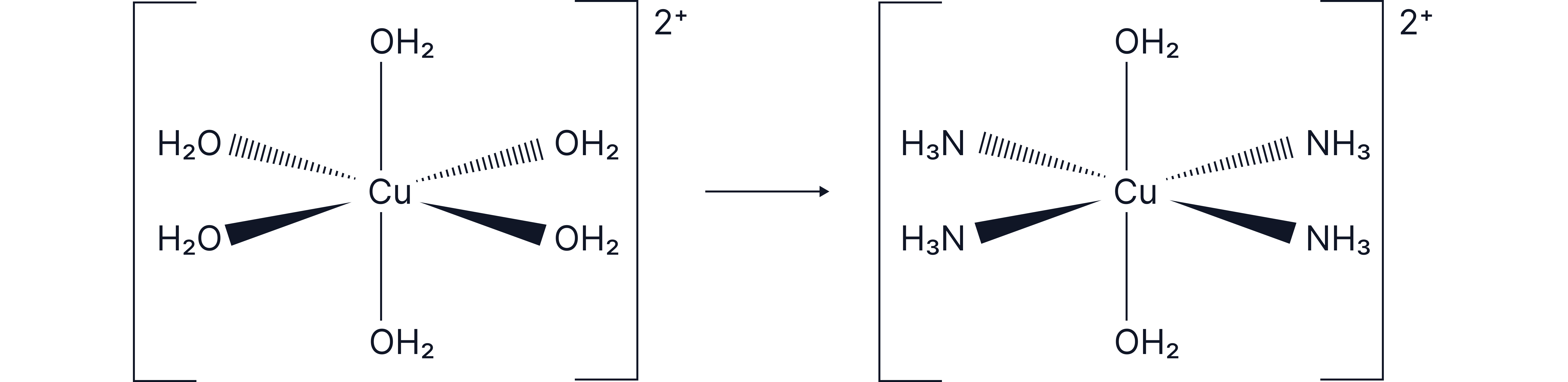
Overall colour change: pale blue solution 🡪 deep blue solution
Octahedral geometry is retained
The product exists as cis/trans isomers
Complete Ligand Exchange With a Change in Coordination Number
The new ligand may be larger or smaller, causing a change in coordination number and possibly shape.
Example:
Exchange of small ligands with large ligands in : Fewer ions can fit around the central metal ion.
Reminder – the overall charge of the ion is determined by the oxidation state of the central metal ion and the charges of the ligands.
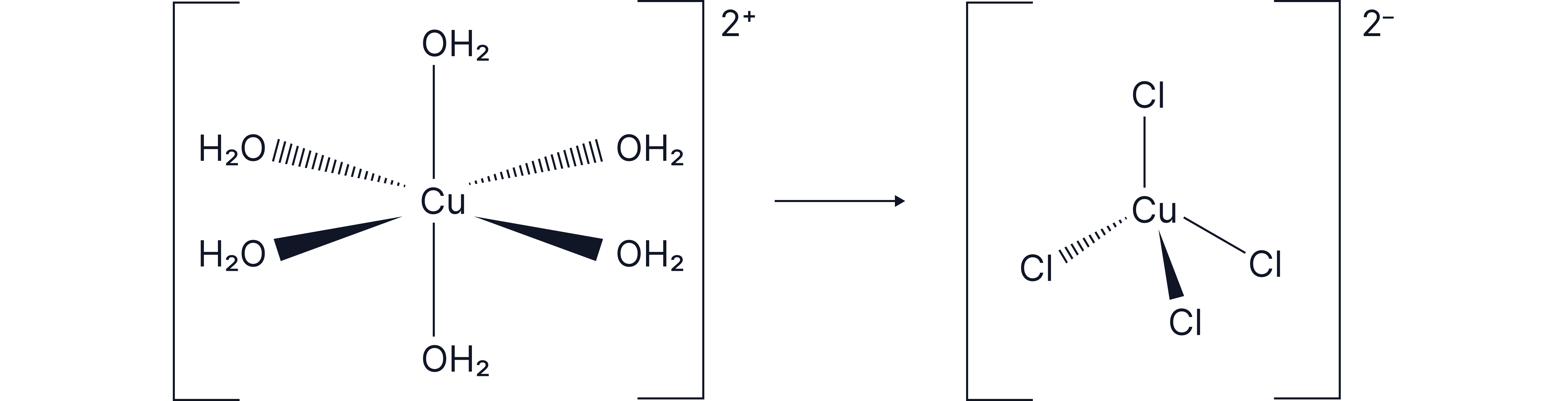

Octahedral → tetrahedral
Pale blue → yellow-green solution
Coordination number changes from 6 to 4
Note: This reaction is reversible; adding water shifts the equilibrium back.
Stability of Complexes
Ligand exchange often occurs because a more stable complex forms.
Stability depends on:
Type of ligand (e.g. multidentate ligands form more stable complexes)
Chelate effect: Complexes with bidentate or multidentate ligands are more stable than those with monodentate ligands due to increase in entropy.
Entropy is a measure of disorder or randomness. The more particles there are, the more entropy there is.
Example 1:
Copper Hexahydrate with EDTA⁴⁻
Copper hexahydrate reacts with a hexadentate ligand, forming a more stable complex because EDTA⁴⁻ binds to the copper ion via six coordinate bonds.
Reaction:
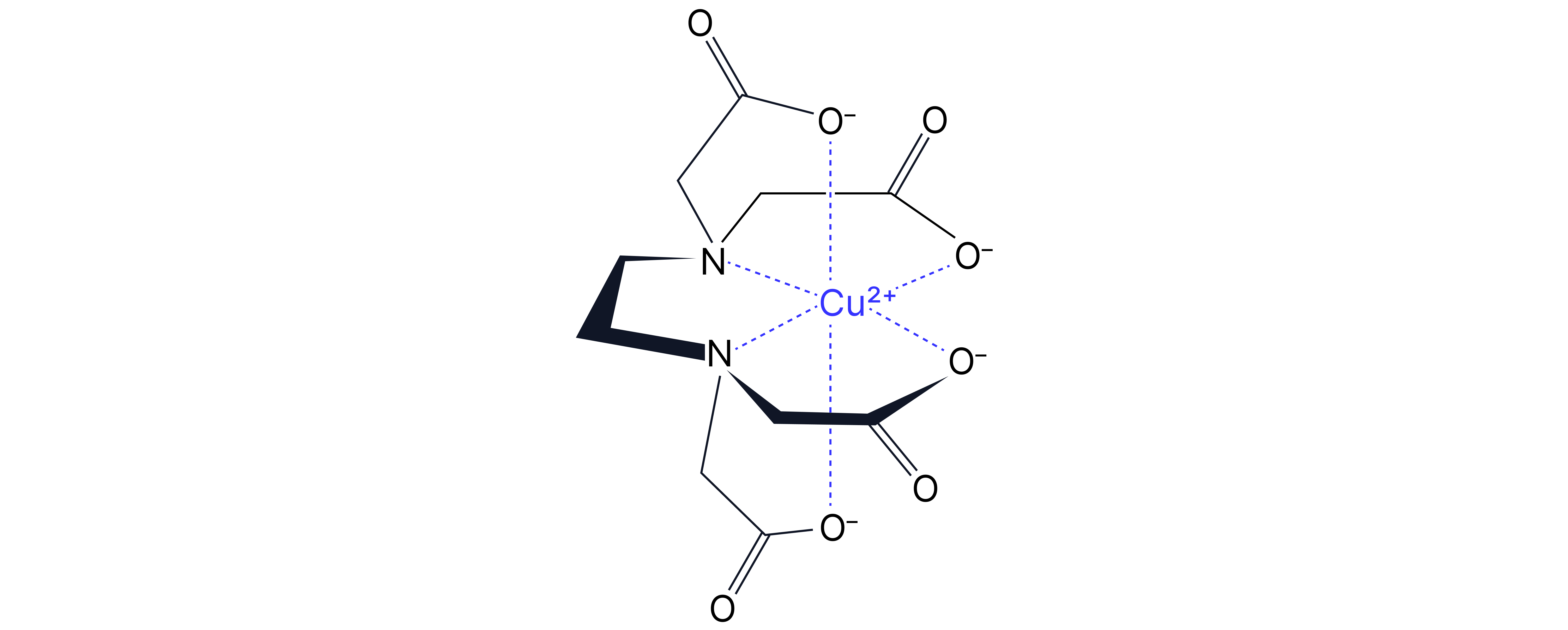
The displaces the six water ligands, forming a hexadentate complex with copper (II)
This complex is more stable due to the large increase in entropy two reactant particles become seven product particles.
Thermodynamics of Ligand Exchange
Gibbs Free Energy ()
A negative indicates a spontaneous reaction.
T is the temperature in Kelvin
Enthalpy Change ()
= bonds broken – bonds formed
Often small, because bond-breaking and bond-forming enthalpies are similar.
Entropy Change ()
Spontaneity is therefore often driven by the change in entropy
A favourable can drive the reaction even if is small or slightly positive.

Example: Replacement of six monodentate water ligands with three bidentate ethanedioate ligands
![Ligand substitution diagram showing hexaaquachromium(III), [Cr(H₂O)₆]³⁺, reacting with bidentate oxalate ligands to form the octahedral chelate complex [Cr(C₂O₄)₃]³⁻.](https://cdn.myedspace.co.uk/images/replacement-of-six-monodentate-water-ligands-with-three-bidentate-ethanedioate-ligands.png)
ΔH is negligible as the same number and same type of bonds are being broken and formed ()
ΔS is large and positive as 4 reactant particles form 7 product particles
A negligible and a positive , results in a negative and therefore a spontaneous reaction.
Haem and Oxygen Transport
Structure of Haem
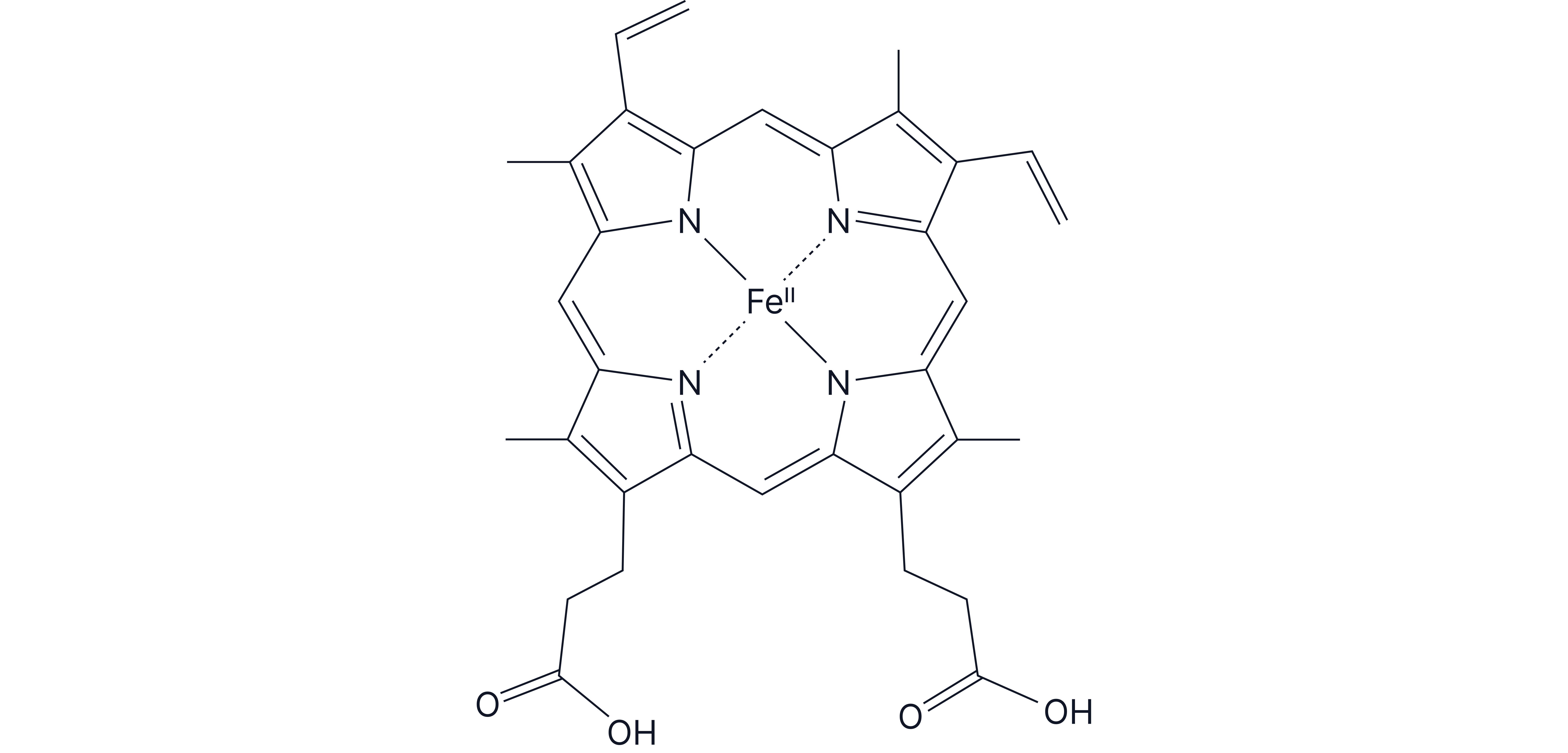
Haem is an iron(II) complex with a multidentate ligand. The central ion in haem is coordinated to four nitrogen atoms from a porphyrin ring (a large, multi-dentate ligand), and a fifth ligand is bound to the oxygen atom in haemoglobin.
is at the centre of the haem group.
The iron ion also forms a sixth coordinate bond to oxygen when oxygen binds to the haemoglobin molecule.
Haemoglobin and Oxygen Transport
In haemoglobin, the iron(II) centre in the haem group is able to form a coordinate bond with oxygen (O₂). This allows haemoglobin to bind oxygen in the lungs and release it in tissues where oxygen is needed.
How it works:
When is inhaled, it binds to the in haemoglobin, forming a coordinate bond with the iron ion in each of the four haem groups.
The iron(II) is coordinated to four nitrogen atoms in the porphyrin ring, with a fifth coordination to , enabling efficient oxygen transport via the bloodstream.
Toxicity of Carbon Monoxide (CO)
Carbon monoxide (CO) is toxic because it binds to the iron(II) in haemoglobin much more strongly than oxygen, and in doing so, it prevents oxygen from binding. When carbon monoxide is inhaled:
1. CO replaces at the centre in the haemoglobin complex.
2. This forms carboxyhemoglobin , where is tightly bound to the iron.
3. As a result, haemoglobin cannot carry oxygen effectively, leading to oxygen deprivation in tissues and organs.
The oxygen-carrying capacity of blood is significantly reduced, which can lead to fatal consequences.
Worked Questions
Question:
Give an example of a ligand exchange reaction that results in a change in coordination number. Include the equation, complex shapes, and colours or solutions.
Answer:
ions displace the water ligands:
Coordination number changes from 6 to 4
Shape changes from octahedral to tetrahedral
Solution changes from pale blue to yellow
Summary of some key examples
Type of Ligand Exchange | Example | Coordination Number Change? | Solution Colour Change |
|---|---|---|---|
Complete | ❌ | Pink → Yellow (not assessed) | |
Complete | ✅ (6 → 4) | Pale blue → Yellow-green | |
Incomplete | ❌ | Pale blue → Deep blue | |
Complete – multidentate (chelate effect) | ❌ | Pink → Maroon (not assessed) |
Key Tips
Always state whether the coordination number or shape changes
For EDTA or bidentate ligands, refer to the chelate effect and entropy