Variable Oxidation States
Lajoy Tucker
Teacher

Contents
What Are Variable Oxidation States?
Oxidation state (oxidation number) is the charge an atom would have if the bonding were purely ionic. Transition metals are known for having multiple stable oxidation states, unlike and block elements.
Why Do Transition Metals Have Variable Oxidation States?
Electrons are lost from both the and orbitals, which are close in energy.
Different numbers of electrons can be lost easily, forming multiple oxidation states.
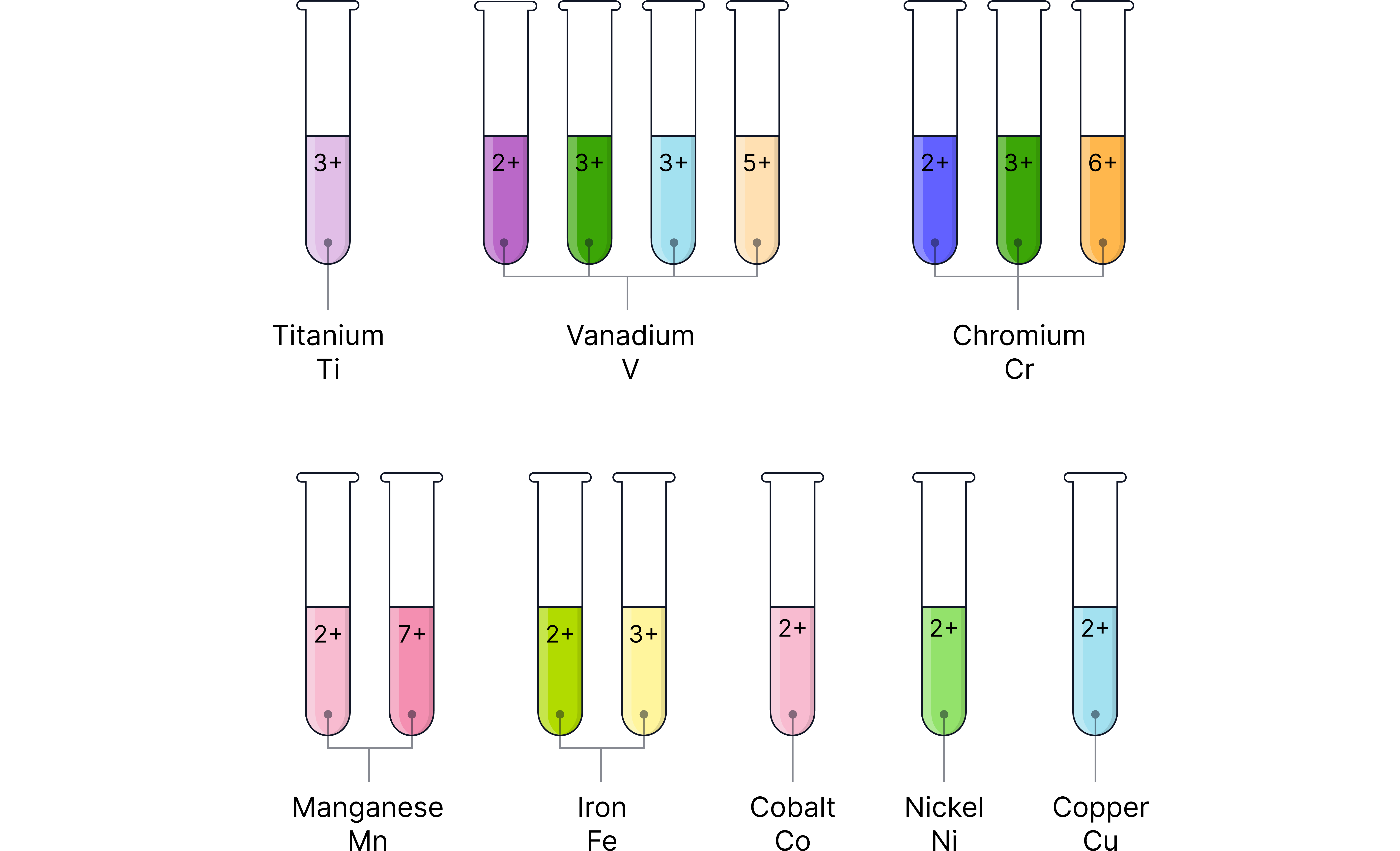
Common Oxidation States of Transition Metals
Element | Common oxidation state(s) | Colour in solution |
Colourless | ||
Purple, colourless | ||
Violet → Yellow across series | ||
Green, orange | ||
Pale pink, purple | ||
Green, yellow | ||
Pink, blue | ||
Green | ||
Blue | ||
Colourless |
and aren’t true transition metal elements as they do not have variable oxidation states.
Catalysts and Variable Oxidation States Explained
Redox Chemistry of Transition Metals
Transition metals often undergo redox reactions where they are oxidised or reduced, changing oxidation state.
Example: Vanadium Redox Series (in acidic solution)
Ion | Oxidation State | Colour |
Yellow | ||
Blue | ||
Green | ||
Violet |
Each step involves electron gain and a visible colour change.
Colours of these species in solution can be assessed. They are easily remembered using the mnemonic ‘You Better Get Vanadium‘ (Yellow Blue Green Violet).
Practice Question
Question 1
Which transition metal can exist in oxidation states of and and shows a series of colours from violet to yellow?
a) Chromium
b) Iron
c) Vanadium
d) Manganese
Answer: C
Question 2
The dichromate ion is used in redox titrations.
a) Determine the oxidation state of chromium in
b) Write a half-equation for the reduction of to in acidic solution.
c) What is the colour change during this reaction?
Answer:
a) Let
So, chromium is in oxidation state
b)
c) Colour change: Orange
Question 3
In a reaction, a solution of VO₂⁺ is reduced stepwise to V²⁺ by zinc in acidic conditions.
a) Write balanced half-equations for each of the two reduction steps:
VO₂⁺ to VO²⁺
VO²⁺ to V³⁺
b) Describe the observed colour changes during this process.
Answer:
a)
VO₂⁺ + 2H⁺ + e⁻ → VO²⁺ + H₂O
VO²⁺ + 2H⁺ + e⁻ → V³⁺ + H₂O
b)
Colour changes:
Yellow (VO₂⁺) → Blue (VO²⁺)
Blue (VO²⁺) → Green (V³⁺)