Catalysis
Lajoy Tucker
Teacher

What is Catalysis?
Catalysis is the increase in the rate of a chemical reaction by a catalyst, which is not used up during the reaction. Catalysts provide an alternate reaction pathway with a lower activation energy.
- A transition metal can act as a catalyst because it can:
- Exist in variable oxidation states
- Form complexes with reactants
- Provide a surface for the reaction to take place
Catalysis Explained
Types of Catalysis
Heterogeneous Catalysis
Catalyst in a different phase from reactants (usually a solid catalyst with gaseous or aqueous reactants).
Reaction occurs on the surface of the catalyst.
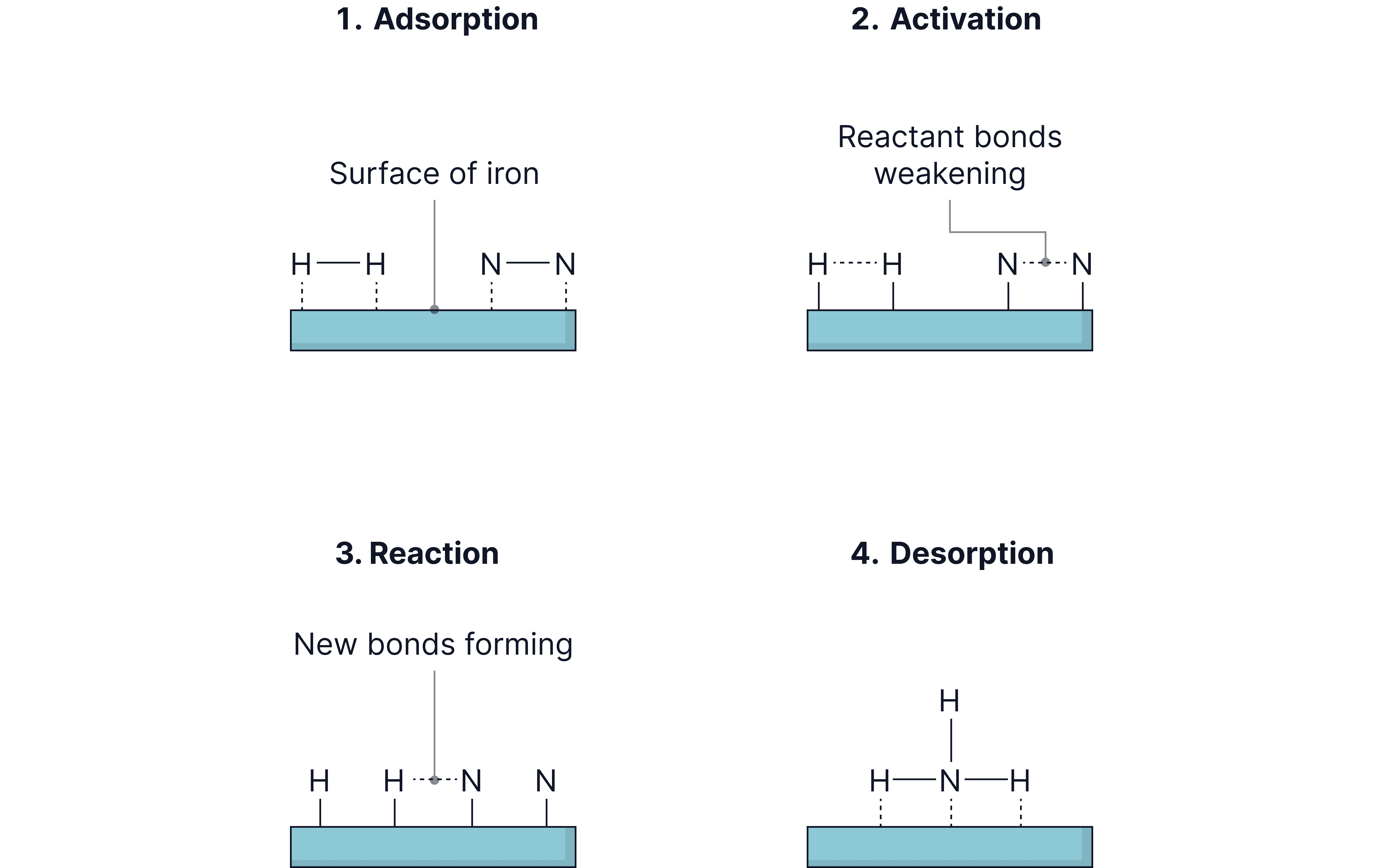
Steps in Heterogeneous Catalysis
1. Adsorption of reactants onto active sites on the catalyst surface.
2. Bond weakening / activation of reactants.
3. Reaction occurs.
4. Desorption of products.
Example 1: Haber Process
Overall reaction:
Catalyst:
Impurities in the reaction mixture may also bind to the catalysts surface and block the active sites.
This is called catalytic poisoning, which:
Reduces the surface area available to the reactant, slowing down the reaction.
Increases costs of chemical process because less product can be made in a certain time or with a certain amount of energy. Catalyst may even need replacing or regenerating.
Example 2: Contact Process
Overall reaction:
Catalyst: (Vanadium (V) oxide)
Note: The 'V' here relates to the +5 oxidation state of the vanadium.
Catalytic steps:
1.
2.
The catalyst is regenerated.
Homogeneous Catalysis
Catalyst is in the same phase as the reactants (usually all aqueous).
They work by combining with the reactants to form an intermediate species which then reacts to form the product and re-form the catalyst.
The reaction pathway that proceeds via this intermediate has an activation energy lower than the direct route.
Example: Reaction between and .
Overall reaction:
Catalyst:
Catalytic steps:
1.
2.
Note: This reaction can also be catalysed by ions which would just switch the order of the catalytic steps and make the intermediate.
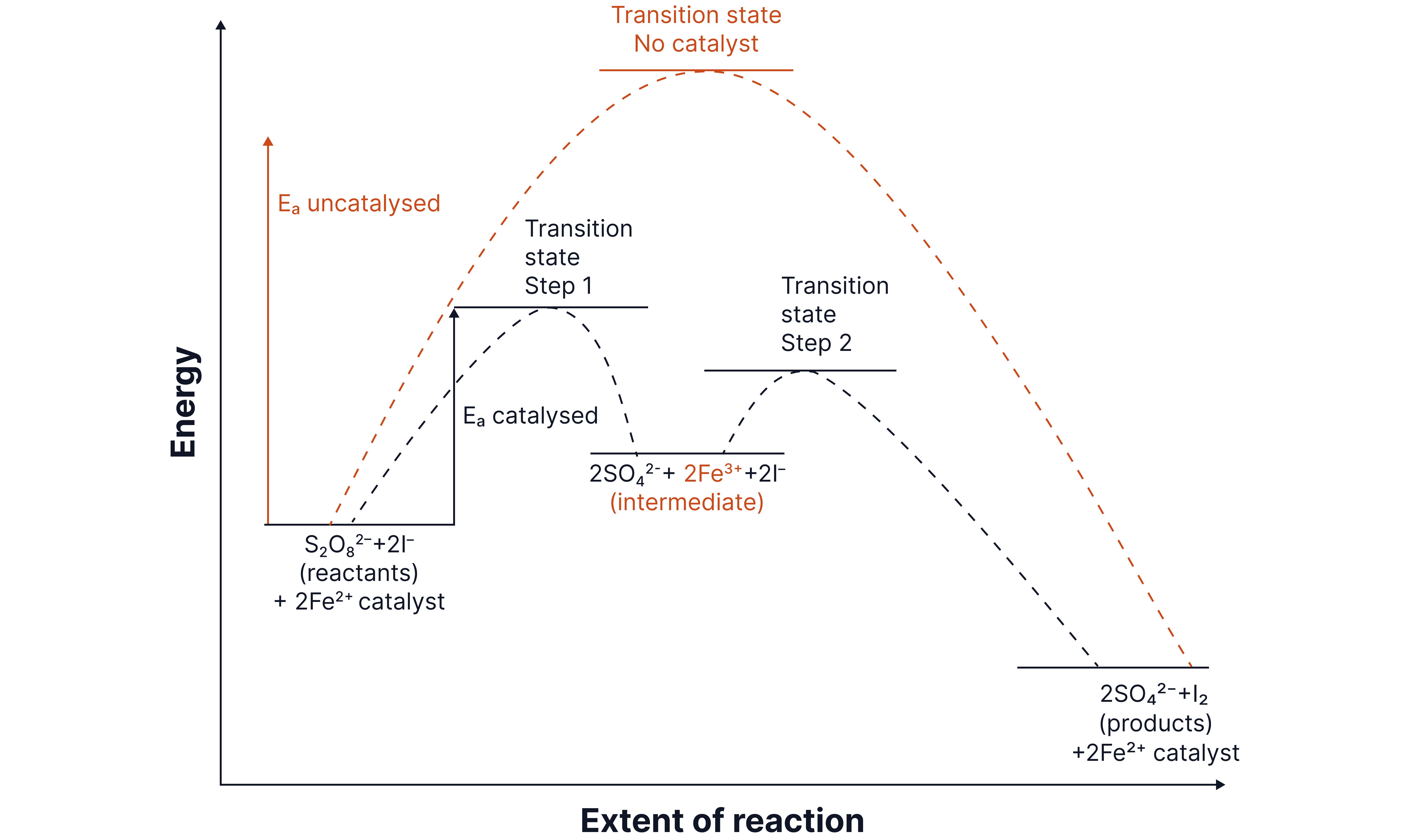
This reaction is very slow without the presence of a catalyst as both reactant ions are negatively charged. A large amount of energy is required to overcome the repulsion between these ions resulting in a high activation energy.
is a suitable catalyst due to its:
Variable oxidation states (catalyst) (intermediate) (regenerated) .
Positive charge allowing it to attract the negative ions and lower the activation energy.
The ions act as the intermediate as they are formed after the first step but used up again by the end of the reaction.
Autocatalysis
Reaction where a product acts as a catalyst.
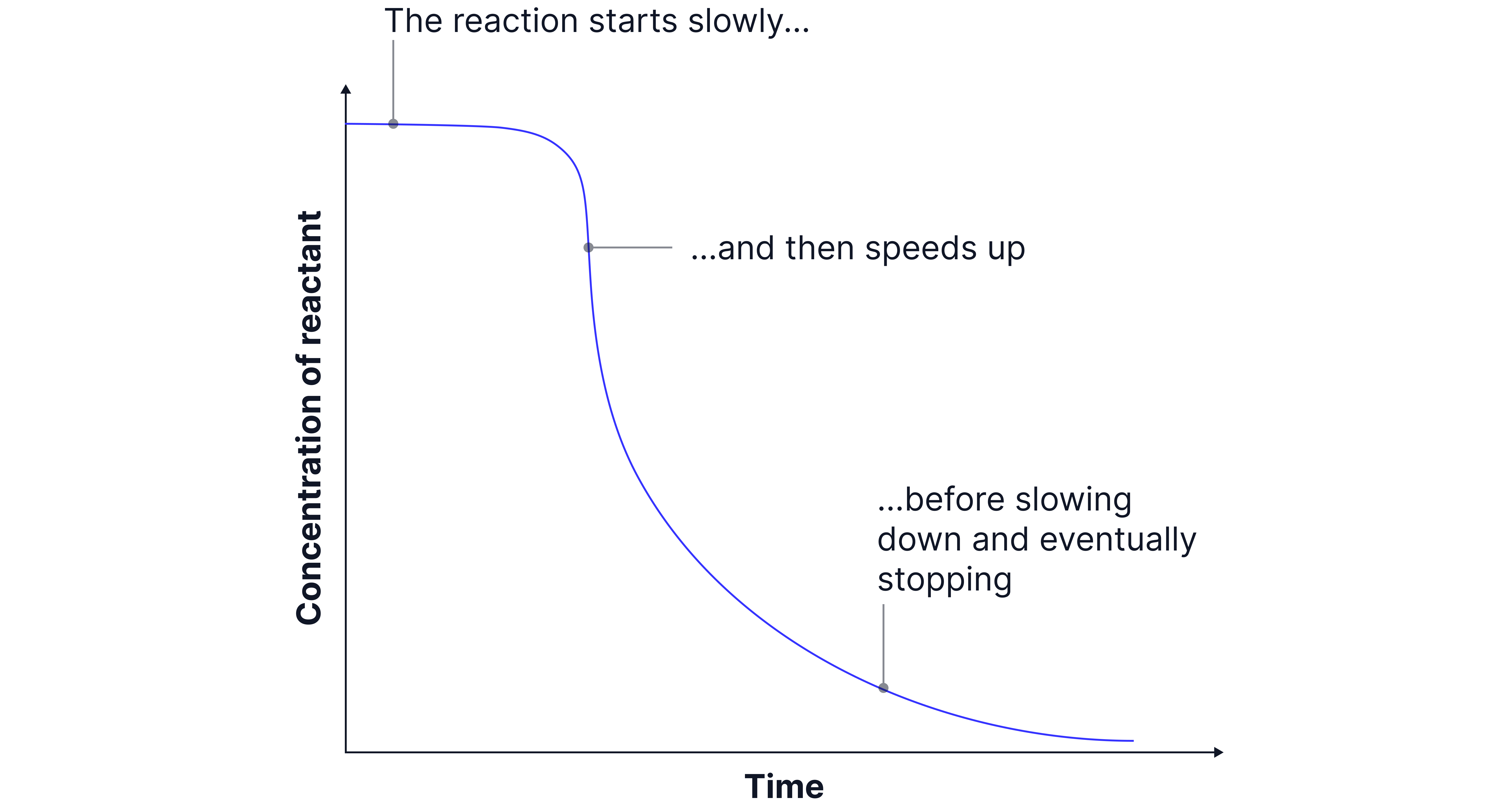
Such reactions start slowly as there is no catalyst present.
As the concentration of the product that is also the catalyst builds up, the reaction speeds up to the catalysed rate.
From then on it behaves like a normal reaction, gradually slowing down as reactants are used up.
Example: Reaction between and
Overall reaction:
Catalyst:
Catalytic steps:
1. react with to produce
2. The intermediate reacts with
Multiplying step 1 by two and step 2 by five to cancel the intermediate returns the overall equation.

Practice Questions
Question 1
Which of the following statements about catalysts is correct?
a) Catalysts increase the yield of a reaction
b) Catalysts increase the rate by raising the activation energy
c) Catalysts are always in the same phase as reactants
d) Catalysts are not used up in the overall reaction
Answer: D
Question 2
Explain why transition metals are often used as catalysts.
Answer:
Transition metals have variable oxidation states enabling alternative reaction pathways with lower activation energy.
Question 3
The overall reaction is:
→
a) This reaction is slow without a catalyst. Suggest why.
b) Write the two steps showing as a catalyst.
Answer:
a) Because both and are negatively charged, they repel, so the uncatalysed reacting has a high activation energy.
b)
1.
2.
Question 4
In the autocatalysed reaction between and , the rate increases as the reaction proceeds.
Explain why this happens.
Answer:
is produced during the reaction and acts as a catalyst, increasing the rate – this is autocatalysis.
Key Notes and Reminders
'Homo' means same and 'hetero' means different.
You must be able to construct all the equations in this revision note as they appear in the specification.