Redox Titrations
Dr. Davinder Bhachu
Teacher

Contents
Introduction
Redox titrations are a method used to determine the concentration of an unknown substance by a redox (electron transfer) reaction.
They may be used to:
Estimate the concentration of a transition metal ion in an unknown solution.
Calculate the percentage composition of a metal in a solid of a compound or alloy.
These are reactions of an oxidising agent against a reducing agent.
As transition metals formed coloured compounds, most redox titrations result in a colour change at the end point so do not require an indicator.
An indicator is still sometimes added to make the end-point more distinct.
These titrations commonly involve:
Transition metal ions (e.g., )
Oxidising agents (e.g., acidified )
Reducing agents (e.g., )
Redox Titration Practice Question Video
Acidified Potassium Manganate(VII) Titrations
as an Oxidising Agent
In acidic solution, is reduced:
The solution is purple, is colourless, so the end-point is a permanent pink (very pale purple) colour formed.
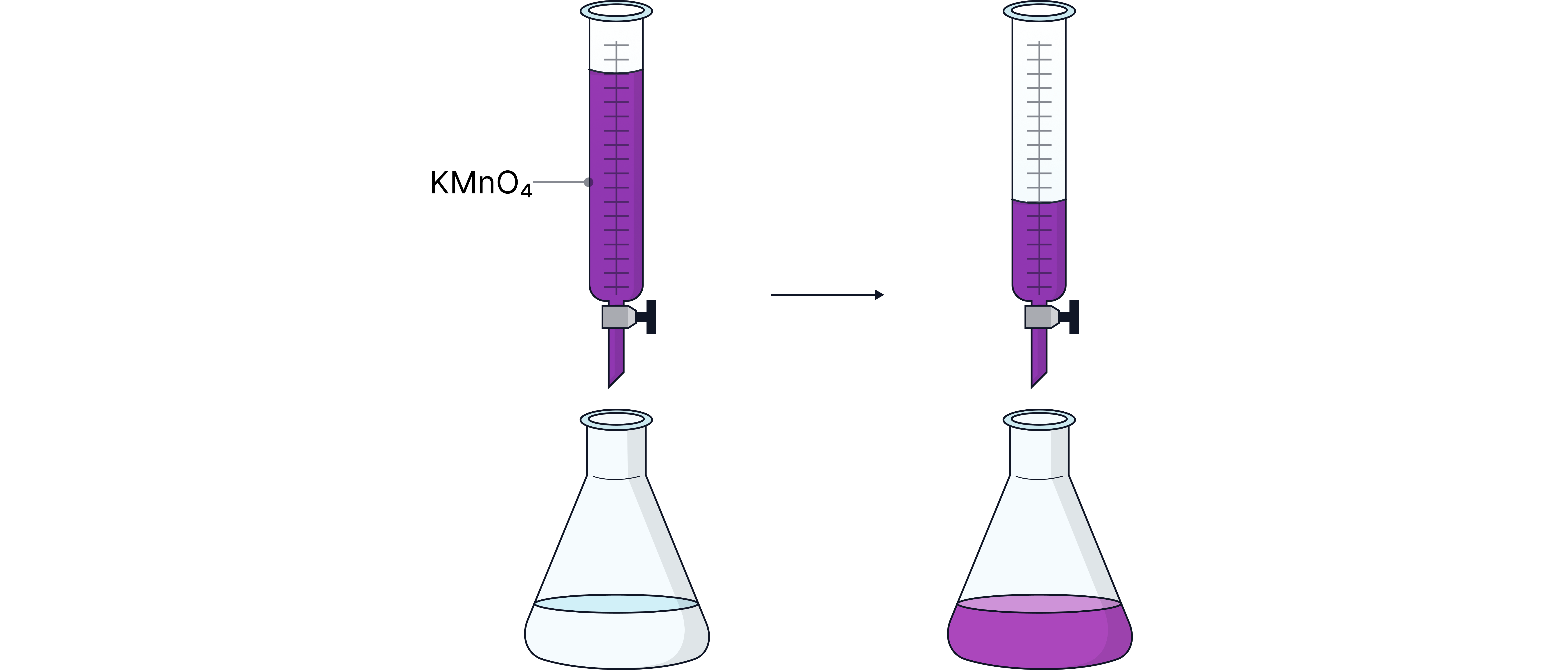
First image - reacting immediately with species in flask
Second image - Species in flask fully reacted so does not react and the colour persists
Common Redox Titration Reactions
a) with
Half equations:
Overall Reaction:
is oxidised to , is reduced to .
b) (oxalate) with
Half equations:
Overall Reaction:
Note - You do not need to memorise these ionic equations but must be able to quickly construct them from half equations.
Procedure Summary
1. Add a standard solution of in the burette, record the initial volume.
2. Pipette a measured volume of into a conical flask.
3. Add excess dilute sulfuric acid (to provide in overall reaction)
4. Titrate until first permanent pink colour persists, record final burette volume.
5. Repeat, adding dropwise from the burette as the endpoint is approached.
6. Repeat until concordant titres are achieved
Calculations
When in doubt… MOLES.
1. Write balanced ionic equation
2. Calculate moles of what you can (usually from burette)
3. Use molar ratio to find moles of the other species (usually )
4. Determine unknown quantity accordingly e.g. mass, , concentration)
Worked Example
of the wire was dissolved in excess of dilute sulphuric acid and the solution was made up to in a volumetric flask. A portion of this solution was pipetted into a conical flask. of was needed for complete oxidation. Determine the percentage of iron in a sample of steel wire.
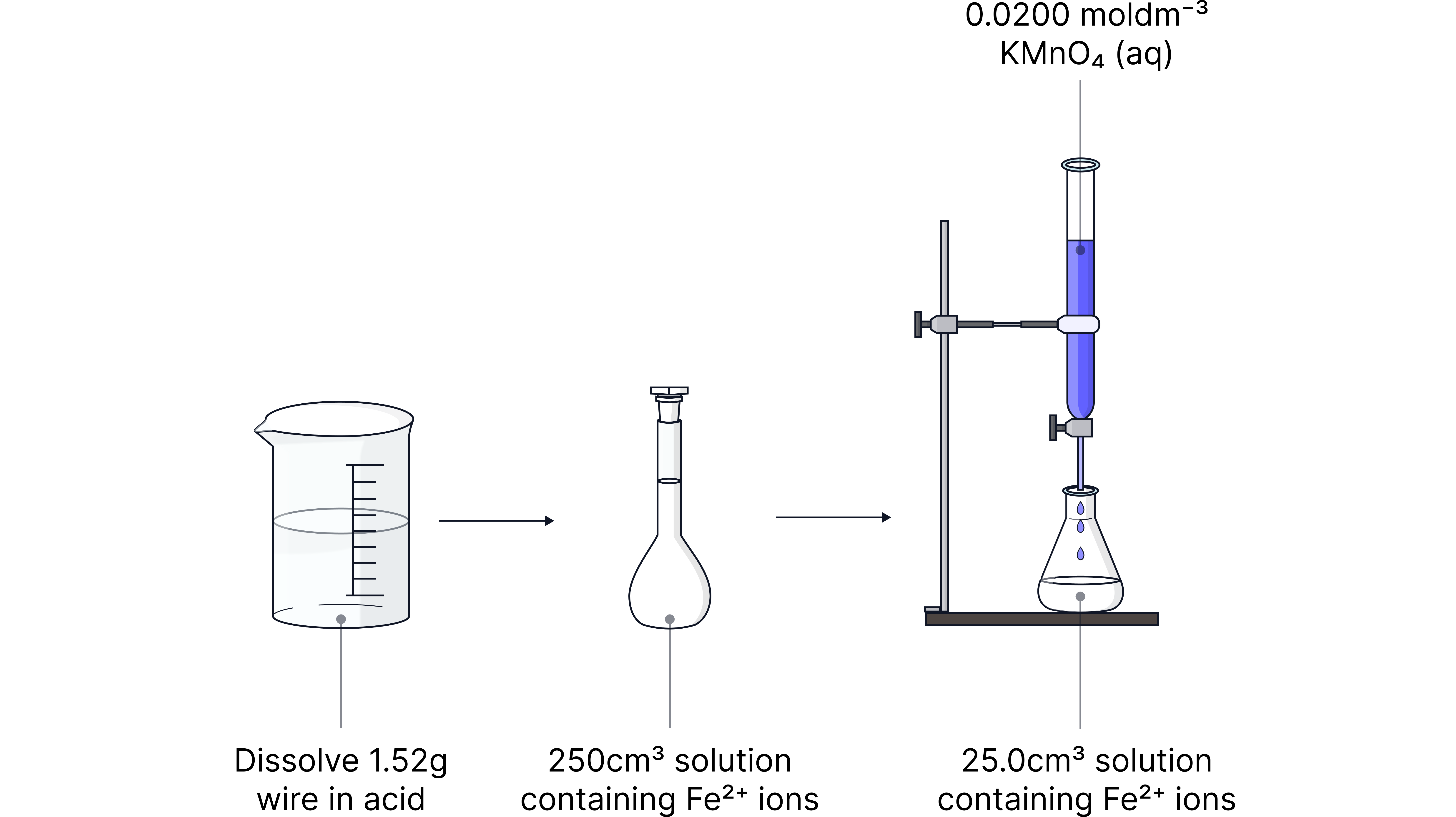
Answer
Calculate moles of from concentration and volume
Use molar ratio to determine moles of ions in portion
Scale the moles to the number in the original solution
Percentage = (mass of /mass of wire) x 100
Percentage % (3 sig figs)