Intermolecular Forces
Lajoy Tucker
Teacher

Contents
Introduction to Intermolecular Forces
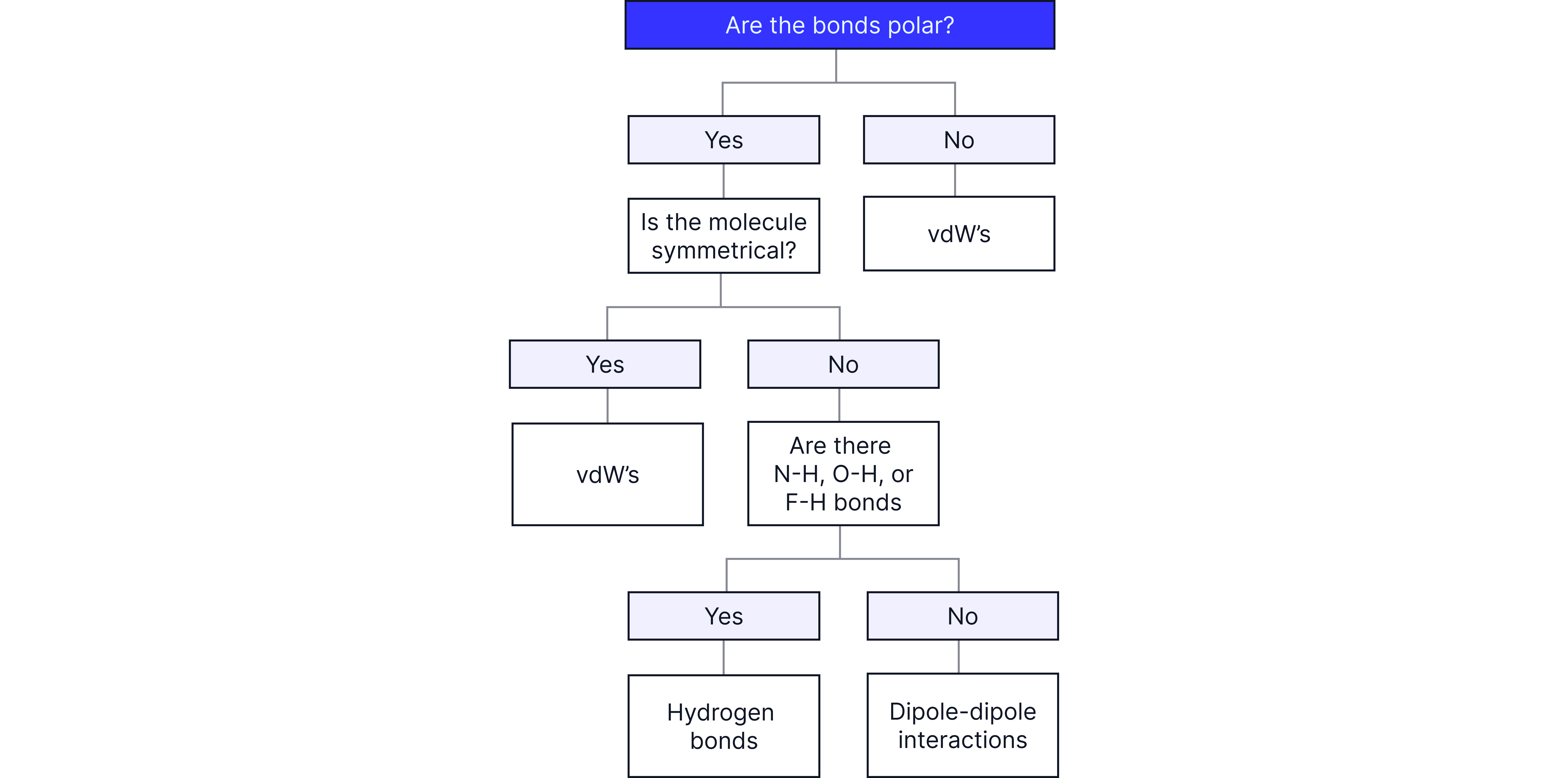
Intermolecular Forces (IMFs): Forces of attraction between molecules — weaker than covalent/ionic/metallic bonds.
Molecular Shape: Determined by electron pair repulsion around the central atom.
Permanent Dipole - an uneven distribution of electron density within a polar molecule
Understanding both allows prediction of physical properties such as boiling point and solubility.
Types of Intermolecular Forces
1. Van der Waals (London) Forces
Temporary dipoles due to fluctuating electron density.
Present in all molecules.
Stronger in larger molecules with more electrons.
How do they arise
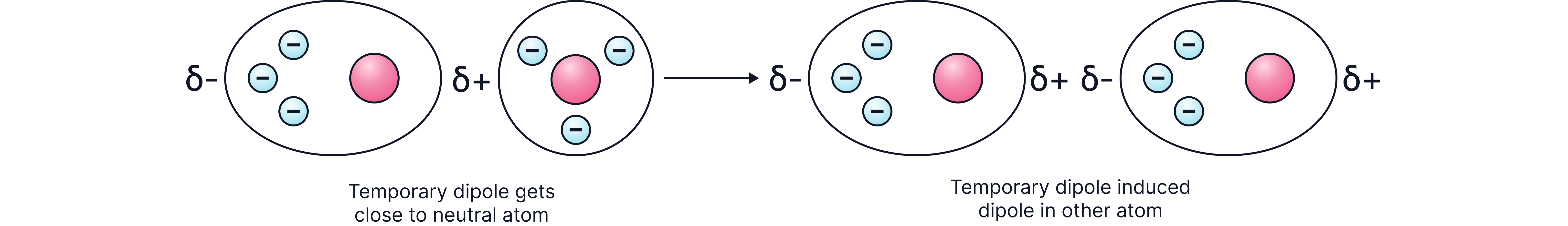
Electrons move quickly in orbitals, so their positions are constantly changing; at any given time, they could be anywhere in an atom.
The possibility exists that one side has more electrons than the other. This will give rise to an instantaneous dipole.
The instantaneous dipole on one atom polarises neighbouring atoms/molecules
Atoms are now attracted to each other by a weak forces
The greater the number of electrons, the stronger the attraction and the greater the energy needed to separate the particles.
Factors affecting strength of van der Waal’s forces
Size of molecule
Larger molecules have more electrons and therefore larger electron clouds that are more easily polarised. This results in temporary dipoles that are more easily formed and longer-lasting,
For example, boiling point of the halogens increases down the group (F2 to I2)
Molecular packing
Linear, straight chain, and flat molecules can pack together more closely
This results in increased surface area of contact and reduced distance between molecules for more and stronger van der Waal’s forces to form.
For example, both pentane and dimethylpropane have the molecular formula C5H10 but 2,2-dimethylpropane has a lower boiling point than pentane.
Straight chain pentane molecules efficiently pack together
Branched dimethylpropane molecules cannot pack as closely together
2. Permanent Dipole–Dipole Interactions
Between molecules with permanent dipoles (polar molecules).
Stronger than van der Waals.
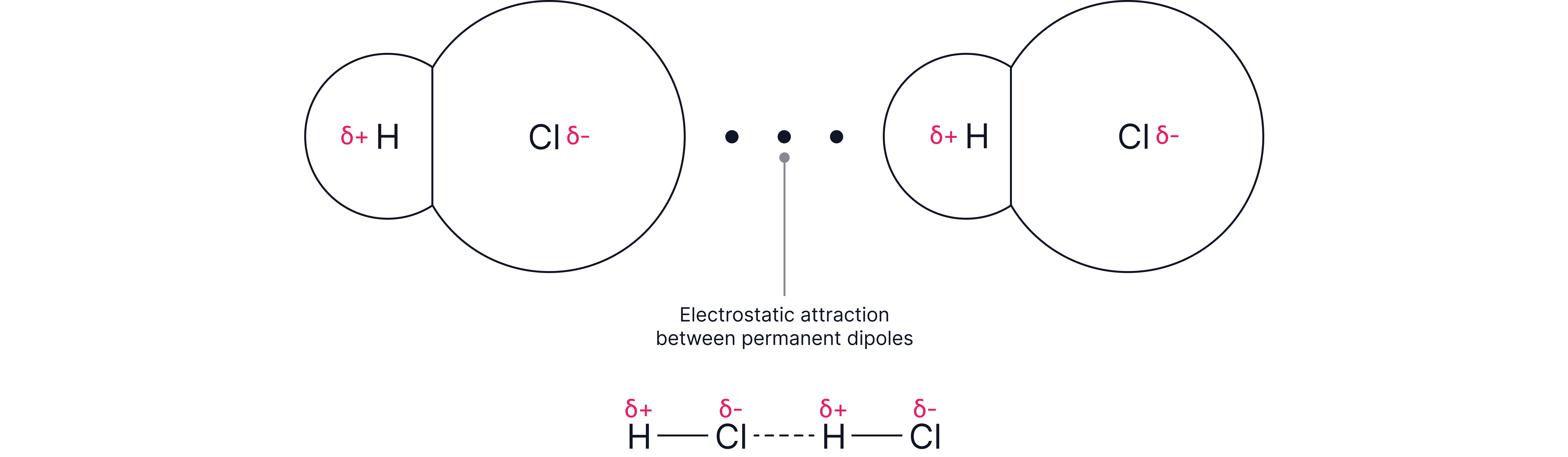
How do they arise?
Attraction between the partially positive atom on one molecule and the partially negative atom on a NEIGHBOURING molecule
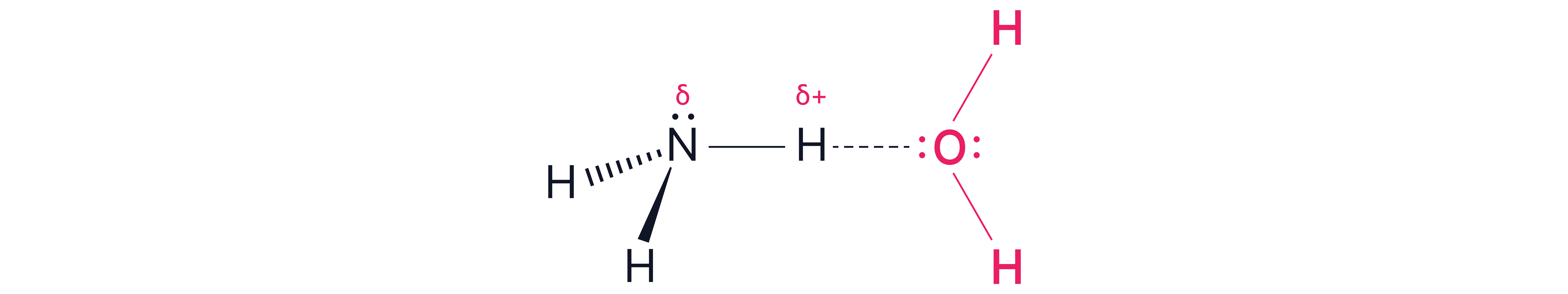
3. Hydrogen Bonding
Between molecules with F–H, O–H, or N–H bonds.
Strongest IMF.
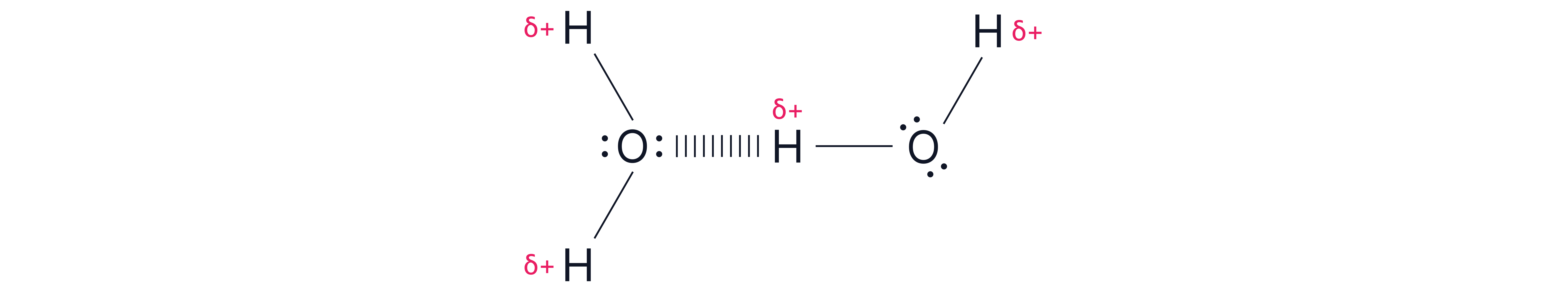
How do they arise?
N, O, and F are very electronegative and can pull electron density away from the bonded hydrogen atom
Electrostatic force of attraction between a lone pair on an N, O, or F atom and a partially positive H atom on a neighbouring molecule
ALL simple molecules have vdWs forces between them
Polar molecules have vdWs and permanent dipole-dipole interactions between them
Molecules with N-H, O-H, of F-H bonds have vdWs forces and dipole-dipole and hydrogen bonds between them
Worked Example
Q: What is the strongest type of IMF present in each substance?
Substance | Strongest Intermolecular Force (IMF) Present |
|---|---|
CH₄ | van der Waals (London dispersion) forces |
HCl | Permanent dipole–dipole interactions |
NH₃ | Hydrogen bonding |
Melting/Boiling point
For simple molecules, melting or boiling the substance depends on the strength of the intermolecular forces.
The stronger the intermolecular force, the more energy is required to overcome them, and the higher the melting/boiling point.
In general,
vdW’s < Dipole-dipole < Hydrogen bonds
Example Question 1
Q: Explain why ethanol () has a higher boiling point than ethane ()
IMF
Comparative strength
Comparative energy
Ethanol has hydrogen bonds between molecules which are stronger than the van der Waal’s forces between ethane molecules and therefore, require more energy to overcome
HOWEVER, for larger molecules, the strength of the van der Waal’s forces can be greater than the permanent dipole-dipole or even hydrogen bonds of a much smaller molecule.
Example Question 2
Explain why has a lower melting point than Include an explanation of the shapes of these molecules in your answer
PF₃ | XeF₄ | |
|---|---|---|
Deduce shape | 3 bonding pairs, 1 lone pair | 4 bonding pairs, 2 lone pairs |
Shape | Tetrahedral | Square planar |
Molecule polarity | Polar — P–F bonds are polar; asymmetric so dipoles do not cancel | Non-polar — Xe–F bonds are polar but molecular symmetry causes dipoles to cancel |
Strongest IMF | Permanent dipole–dipole | van der Waals (London dispersion) |
is a much larger molecule than therefore the van der Waal’s forces between molecules are stronger than the permanent dipole interactions between molecules and therefore require more energy to overcome.
Solubility
A substance's ability to dissolve in a solvent is primarily based on the strength of the interactions between the solute and solvent molecules.
Substances with similar intermolecular forces will be soluble in each other. In other words, ‘like dissolves like’.
For example, ammonia is soluble in water because hydrogen bonds can form between the molecules
Practice Questions
Question 1
Which molecules form hydrogen bonds with water?
A. CH₄
B. CH₃F
C. CH₃OH
D. HCl
A: CH₃OH (has –OH group to H-bond with H₂O)
Question 2
Explain why H₂O has a higher boiling point than H₂S?
Answer:
H₂O forms hydrogen bonds between molecules, H₂S does not.
Overcoming those stronger H-bonds requires more energy than overcoming H₂S’s intermolecular forces
Key Tips
Melting/boiling substances with simple molecular structure requires overcoming "intermolecular forces", not "bonds" when referring to IMFs
Make sure you can deduce whether or not a molecule will be polar based on its shape and types of bonds (link to ‘Polarity’ note)