Electronegativity and Polarity
Lajoy Tucker
Teacher

Introduction and Definitions
While covalent molecules involve the sharing of pairs of electrons, the electron density is not always evenly distributed as atoms of some elements more strongly attract electron density towards themselves resulting in an uneven distribution of charge.
Electronegativity
Electronegativity: The tendency of an atom to attract a bonding pair of electrons toward itself (in a covalent bond).
Increases across a period ()
Decreases down a group ()
Most electronegative elements:
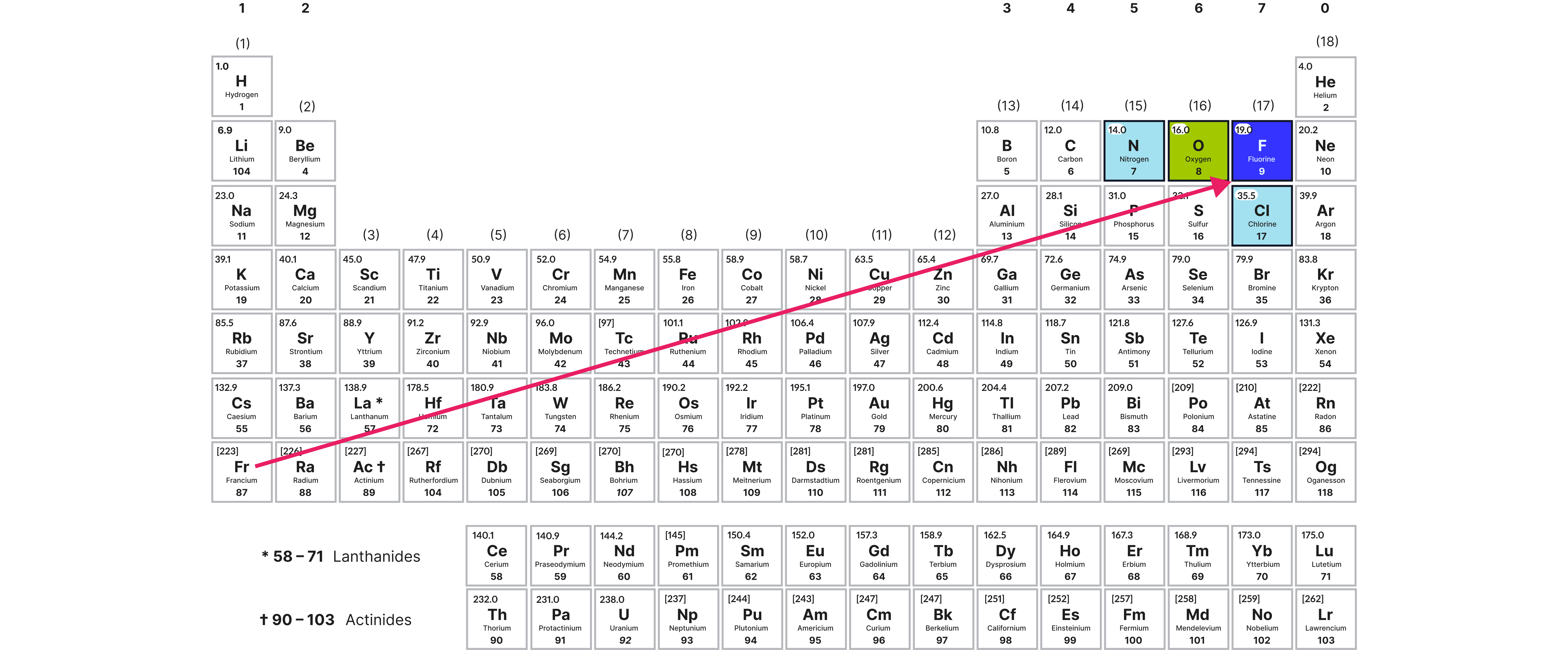
Electronegativity increases across a period as:
The nuclear charge (number of protons increases)
The number of shells and therefore shielding effect remains the same
The atomic radius decreases
There is a stronger attraction between the nucleus and the electrons in the covalent bond
Electronegativity increases up a group as:
Fewer shells and therefore lesser shielding effect
Smaller atomic radius
There is a stronger attraction between the nucleus and the electrons in the covalent bond
What Is Polarity?
Polarity refers to uneven distribution of electrons.
| Concept | Description |
| Bond Polarity | Caused by difference in electronegativity |
| Molecular Polarity | Depends on overall shape + symmetry |
The Bonding Spectrum
The difference in electronegativity between the bonded atoms determines the type of bond.
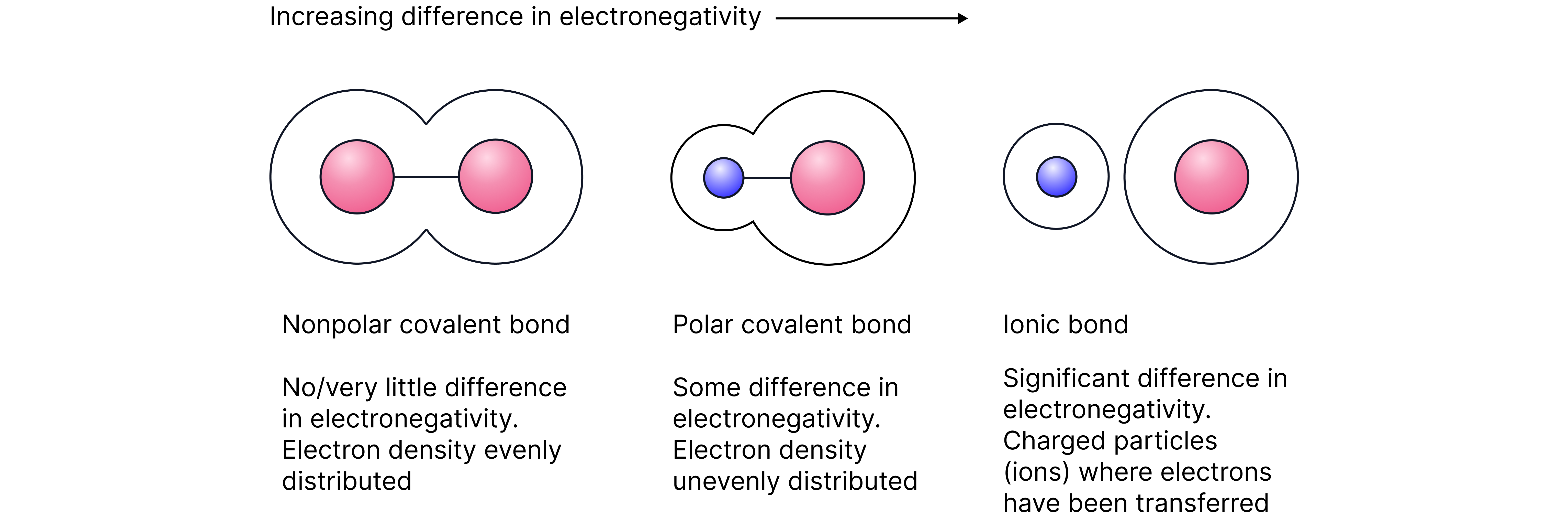
Note – The Pauling scale gives quantitative electronegativity values. The type of bond can then be determined by calculating the difference in electronegativity. In AQA, only qualitative differences are considered.

Bond Polarity
Non-Polar Bonds
Between atoms of the same element – electrons are shared equally resulting in non-polar bonds. e.g

C-H bonds commonly seen throughout organic chemistry are also non-polar due to C and H having similar electronegativities.
Polar Bonds
A difference in electronegativity between bonded atoms results in a polar bond. The more electronegative atom pulls the electron density towards itself resulting in a partially negative charge shown as .
The less electronegative atom which is now electron deficient is shown as .
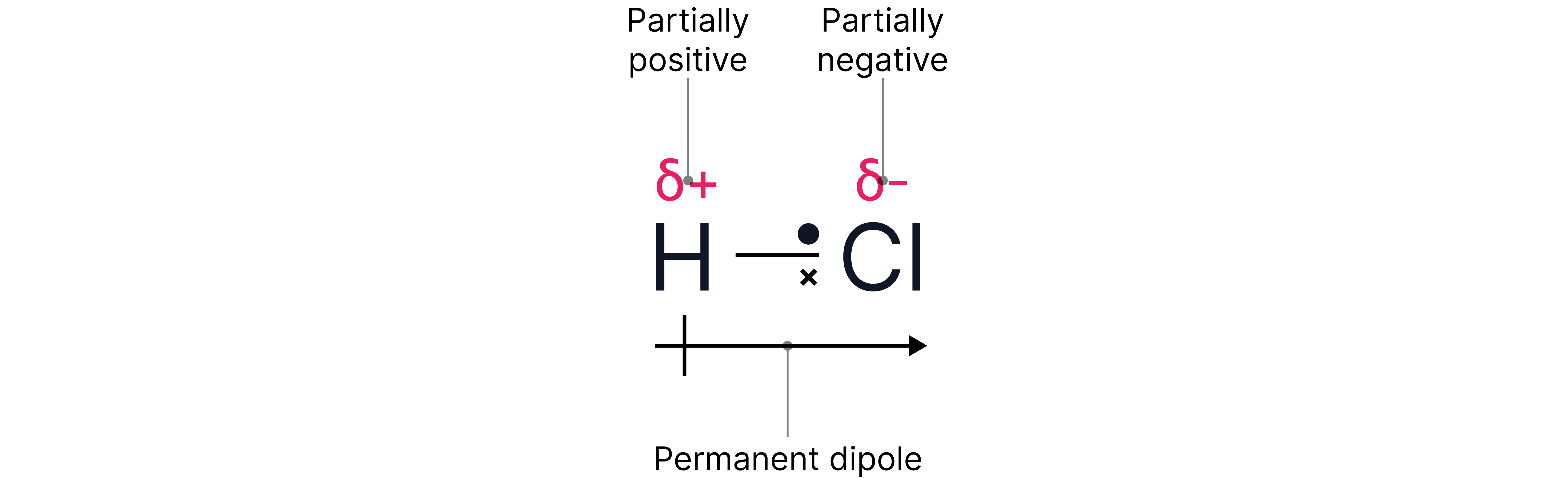
As a general rule, if a non-metal atom is bonded to N, O, F, or Cl (the most electronegative elements) the bond will be polar.
e.g.
Quick Check!
Which of the bonds below are polar?
Answer:
Polar Molecules
A molecule is polar if:
It has polar bonds - difference in electronegativity between covalently bonded atoms.
The molecule is asymmetrical - the dipoles do not cancel out.
Molecules with polar bonds may still be non-polar if the dipoles cancel out due to symmetry.
To deduce whether a molecule is polar, you must first determine its shape. This is covered in the 'Shapes of Molecules and Ions'note.
Worked Examples
1.
Are the bonds polar?
Yes, there is a difference in electronegativity between O and H (remember to look out for N, O, F, or Cl)
Is the molecule asymmetrical?
is bent. The molecule is asymmetrical therefore the dipoles do not cancel out. There is a net dipole moment pointing up towards the . The molecule is polar.
2.
Are the bonds polar?
Yes, there is a difference in electronegativity between B and F (remember to look out for N, O, F, or Cl).
Is the molecule asymmetrical?
is trigonal planar. Three bond dipoles in opposite directions, of the same magnitude. The molecule is symmetrical therefore the dipoles cancel out. There is no net dipole moment. The molecule is non-polar.
Note - represents a permanent dipole showing the direction in which the electron density is being pulled.
More Examples
Molecule | Polar Bonds? | Shape | Dipoles Cancel? | Overall Polarity |
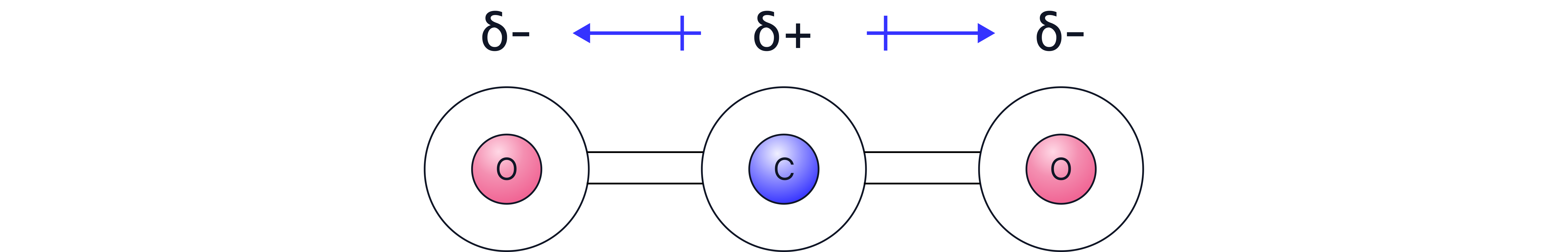
CO₂ | Yes | Linear | Yes | Non-polar |
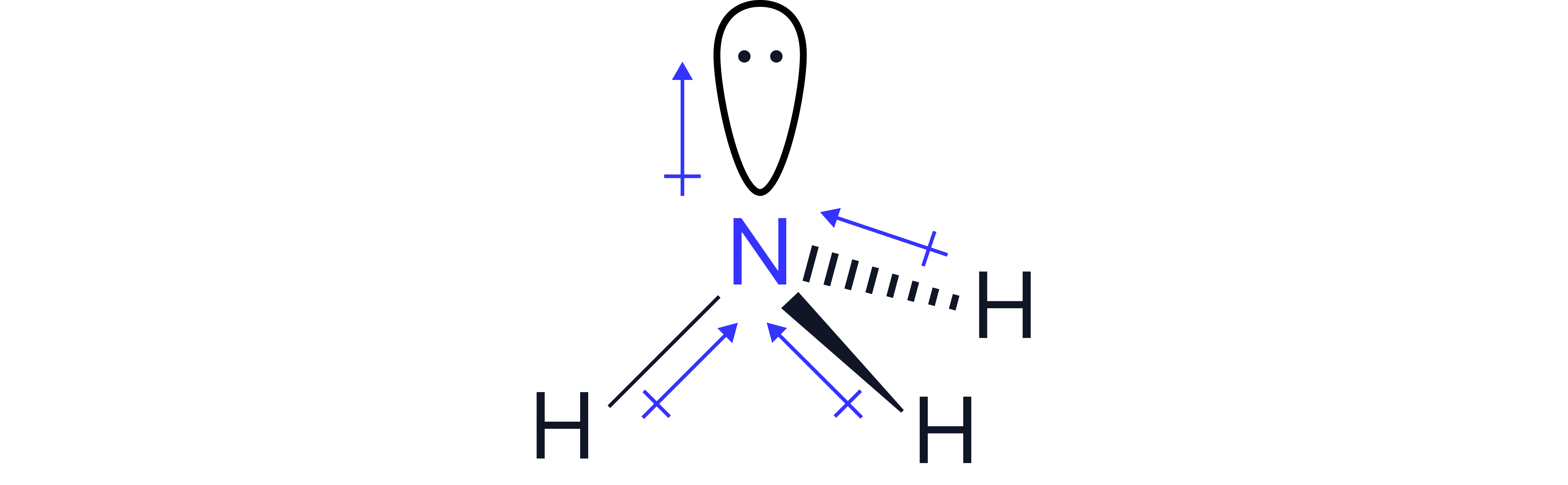
NH₃ | Yes | Trigonal pyramidal | No | Polar |
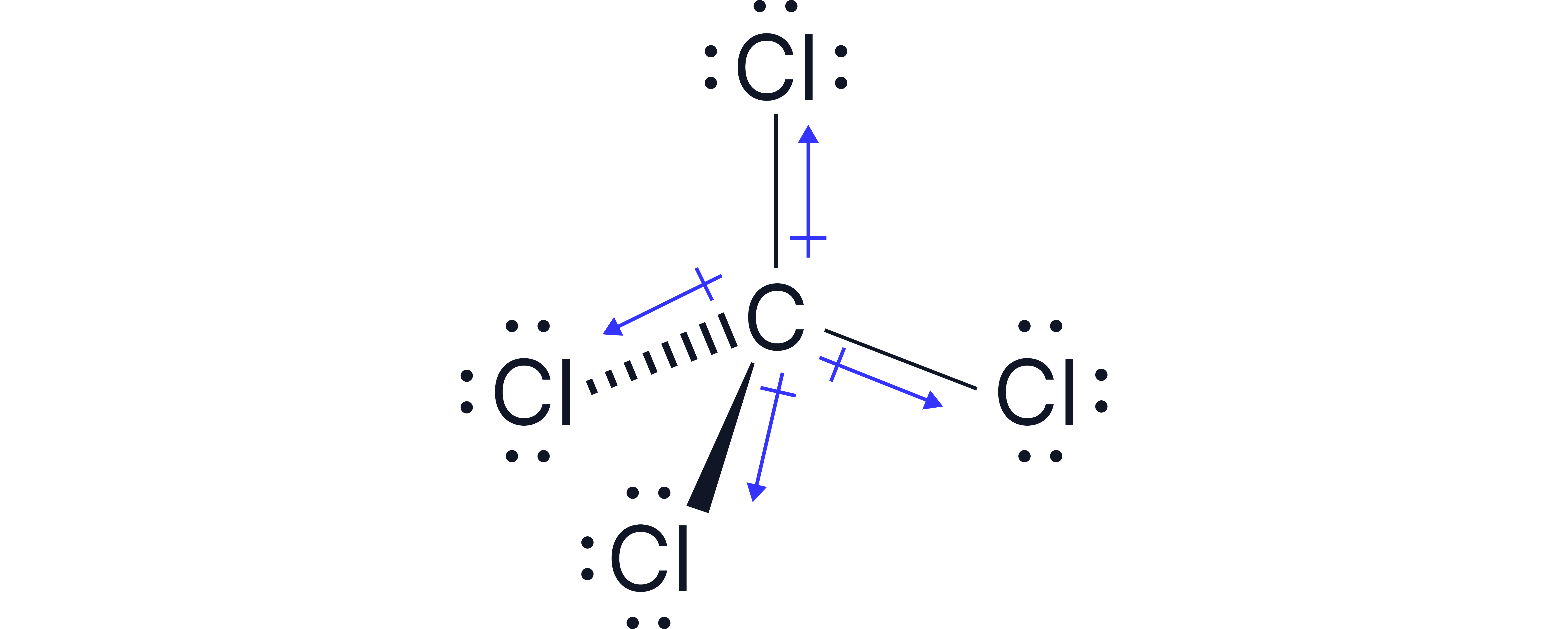
CCl₄ | Yes | Tetrahedral | Yes | Non-polar |
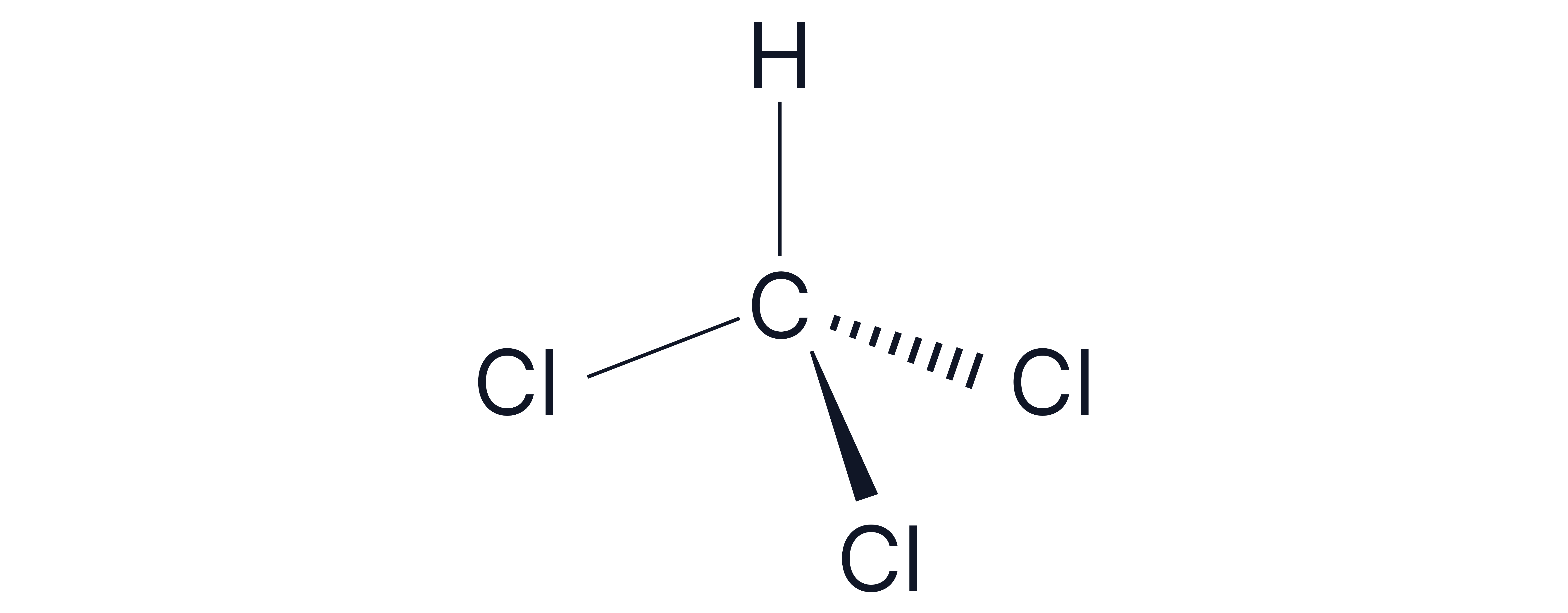
CHCl3 | C-Cl Yes C-H No | Tetrahedral | No | Polar |
Question
Which molecule is non-polar despite having polar bonds?
A. Water,
B. Ammonia,
C. Carbon Dioxide,
D. Hydrogen Fluoride,
Answer
All have polar bonds, but CO₂ has polar bonds but is symmetrical, so answer is Carbon Dioxide.
Use symmetry of shape to determine if dipole cancels
Key Tips
How to assess molecular polarity:
Identify all bonds → Are they polar?
Draw molecular shape (e.g., linear, bent, tetrahedral)
Use dipole arrows to show direction
Determine if dipoles cancel (symmetrical) or add up (asymmetrical)
Always consider both shape and bond polarity when deciding molecular polarity.
Common Mistakes to Avoid
Saying a molecule is polar just because it contains polar bonds
Forgetting lone pairs affect molecular shape
Confusing bond polarity with molecular polarity