Mass spectrometry
Lajoy Tucker
Teacher

Contents
What is Mass Spectrometry?
Mass spectrometry is an analytical technique that identifies compounds by measuring the mass-to-charge ratio (m/z) of ions formed from molecules.
How Mass Spectrometry Works:
1. Ionization: Sample molecules are bombarded with high-energy electrons (electron impact ionization).
2. Acceleration: Ions are accelerated through an electric field.
3. Deflection: Ions are separated based on their m/z ratios in a magnetic field.
4. Detection: Ions are detected and recorded as peaks on a mass spectrum.
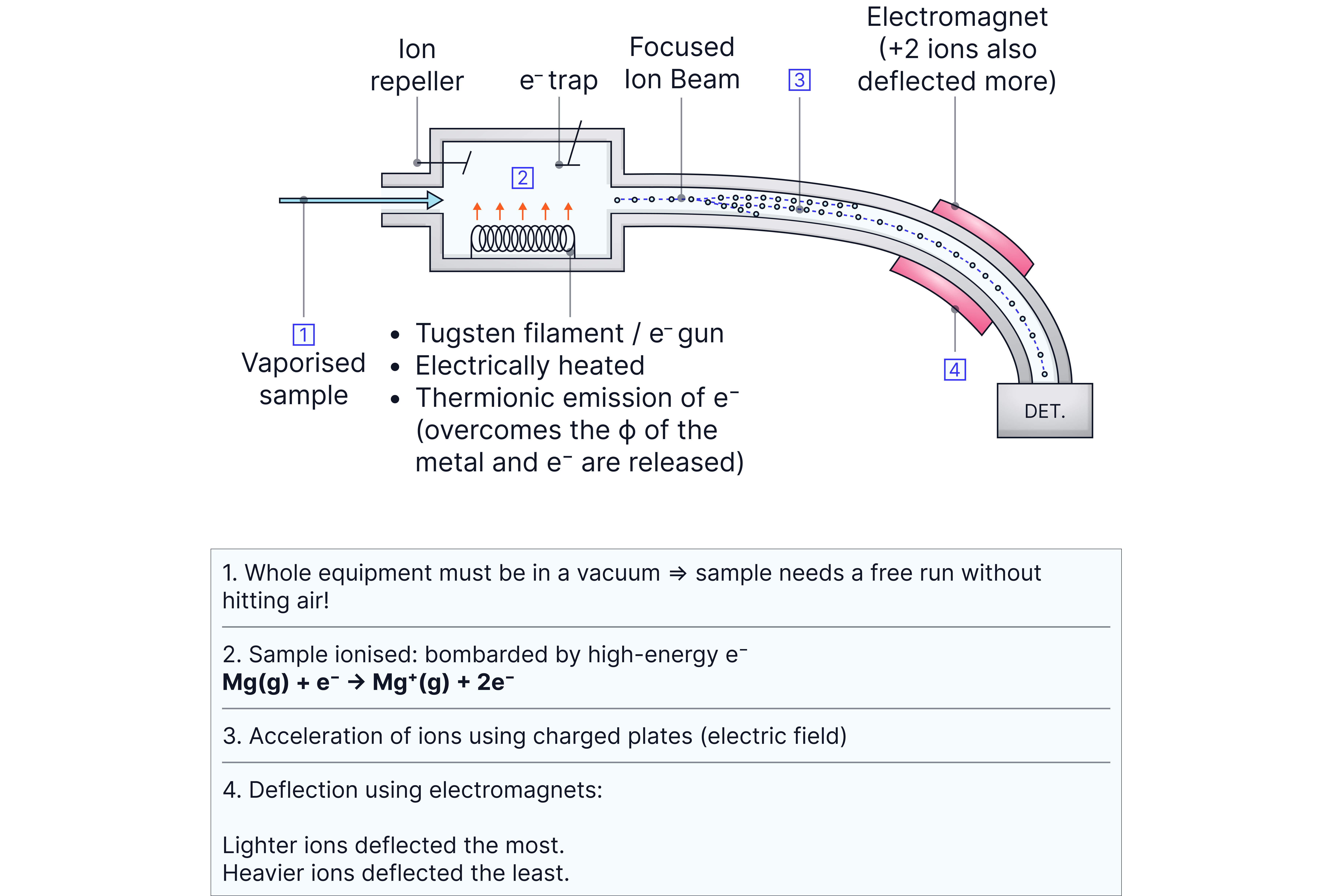
We measure m/z ratios rather than just masses because the technique separates ions based on their mass-to-charge ratio. Since most ions have a +1 charge, m/z effectively equals the mass.
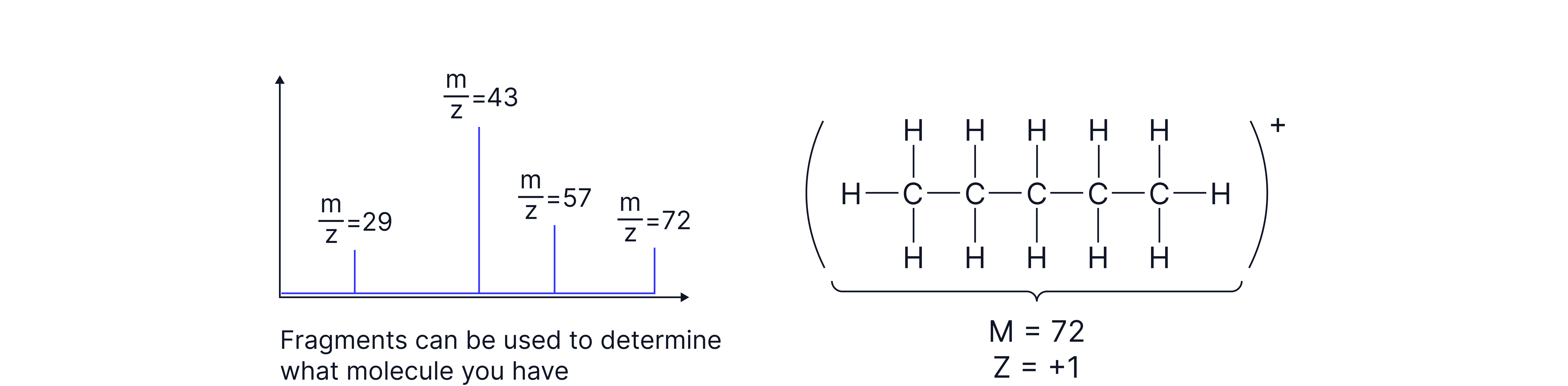
Purpose of Mass Spectrometry in Organic Analysis
Mass spectrometry can be used to determine the molecular formula of a compound by using:
• Precise relative atomic masses (to 4 decimal places)
• The accurate molecular mass (Mr) from the molecular ion peak (M⁺) in a high-resolution mass spectrum
Key Features of Mass Spectra
The Molecular Ion Peak (M⁺)
• The molecular ion is the species formed when a molecule loses one electron, but no atoms:
M → M⁺ + e⁻
This peak appears at the highest m/z value (excluding minor isotopic peaks) and represents the molecular mass of the compound.
Important: Some molecules don't show clear molecular ion peaks because the molecular ion is unstable and fragments immediately.
The Base Peak
• The base peak is the most abundant ion in the spectrum (assigned 100% relative abundance).
This is often NOT the molecular ion peak.
Usually represents the most stable fragment ion.
The M+1 Peak (Isotope Peak)
• M+1 peak appears one mass unit higher than the molecular ion
• Caused by ¹³C isotopes (1.1% natural abundance)
• Rule of thumb: M+1 peak height ≈ 1.1% × number of carbon atoms
• Example: C₄ compound shows M+1 peak at ~4.4% of molecular ion peak
Fragmentation Patterns
When molecular ions break apart, they form fragment ions. Common losses include:
• -15: Loss of CH₃ (methyl group)
• -29: Loss of CHO (aldehyde group) or C₂H₅ (ethyl group)
• -31: Loss of OCH₃ (methoxy group)
• -45: Loss of COOH (carboxyl group)
Determining Molecular Formula with High-Resolution Mass Spectrometry
Why High-Resolution is Essential?
High-resolution MS can:
• Differentiate between compounds with similar integer masses (e.g. C₂H₄O vs. CH₄N₂)
• Identify molecular formulas to 4 decimal places, allowing accurate deduction.
• Avoid ambiguity: Many compounds have the same integer molecular mass, but precise atomic masses allow accurate identification
Step-by-Step Method:
1. Obtain the molecular ion peak value from the spectrum
2. Use a list of possible empirical formulae (consider C, H, O, N, halogens)
3. Add up the accurate relative atomic masses for combinations
4. Match to the given precise molecular mass (within ±0.0005 tolerance)
5. Check: Does the formula make chemical sense?
Reference Table of Precise Atomic Masses
Atom | Precise Mass (u) |
|---|---|
H | 1.0078 |
C | 12.0000 |
O | 15.9949 |
N | 14.0031 |
F | 18.9984 |
Cl | 34.9689 |
Example Calculation:
Given M⁺ = 60.0211, what is the molecular formula?
Try C₂H₄O₂:
• C (12.0000 × 2) = 24.0000
• H (1.0078 × 4) = 4.0312
• O (15.9949 × 2) = 31.9898
• Total = 60.0210 ✓ (within tolerance ±0.0005)
Answer:
C₂H₄O₂ (ethanoic acid or methyl ethanoate)
Worked Examples
Example 1: Standard Calculation
A molecular ion peak appears at m/z = 88.0524. What is the likely molecular formula?
Try C₄H₈O₂:
• C = 12.0000 × 4 = 48.0000
• H = 1.0078 × 8 = 8.0624
• O = 15.9949 × 2 = 31.9898
• Total = 88.0522 ✓
Answer:
C₄H₈O₂ (e.g. ethyl ethanoate or butanoic acid).
Example 2: Distinguishing Similar Masses
Explain why accurate atomic masses are needed when using mass spectrometry to determine a molecular formula.
Answer:
Because many compounds have similar integer molecular masses, only precise atomic masses allow accurate identification and avoid ambiguity between potential formulae.
Example: C₂H₄O and CH₄N₂ both have integer mass of 44, but:
• C₂H₄O: Precise Mr = 44.0262
• CH₄N₂: Precise Mr = 44.0375
High-resolution MS gives precise values to 4 d.p., so only one matches exactly.
Practice Questions
Question 1
A molecular ion peak is observed at m/z = 46.0419. Suggest a molecular formula and show your calculation.
Try C₂H₆O:
• C = 12.0000 × 2 = 24.0000
• H = 1.0078 × 6 = 6.0468
• O = 15.9949 × 1 = 15.9949
• Total = 46.0417 ✓
Answer:
C₂H₆O (ethanol or dimethyl ether).
Question 2
The molecular ion peak for a compound is 74.0368. Which of the following is a likely molecular formula?
A. C₃H₈O₂
B. C₂H₆O₂
C. C₄H₁₀O
D. C₃H₆O₂
Sample calculation for D:
C₃H₆O₂:
• C = 36.0000
• H = 6.0468
• O = 31.9898
• Total = 74.0366 ✓ (Closest match)
Correct answer: D
Common Student Mistakes to Avoid
1. Confusing molecular ion with base peak: The molecular ion might not be the tallest peak!
2. Forgetting other elements: Always consider N, F, Cl if C, H, O don't work.
3. Arithmetic errors: Double-check decimal place calculations.
4. Ignoring chemical sense: Make sure your formula could actually exist.
5. Wrong tolerance: Only accept matches within ±0.0005.
Connections to Other Techniques
Mass spectrometry works best when combined with:
• IR spectroscopy: Identifies functional groups.
• NMR spectroscopy: Determines structure and connectivity.
• Melting/boiling points: Helps distinguish between isomers.
Real-world applications:
• Drug testing and forensic analysis.
• Environmental monitoring • Protein identification in biochemistry.
• Quality control in pharmaceutical industry.
Key Tips for Success
• Remember: MS gives molecular formula, NOT structure (isomers have same formula) • Always show working: Full calculations earn method marks
• Check your answer: Does the formula make chemical sense?
• Practice calculations: Speed and accuracy come with repetition
• Link to other topics: Refer back to Atomic Structure (3.1.1) for more on Mass Spec fundamentals