Maxwell–Boltzmann Distributions
Dr. Davinder Bhachu
Teacher

Introduction
This curve shows the spread of kinetic energies that particles in a sample have at a specific temperature.
Not all particles have the same energy.
Most have energies around a peak value – this is called the most probable energy (Eₘₚ).
The mean energy is slightly to the right of Eₘₚ.
A small proportion of particles have energy equal to or greater than Eₐ, and only these can react.
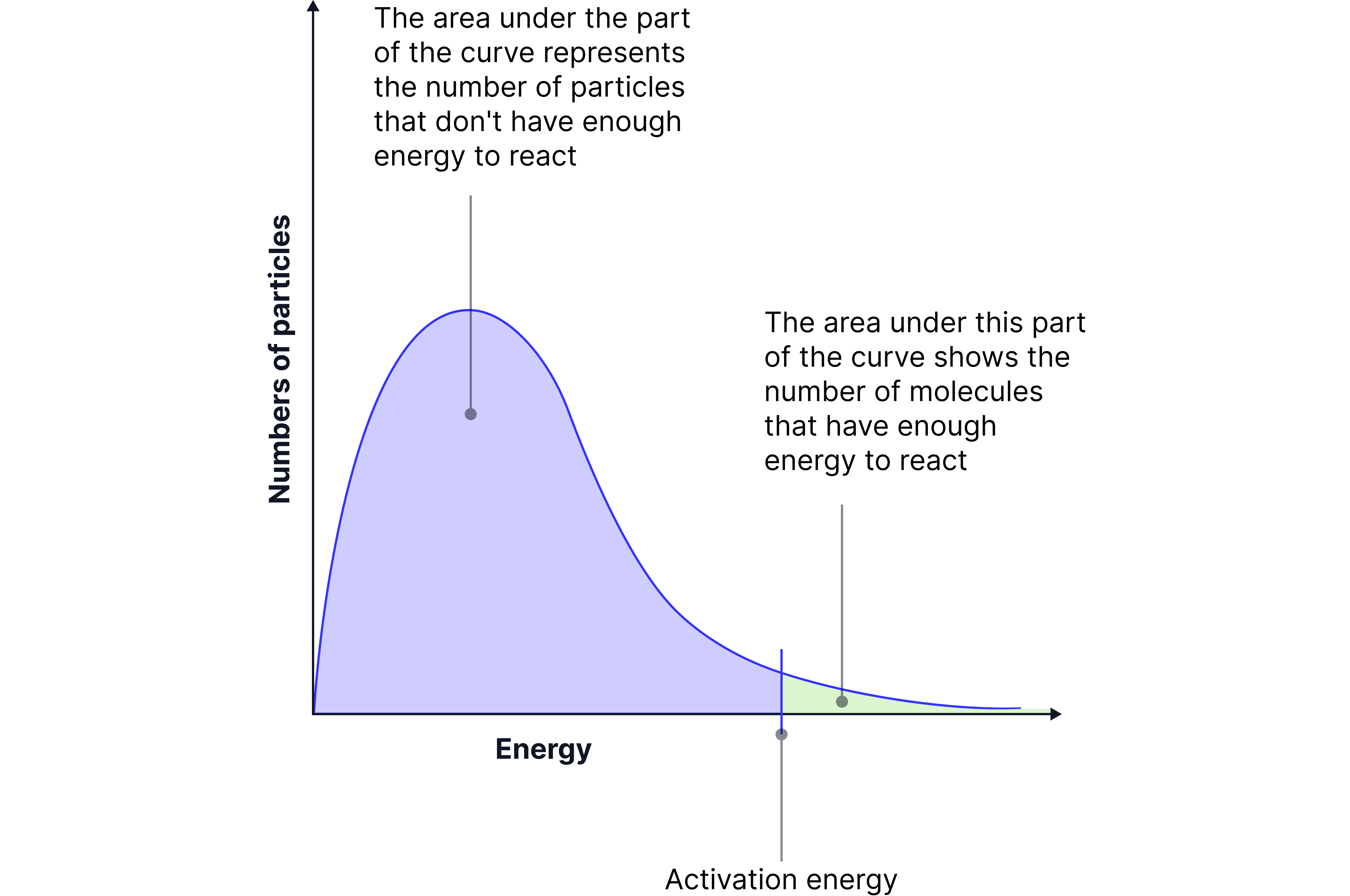
📌 Key features:
The curve starts at the origin (no particles have zero energy).
It rises to a peak (Eₘₚ), then falls.
It never touches the x-axis – there's no maximum possible energy.
Effect of Temperature on Distribution
When temperature increases:
The mean and most probable energies shift to the right (higher values).
The curve flattens and broadens.
A greater proportion of particles have E > Eₐ.
This means the rate of reaction increases, even though the total number of particles stays the same.
📘 Important:
The area under the curve stays constant (same number of particles).
Diagram to redraw (two curves – low temp vs high temp):
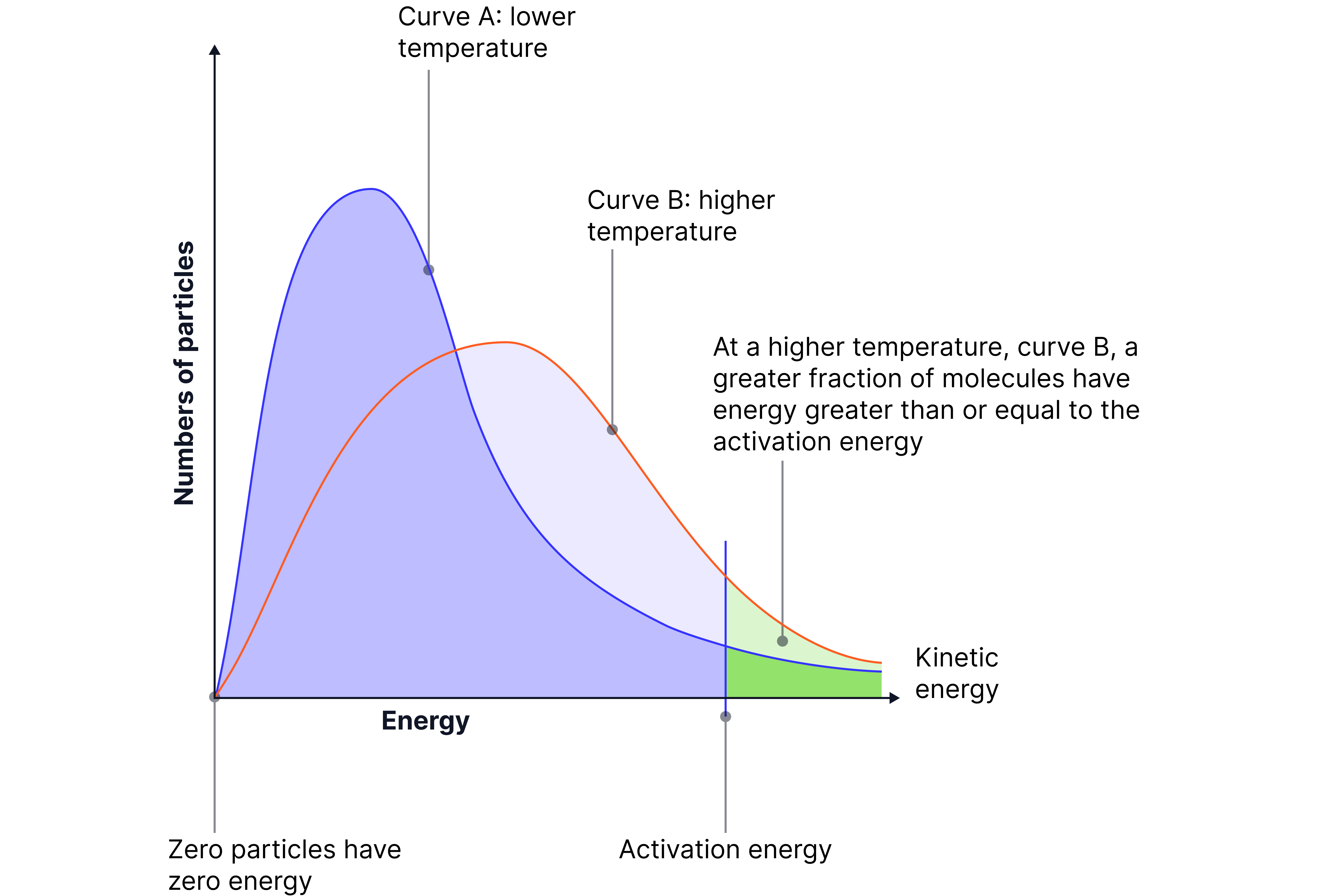
Curves:
Blue curve: Lower temperature – peak is higher and closer to the left
Red curve: Higher temperature – peak is lower and shifted to the right
Worked Examples
Worked Example 1: Effect of Temperature on Molecular Speeds
Question: Consider the reaction between hydrogen () and iodine () to form hydrogen iodide (HIHI):
At 25°C, the activation energy is 50 kJ/mol, and only a small proportion of the molecules have enough energy to react. If the temperature is raised to 45°C, how does this affect the reaction rate?
Answer:
At 45°C, more molecules have energy greater than or equal to the activation energy.
The Maxwell-Boltzmann distribution curve shifts to the right and the peak gets lower and wider, increasing the number of molecules with sufficient energy to react.
The reaction rate will increase, as more molecules collide with effective energy, increasing the number of successful collisions.
Worked Example 2: Effect of Increasing Temperature on the Distribution Curve
Question: A gas is at 300 K and the activation energy for the reaction is 60 kJ/mol. Draw the Maxwell-Boltzmann distribution curve for this system. Then, explain what happens to the distribution curve when the temperature is increased to 350 K.
Answer:
At 300 K:
Most molecules will have energy below the activation energy.
Few molecules will have enough energy to react, resulting in a slow reaction rate.
At 350 K:
The peak of the curve shifts to the right, and the area under the curve becomes wider.
More molecules have enough energy to overcome the activation energy.
The reaction rate increases as more effective collisions occur.
Questions
Question 1: Draw and label the Maxwell-Boltzmann distribution curve at two different temperatures: 300 K and 400 K. Highlight the area corresponding to the activation energy.
Answer 1:
At 400 K, the peak shifts right, and the curve flattens.
More particles have sufficient energy to overcome the activation energy.
Question 2: Explain how the Maxwell-Boltzmann distribution curve can explain why increasing the temperature of a reaction increases the rate of reaction.
Answer 2:
As temperature increases, the average energy of particles increases, shifting the distribution curve to the right.
This means more molecules have energy equal to or greater than the activation energy, leading to more effective collisions and thus a higher reaction rate.
Key Points to Remember:
Maxwell-Boltzmann distribution shows the spread of kinetic energies of molecules in a system.
Increasing temperature causes a rightward shift of the curve, increasing the number of molecules with sufficient energy to react.
The activation energy is the energy threshold for a reaction to occur, and effective collisions are those that exceed this energy.
Be clear on the concept of activation energy. It's crucial to understand that only molecules with energies equal to or greater than Ea can react when they collide.
Describe the effect of temperature changes clearly. Higher temperatures increase the number of molecules with sufficient energy, leading to a higher reaction rate.
Maxwell-Boltzmann curve changes: When drawing or analyzing the graph, always note the shift in the peak and the spread of energies at different temperatures.