Metal-Aqua Ions
Lajoy Tucker
Teacher

Contents
Introduction to Metal-Aqua Ions
When metal salts dissolve in water, complex ions form in which water molecules act as ligands, coordinating to the central metal ion. These metal-aqua ions are usually in an octahedral geometry.
General formula: , where = metal ion, = oxidation number
Examples:
(pale green solution)
(colourless solution)
Metal-Aqua Ions Explained
Acidity of Metal-Aqua Ions
Metal-aqua ions are acidic as water ligands partially dissociate to release H+ ions into solution.
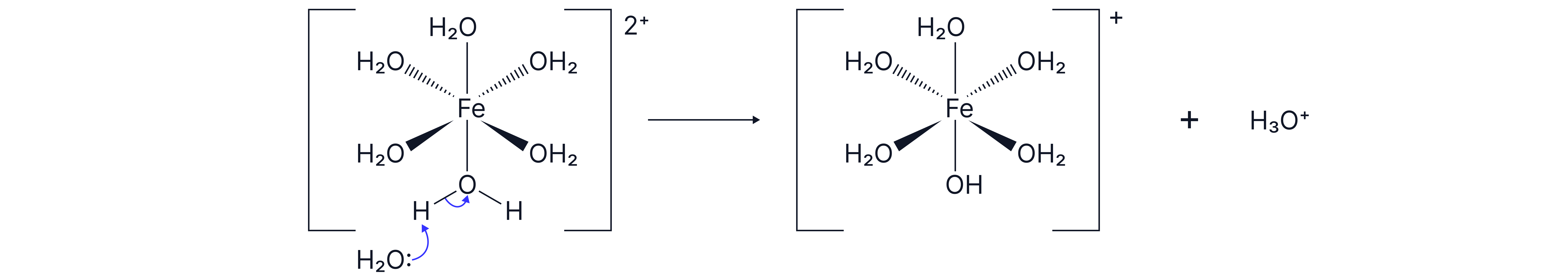
e.g
OR
In general:
OR
The charge of the complex decreases by 1 as the neutral water ligand has become a negatively charged hydroxide ligand () due to the loss of
Comparing acidity
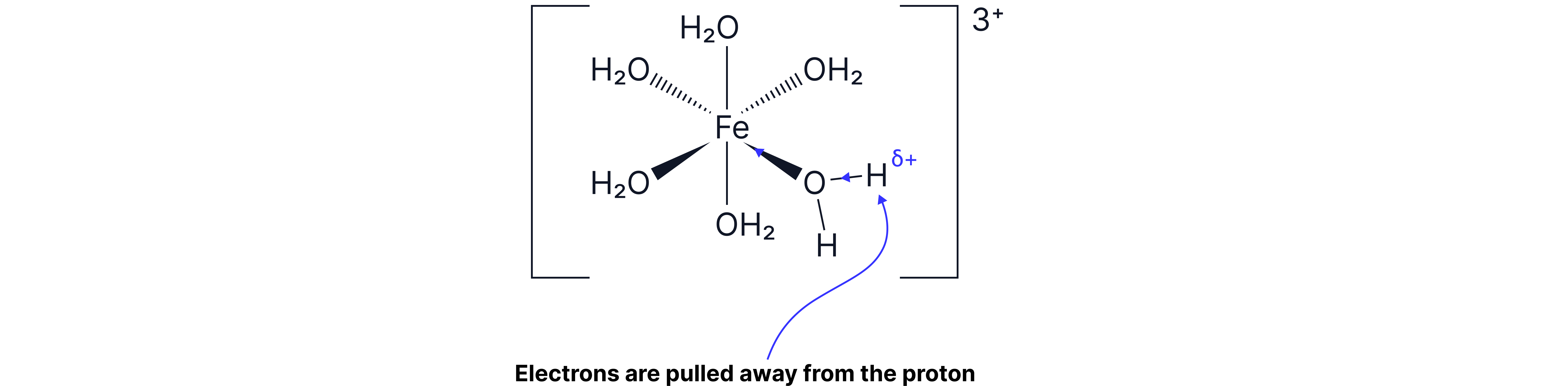
3+ ions are more acidic than 2+ ions.
The high charge density (greater ion charge and smaller ion size) of 3+ ions polarises the O-H bonds in coordinated water molecules more strongly, making it easier for H⁺ ions to be released.
Colours of Metal-Aqua Ions
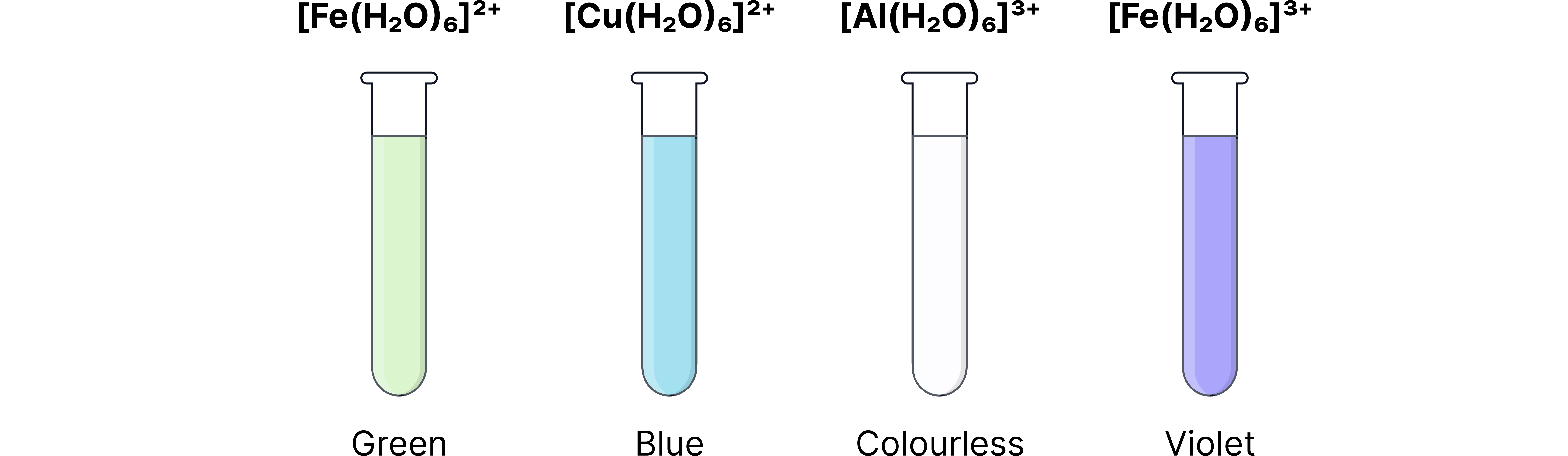
Practice Question
Question
Write an ionic equation that demonstrates the acidic behaviour of in solution
Answer:
OR
Explanation - The ions that make up are and . In solution, the exist as the metal-aqua complex .
Key Points and Reminders
In aqueous solution, metal ions form metal-aqua complexes of the form
When comparing acidity consider the charge of the metal ion AND its size (i.e. charge density)
If asked about the acidity of a metal salt in solution, ignore the negative ion and just consider the metal ion coordinated by six water ligands e.g. treat e.g. as . The nitrate ions are spectators.
Metal-aqua solution colours to know:
Ion | Solution colour |
Blue | |
Pale green | |
Violet/purple (often appears yellow/brown due to presence of | |
Colourless |